Clear Sky Science · en
Simultaneous spatial transcriptomics and morphology profiling as tools to explore how microglia change with age
Why Brain Immune Cells Matter as We Grow Older
As we age, our brains do not just lose neurons; the support and immune cells around them also change in subtle ways that can shape memory, mood and vulnerability to disease. This study looks at microglia—the brain’s resident immune cells—and asks a deceptively simple question: how does the fine wiring of these cells, and the way their genetic messages are positioned inside them, shift with age? By combining powerful imaging and gene-reading tools, the authors show that where key molecules sit inside a microglial cell can influence its shape and behavior, and that this spatial organization is remodeled over the lifespan.
Looking Inside the Aging Brain
To explore these questions, the researchers examined brain slices from young adult mice (roughly early adulthood in human terms) and very old mice. They used an imaging method called MERFISH to detect hundreds of different RNA molecules—the working copies of genes—at near single-molecule resolution, while simultaneously staining microglia so that each cell’s outline and branching pattern could be captured. This allowed them to map more than 3,900 individual microglia across seven brain regions, linking each cell’s detailed shape to its gene activity and to the exact locations of transcripts within its thick central body and its thin, tree-like processes. 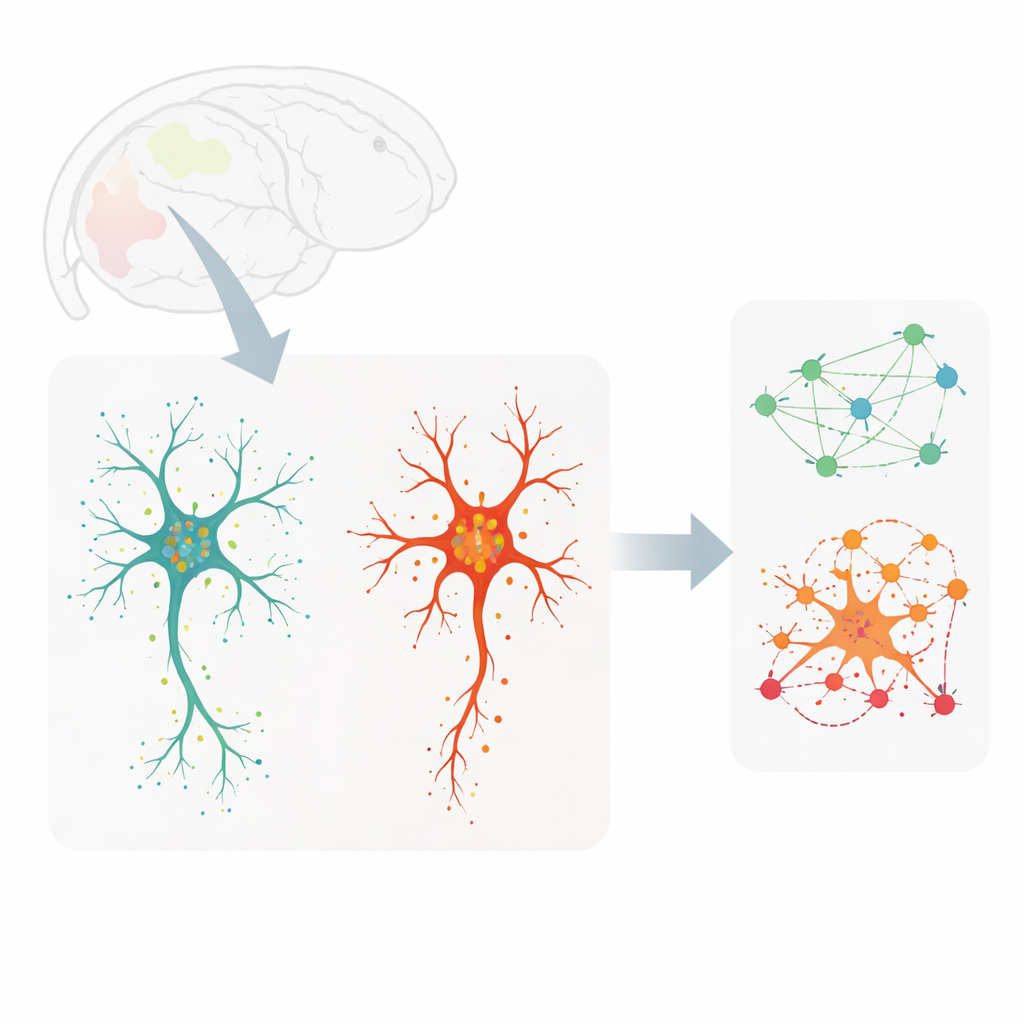
Shapes and States Do Not Always Match
Traditionally, microglia have been divided into two broad camps: ramified cells with many branches that quietly survey the brain, and rounder, amoeboid cells thought to be in an “activated” or disease-associated state. The new maps complicate that story. When the team clustered microglia by their gene-expression profiles, they found three main groups: homeostatic, transitioning and disease-associated-like cells. Separately, when they clustered cells purely by shape using a neural network and dozens of geometry measurements, they uncovered a smooth spectrum from least to most branched. Comparing the two revealed a surprising mismatch. Some disease-associated-like cells retained extensive branching, while some ostensibly “resting” cells were more amoeboid. This means that one cannot reliably infer what a microglial cell is doing from its outline alone.
Genetic Messages That Track with Branching
To dig deeper, the authors examined how specific genes related to microglial geometry. They identified sets of genes whose activity correlated strongly with features such as cell area, the number of branch tips and the complexity of branching patterns. Notably, several genes involved in handling the neurotransmitter glutamate, including the transporter gene Slc1a2, were more active in highly branched cells and showed RNA molecules extending along the processes. Other genes tied to the cell body, such as those linked to internal vesicles and ingestion of material, tracked with soma size and compactness. By focusing on transcripts that were enriched either in the central body or in the processes, the team showed that process-localized genes were especially informative: using only these, a machine-learning model could distinguish branched from amoeboid microglia with good accuracy. 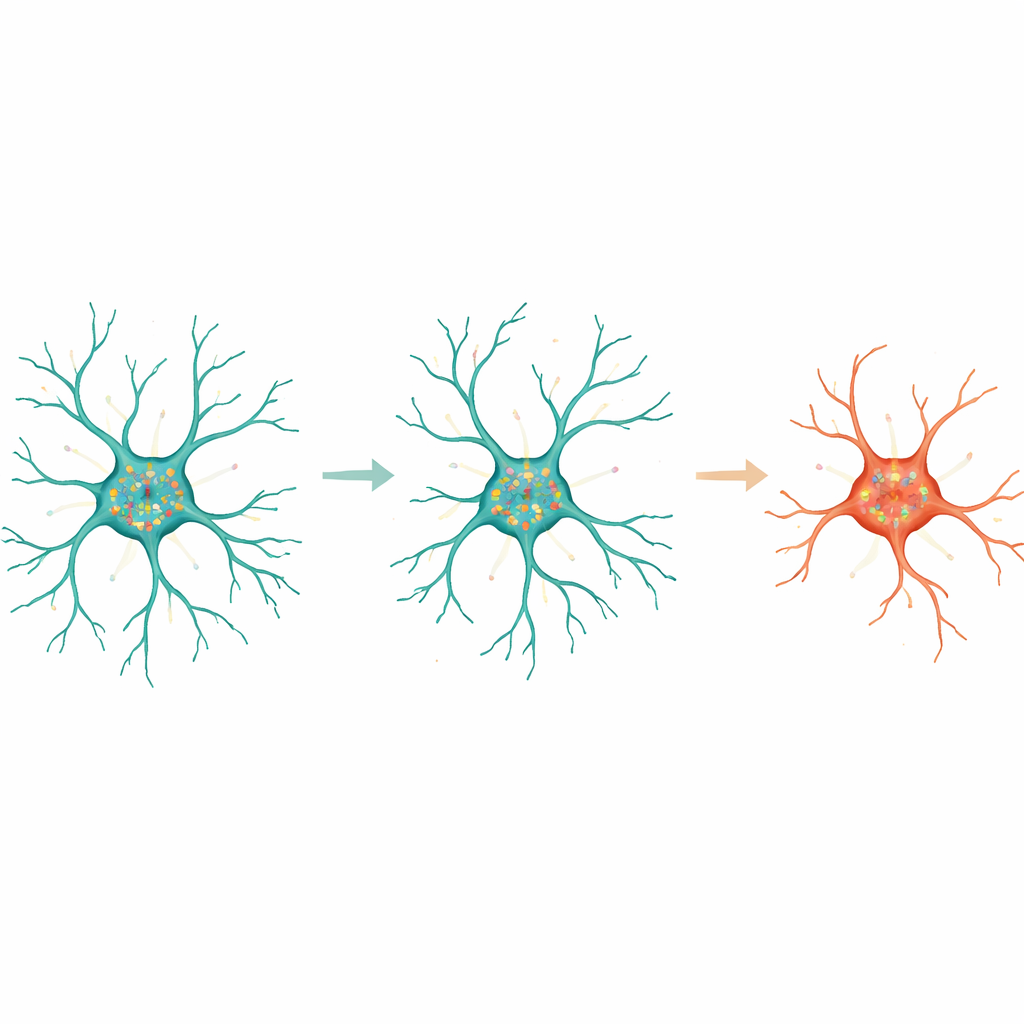
How Aging Rearranges Internal Organization
Age added another twist. In ramified microglia from young mice, many genes showed clear preferences for either the soma or the processes, and their transcripts often clustered together in patterns suggesting coordinated functions, such as managing energy use or pruning synapses. In aged mice, the number of strongly compartmentalized genes dropped, and the networks of transcripts that sat near one another became sparser and different in emphasis. In young cells, tightly co-localized RNAs in the soma tended to be linked to cytokine production and metabolic control, whereas in older cells they shifted toward migration and removal of cellular debris. In the processes, young microglia showed networks tied to local protein handling and synapse refinement, while aged microglia tilted toward uptake and breakdown of material. Despite this simplification with age, a core group of process-localized genes, including Slc1a2 and Pink1, remained key predictors of how branched a cell would be.
What This Means for Brain Health
Taken together, the work shows that the inner geography of RNA molecules inside microglia is not random: it helps define how these cells are shaped and what they do, and it is reshaped as the brain grows old. The classic picture that “activated” microglia must be round and stubby no longer holds; instead, function, shape and molecular state can be partially uncoupled. For a lay reader, the key message is that brain immune cells are far more diverse and adaptable than their textbook portraits suggest. By learning how their internal logistics—where messages are placed and with whom they cluster—change with age, scientists may eventually find ways to nudge microglia toward states that better protect the aging brain from degeneration and disease.
Citation: Henze, D.E., Tsai, A.P., Wyss-Coray, T. et al. Simultaneous spatial transcriptomics and morphology profiling as tools to explore how microglia change with age. Nat Aging 6, 869–885 (2026). https://doi.org/10.1038/s43587-026-01089-z
Keywords: microglia, brain aging, spatial transcriptomics, cell morphology, neuroinflammation