Clear Sky Science · en
PARP inhibitors and breast cancer: from therapeutic breakthrough to resistance challenge
Why this matters for patients and families
Breast cancer is now the most common cancer in women worldwide, and many people are living longer because of better drugs. One newer class of medicines, called PARP inhibitors, has offered fresh hope to patients whose tumors carry certain genetic flaws. This article explains how these drugs work, why they can be so effective, and how cancers manage to outsmart them over time. Understanding this arms both doctors and patients with clearer expectations and points toward future treatments that might keep breast cancer under control for longer.
Targeted drugs that exploit cancer’s weak spots
Breast cancer is not a single disease, but a collection of subtypes that behave differently and require tailored care. Tumors lacking hormone or HER2 signals, such as many triple negative breast cancers, are especially aggressive and hard to treat. PARP inhibitors were developed to target tumors that already have trouble repairing their DNA, especially those with inherited or acquired changes in BRCA1 or BRCA2 genes. By blocking PARP proteins, which normally help mend everyday DNA damage, these drugs push already vulnerable cancer cells past their limit, leading to their death while sparing most healthy cells. Large clinical trials like OlympiA and EMBRACA have shown that PARP inhibitors can extend the time before disease worsens in selected patients.
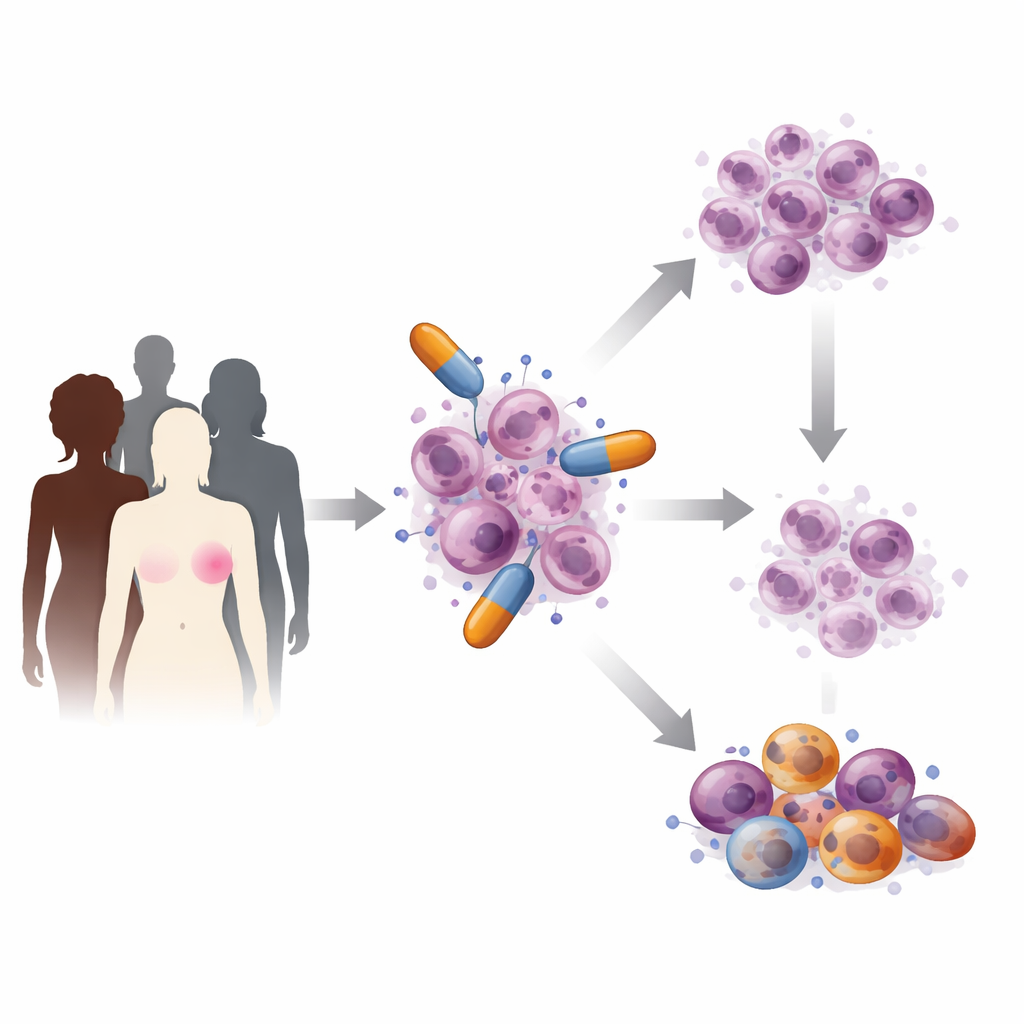
How PARP drugs attack cancer cells
Inside every cell, DNA is constantly being nicked and repaired. PARP proteins act as first responders to certain types of breaks, calling in repair crews and helping keep the copying of DNA on track. PARP inhibitor drugs slip into the active site of these proteins and either block their repair activity or cause them to stick tightly to broken DNA. As a result, small breaks accumulate and collide with the cell’s DNA copying machinery, leading to more serious damage that cells with faulty BRCA genes cannot fix. Scientists have also discovered that these drugs disturb how new DNA strands are finished, create gaps that later collapse into dangerous breaks, alter how cells handle key nutrients for chemical reactions, and even spark immune alarm signals that can help the body’s defenses recognize and attack tumors.
Evidence from trials and real-world treatment
Several PARP inhibitors are now in use or under study for breast cancer. Olaparib and talazoparib are the main options for patients with advanced HER2 negative disease and BRCA mutations, and have also been tested in earlier stages of cancer. Other drugs in this family, such as niraparib, rucaparib, veliparib, fuzuloparib and pamiparib, are being explored in different settings or regions, sometimes in combination with chemotherapy. While many trials report clear delays in tumor growth, overall survival benefits can vary, and side effects like low blood counts remain a concern. The article also notes that data from everyday practice, beyond controlled trials, are still limited, especially for newer agents and for patients from diverse backgrounds.
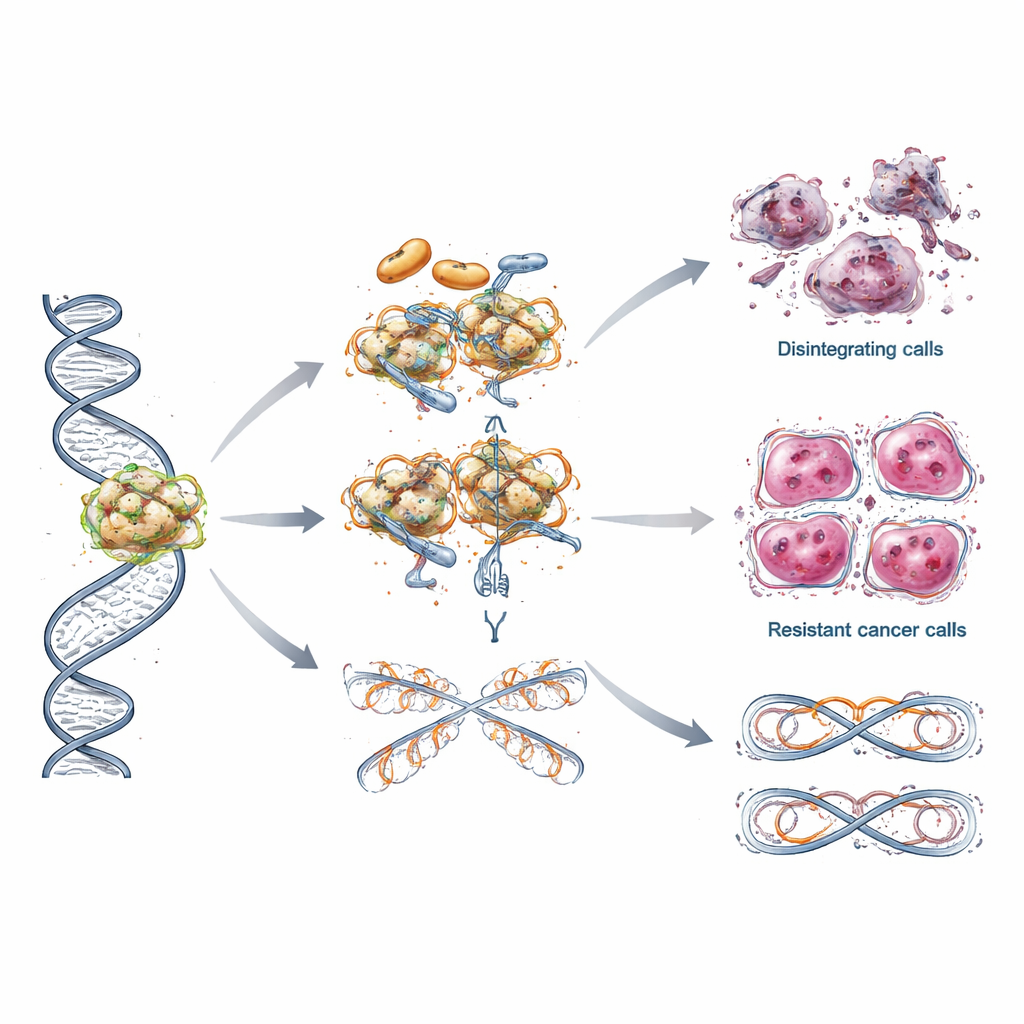
How tumors learn to resist PARP treatment
Despite early success, many patients eventually see their cancer stop responding to PARP inhibitors. The review describes several clever escape routes used by tumor cells. Some regain DNA repair ability by acquiring new mutations that restore BRCA function or by switching gene activity on again through changes in DNA tagging. Others protect the delicate DNA copying machinery so it does not collapse under stress, or adjust how DNA packaging proteins, called histones, are handled so that their chromosomes remain more stable. In some cases, cancer cells simply pump the drug out more efficiently, lowering its concentration inside the cell. These layered resistance tricks help explain why responses often fade and why the same drug may work differently from one patient to another.
Next steps: smarter combinations and wider access
To stay ahead of resistance, researchers are testing PARP inhibitors alongside chemotherapy, immune checkpoint drugs, and medicines that block other stress response routes inside cancer cells. The hope is that well chosen combinations, guided by genetic markers, will make it harder for tumors to escape while keeping side effects manageable. The authors also call for more selective next generation PARP drugs that spare healthy blood cells, better tools to predict who will benefit, and attention to the cost and availability of these treatments in low resource settings. In simple terms, PARP inhibitors have turned a genetic vulnerability in some breast cancers into a real therapeutic opportunity, but long lasting control will require understanding and blocking the many ways in which tumors fight back.
Citation: Wang, W., Cai, C., Qin, S. et al. PARP inhibitors and breast cancer: from therapeutic breakthrough to resistance challenge. Exp Mol Med 58, 981–992 (2026). https://doi.org/10.1038/s12276-026-01673-8
Keywords: PARP inhibitors, breast cancer, BRCA mutations, drug resistance, targeted therapy