Clear Sky Science · en
Fetal-restricted hematopoietic progenitors arise from hemogenic endothelium in vitelline and umbilical arteries
How Early Blood Formation Shapes a Lifetime
Before we are born, our bodies must quickly build a working blood and immune system to keep the growing fetus alive. This article explores a newly uncovered “early generation” of blood-forming cells in mouse embryos that work intensely during fetal life but largely disappear after birth. Understanding how and where these short‑lived cells arise helps explain why children’s blood and immune systems differ from those of adults, and may shed light on diseases that begin before birth.
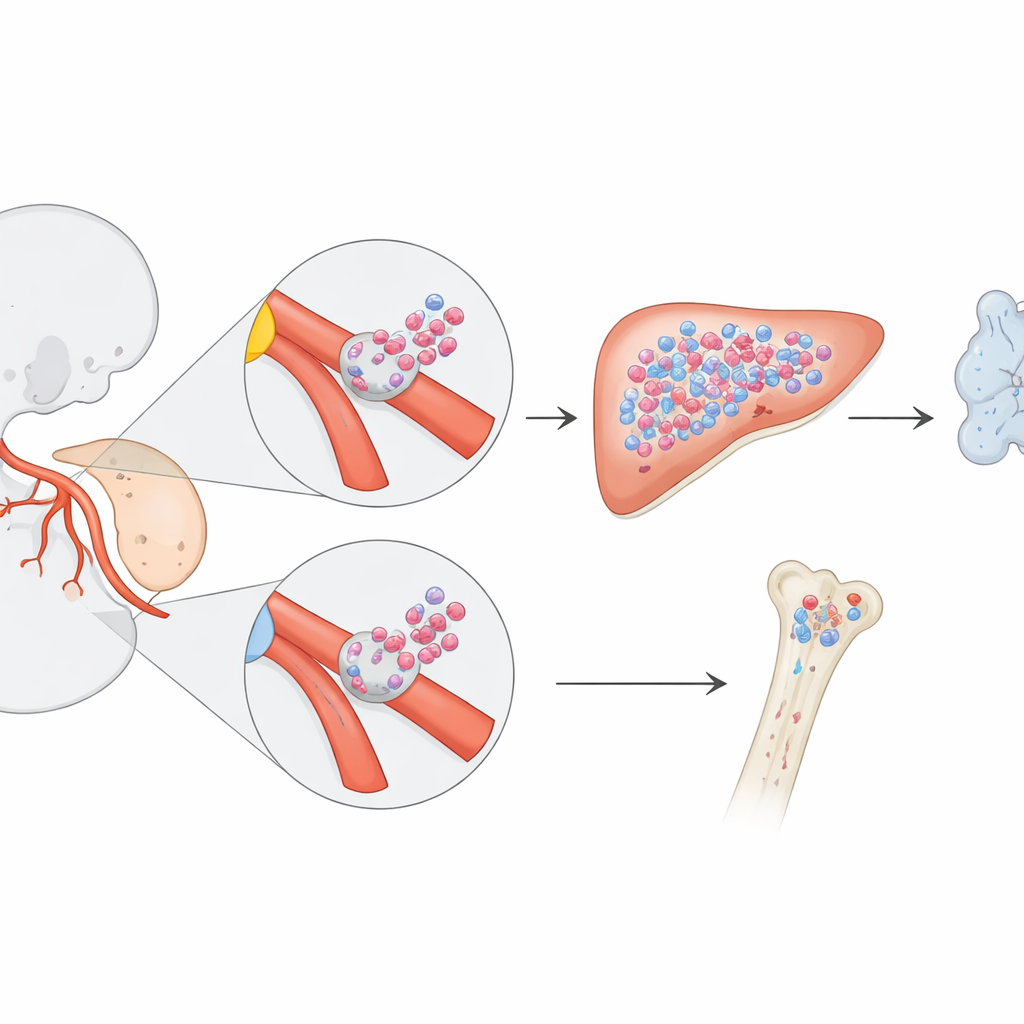
A Hidden Generation of Blood-Building Cells
Blood cells are produced in waves as an embryo develops. Earlier work showed that long‑lived stem cells, which sustain blood production throughout adult life, appear in a region near the embryonic aorta. But researchers suspected there might also be special fetal‑only progenitors—cells that build most of the blood and immune system before birth and then fade away. To track these elusive cells, the authors used genetic “time stamps” in mice. By briefly switching on a marker in specific endothelial cells—the cells that line blood vessels and can transform into blood‑forming cells—they followed the fate of their descendants from early embryonic days into adulthood.
Pinpointing Where Fetal-Only Cells Come From
By changing exactly when they activated the marker, the team could label different waves of emerging blood cells. When they tagged hemogenic endothelium on embryonic day 8.5, they saw a striking pattern: many blood and immune cells in the late‑gestation fetus carried the label, but far fewer did so after birth. Detailed imaging showed that these labeled progenitors first appeared as clusters of cells budding from the walls of two key vessels—the vitelline and umbilical arteries, which connect the embryo and yolk sac to the placenta. These clusters formed earlier than the classic long‑term stem cells in the dorsal aorta and represented a previously unrecognized source of blood-forming cells.
Feeding the Fetal Immune System, Then Bowing Out
The researchers next asked what these fetal‑restricted progenitors actually do. Using flow cytometry to examine blood and organs, they found that cells marked at day 8.5 contributed the majority of fetal B cells, many T cells, and a large portion of fetal myeloid cells—white blood cells that fight infection and help build tissues. Yet after birth, their contribution steadily declined as another wave of blood-forming cells, the classic adult‑type stem cells labeled at a later time point, took over. This “handover” revealed a layered system: an early program optimized for rapid fetal growth and immune seeding, followed by a durable adult program designed for lifelong maintenance.
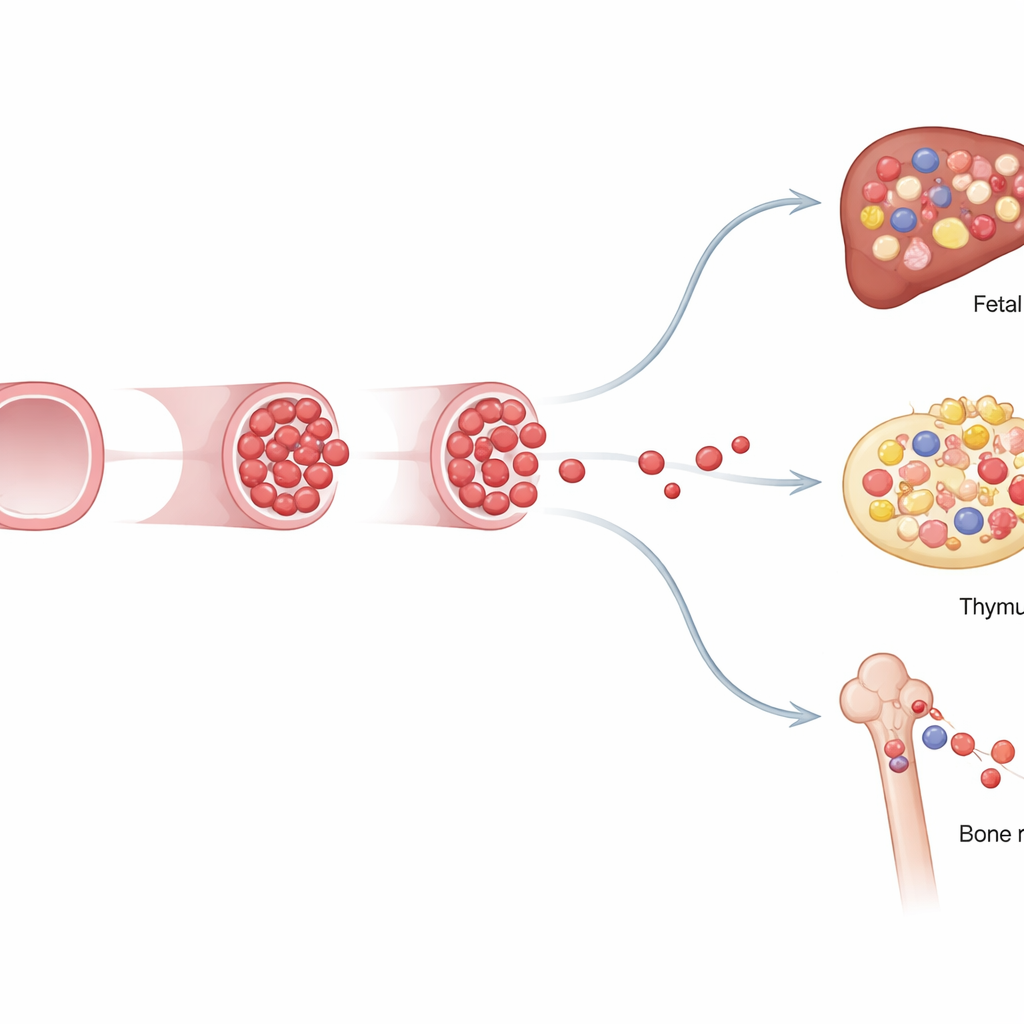
Distinct Identities and Limited Staying Power
To probe how these fetal‑restricted cells differ from adult stem cells, the team transplanted them into irradiated mice and analyzed their genetic activity one cell at a time. Transplants showed that fetal‑restricted progenitors could briefly rebuild multiple blood and immune lineages in recipients, but their contribution waned in secondary transplants, indicating little long‑term self‑renewal. Single‑cell RNA sequencing revealed that these cells carry a mixed identity: some look more like immature stem‑like precursors, while others are already primed to become specific immune cells, especially lymphocytes. Their gene activity and energy‑use patterns also differed depending on whether they came from intra‑embryonic vessels or extra‑embryonic arteries near the yolk sac, underscoring that location shapes their fate.
Why This Early Blood Wave Matters
This study shows that a short‑lived but powerful wave of blood-forming progenitors arises from specialized vessel‑lining cells in the vitelline and umbilical arteries before classical stem cells appear. These fetal‑restricted progenitors build much of the fetus’s blood and immune system, then gradually give way to adult‑type stem cells that reside in the bone marrow. For a lay reader, the key takeaway is that our blood system is not built by a single, timeless stem cell, but by successive generations of builders with different jobs and lifespans. Recognizing this early fetal wave refines our picture of how blood and immune diversity arise and may help explain why some childhood blood disorders and leukemias trace back to events that occur well before birth.
Citation: Barone, C., Quattrini, G., Muratore, A. et al. Fetal-restricted hematopoietic progenitors arise from hemogenic endothelium in vitelline and umbilical arteries. Nat Cardiovasc Res 5, 308–327 (2026). https://doi.org/10.1038/s44161-026-00793-8
Keywords: embryonic hematopoiesis, fetal blood stem cells, vitelline and umbilical arteries, hemogenic endothelium, fetal immune development