Clear Sky Science · en
The contributions of TLR2, TLR8 and TLR3 to direct and antibody-dependent enhancement of dengue virus serotype 2 infection
Why this research matters
Dengue fever is a mosquito-borne disease that can range from a bad flu-like illness to life‑threatening bleeding and shock. People who catch dengue a second time are at higher risk of severe disease, especially when their existing antibodies, instead of protecting them, actually help the virus infect immune cells. This study uncovers key molecules on immune cells that make this dangerous "boosting" of infection and inflammation possible and points to new ways to prevent the most serious forms of dengue.
When helpful antibodies turn harmful
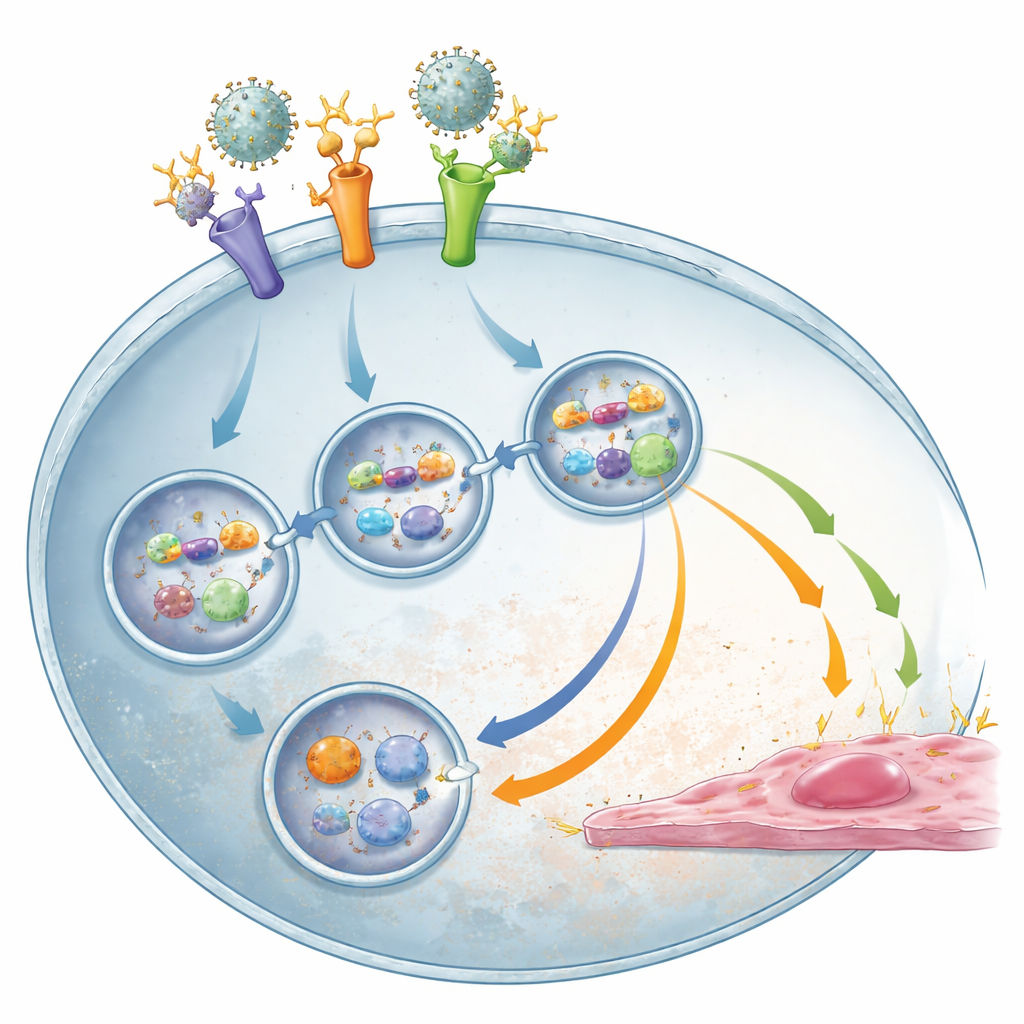
After a first dengue infection, the body keeps antibodies that can recognize the virus. During a later infection with a different dengue type, these antibodies may not fully neutralize the invader. Instead, they can coat the virus and guide it into certain white blood cells through antibody receptors, a process called antibody‑dependent enhancement. The authors used an experimental system based on human blood cells to compare this boosted infection with ordinary infection by dengue virus type 2. They focused on monocytes, a kind of immune cell that both hosts the virus and releases inflammatory substances that can damage blood vessels.
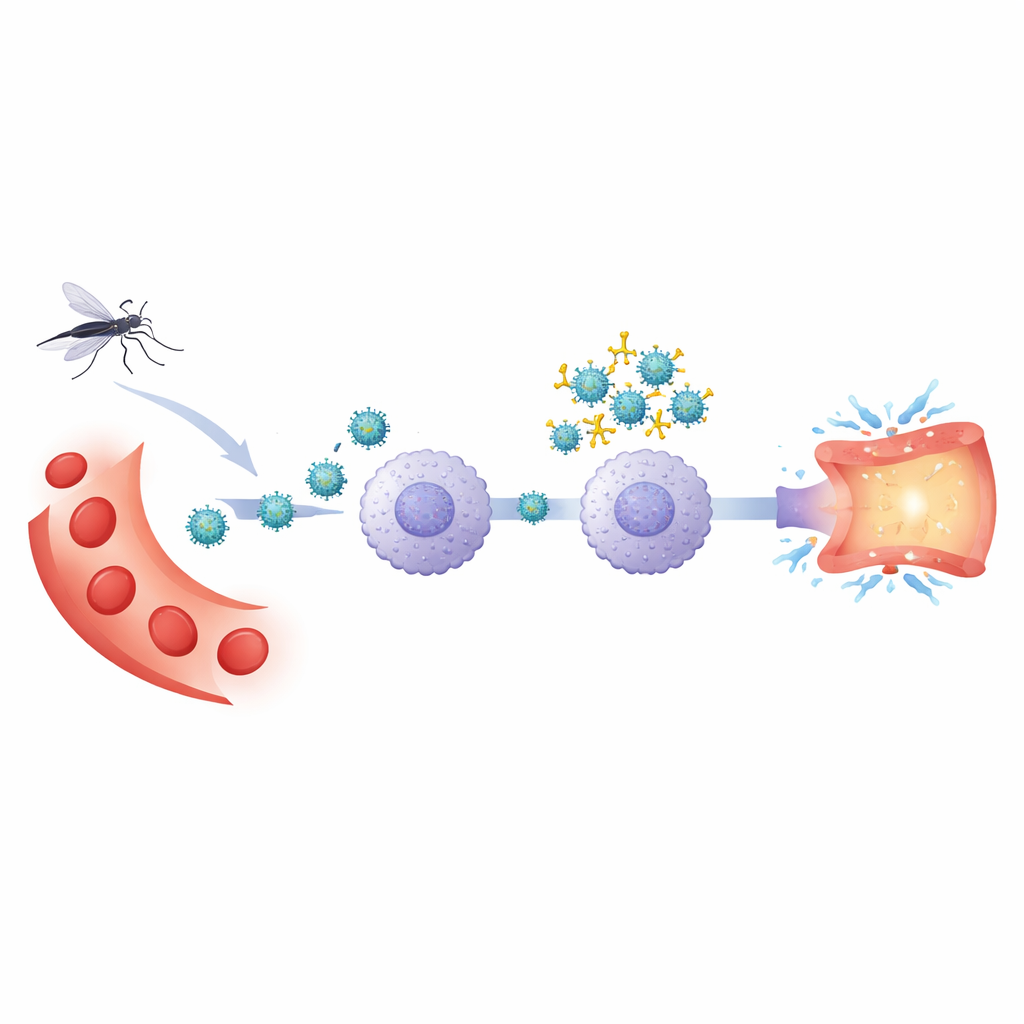
Key gatekeepers on immune cells
The team examined several receptors—molecular "docking stations"—on monocytes. One was CD32, a receptor that recognizes antibodies bound to the virus. Others were sensors that typically detect germs, including TLR2 on the cell surface and TLR3 and TLR8 inside cellular compartments, as well as a signaling enzyme called SYK that links receptors to downstream reactions. By selectively blocking these molecules with antibodies or small inhibitors, they asked which ones were truly necessary for virus entry and for the inflammatory burst that follows.
TLR2 as an essential entry helper
The experiments showed that when dengue virus entered cells with the help of antibodies, blocking CD32 or SYK reduced infection back to the level seen without antibodies. Strikingly, blocking TLR2 went even further: it almost completely prevented infection, both with and without antibodies. This indicates that TLR2 is not just an alarm sensor but also acts as a crucial helper for the virus to get into monocytes under all conditions. Another molecule, CD14, supported this process, while other proposed co‑receptors such as TLR1, TLR6, CD36, or LILRB1 did not play a detectable role in this model.
From viral entry to vessel damage
Severe dengue is marked by leaky blood vessels. To connect events in monocytes to effects on blood vessels, the researchers collected the fluid from infected blood cell cultures and applied it to human vein endothelial cells grown in the lab. These vessel cells responded by switching on surface markers that promote inflammation and leakage, but only when they were exposed to substances released by infected monocytes, not to virus–antibody mixtures alone. Antibody‑enhanced infection made monocytes produce even more of these activating signals than direct infection. Blocking CD32, SYK, or TLR2 in the blood cells strongly reduced this endothelial activation and also lowered levels of antiviral and inflammatory molecules such as interferons and TNF‑alpha.
Hidden viral RNA sparks the storm
To find out what actually triggers the inflammatory storm, the team tested whether non‑infectious virus particles could still activate monocytes. When the viral genetic material was destroyed by ultraviolet treatment, the particles failed to induce vessel‑activating signals, even though TLR2 could still bind them. This pointed to viral RNA as the key trigger. Indeed, when internal RNA sensors TLR3 and especially TLR8 were blocked, monocytes produced much less inflammatory and antiviral substances, particularly under antibody‑enhanced conditions, without reducing how many cells were infected. This suggests a two‑step process: TLR2 first helps bring virus–antibody complexes into the cell, and once viral RNA is exposed inside, TLR3 and TLR8 detect it and drive the damaging immune response, with SYK acting as a shared signaling hub.
What this means for fighting severe dengue
Taken together, the study proposes a clear chain of events linking secondary dengue infection to severe disease. Antibodies from a prior infection can shuttle dengue virus into monocytes via CD32, but successful infection in these cells also depends on TLR2. Once inside, the virus exposes its RNA to TLR3 and TLR8, which, together with SYK, ignite a powerful inflammatory and antiviral program that activates and disrupts blood vessels. By identifying TLR2, TLR3, TLR8, and SYK as central players in both viral entry and harmful inflammation, this work highlights potential drug targets that might one day help prevent mild dengue from progressing into its most dangerous, life‑threatening forms.
Citation: ter Ellen, B.M., Punekar, M., Castillo, J.A. et al. The contributions of TLR2, TLR8 and TLR3 to direct and antibody-dependent enhancement of dengue virus serotype 2 infection. npj Viruses 4, 24 (2026). https://doi.org/10.1038/s44298-026-00190-9
Keywords: dengue virus, antibody-dependent enhancement, innate immunity, Toll-like receptors, vascular leakage