Clear Sky Science · en
Mechanistic insights into the structure and function of the RecA C-terminal tail
How bacteria manage DNA stress
Bacteria are constantly bombarded by things that damage their DNA, from antibiotics to radiation. Yet they often survive and even become harder to kill. This study looks closely at a key bacterial protein called RecA, which helps repair broken DNA and switch on an emergency program known as the SOS response. The authors focus on a small, flexible tail at one end of RecA that had long escaped structural studies, and show how this tiny piece acts as a built‑in brake and switch for DNA repair and antibiotic survival.
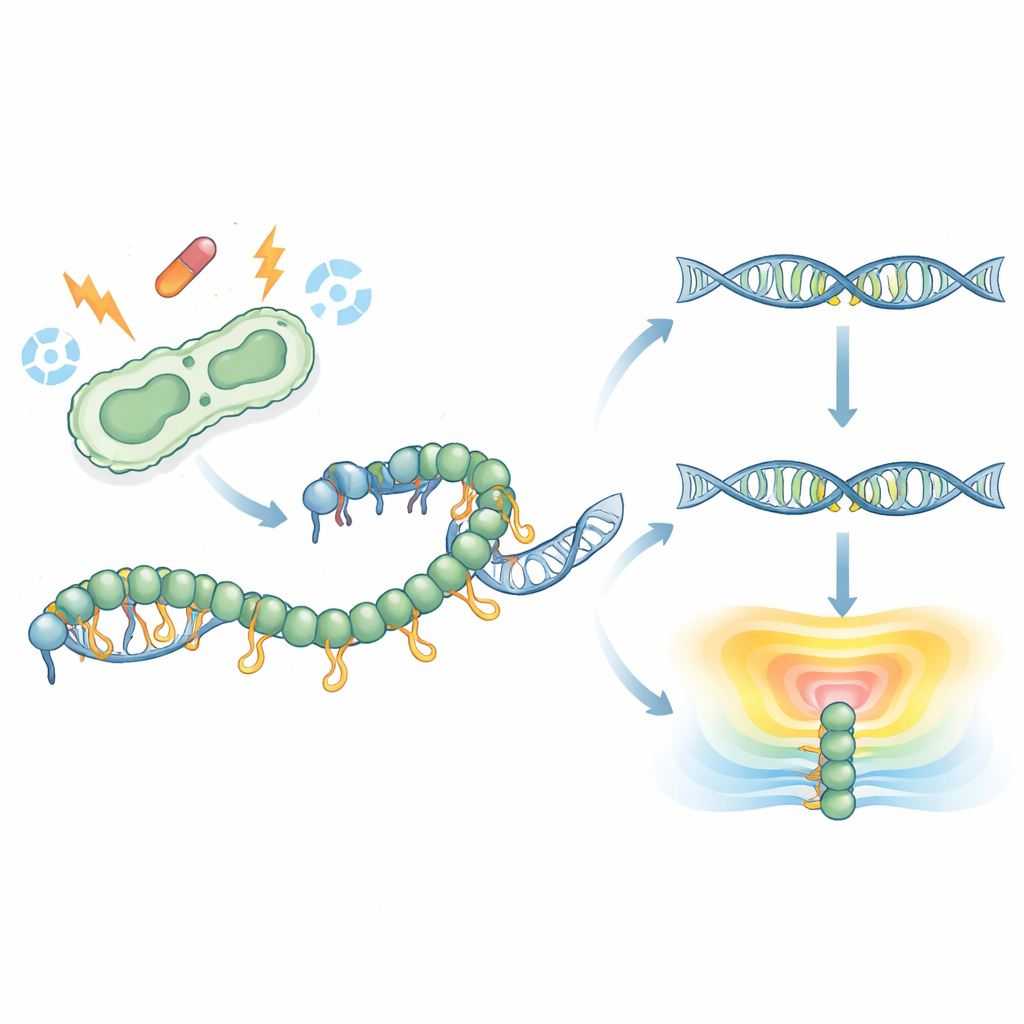
A molecular first look at a hidden tail
RecA wraps around single strands of DNA to form a filament that drives DNA repair and activates the SOS response. Earlier structures had never captured the floppy C‑terminal tail of RecA, leaving its role mysterious. Here, the researchers solved a high‑resolution crystal structure of full‑length RecA from the pathogen Klebsiella pneumoniae bound to an ATP‑like molecule. To their surprise, part of the tail folded into a short, ordered strip and reached over to contact the central motor region of a RecA molecule in a neighboring filament. This interaction joins negative charges on the tail with positive charges on the core, revealing a previously unseen way that RecA molecules can touch each other and assemble into higher‑order structures.
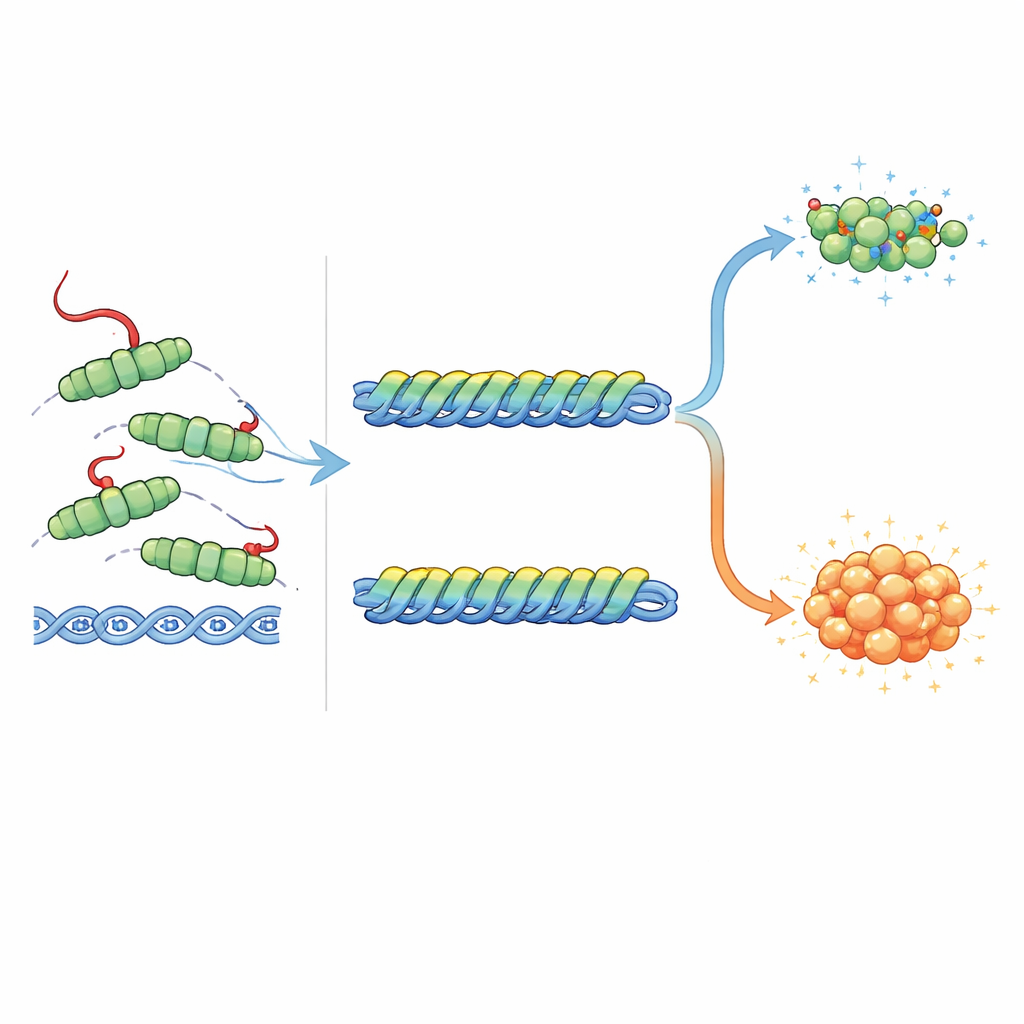
A built‑in brake on DNA binding and repair
Because the tail sits close to the DNA‑binding regions of RecA, the team tested how it affects contact with DNA. Using gel‑shift assays, they showed that RecA versions lacking the tail, or carrying charge‑altering changes in the tail, grabbed both single‑stranded and double‑stranded DNA more tightly than the normal protein. Electron microscopy confirmed that these altered proteins built longer RecA filaments on short DNA pieces than the full‑length protein did. In direct strand‑exchange experiments, which mimic the heart of homologous recombination, tail‑less or tail‑mutant RecA carried out DNA swapping more efficiently. Together, these results show that the tail normally acts as an autoinhibitory element: it makes it harder for RecA to bind DNA, extend along it, and carry out genetic strand exchange, helping to prevent runaway recombination on intact chromosomes.
Fine‑tuning the bacterial SOS alarm
RecA’s job is not only to fix DNA but also to trigger the SOS response by helping two other proteins, LexA and UmuD, cut themselves. LexA normally keeps SOS genes silent; when it is cleaved, those genes switch on. UmuD cleavage produces a partner for a special error‑prone DNA polymerase that can copy past damage but also introduces mutations. The authors found that the RecA tail has opposite effects on these two self‑cleavages. Full‑length RecA, with its intact tail, was best at promoting LexA cleavage, while tail‑less or strongly altered versions were worse. In contrast, the same tail‑less and mutant forms were better at stimulating UmuD cleavage than the normal protein. Structural comparisons suggest why: the tail can sit comfortably near bound LexA and even form favorable charge interactions, but it would clash physically and electrostatically with bound UmuD′. In other words, the tail helps turn on the SOS alarm while restraining the most mutation‑prone arm of the response.
Impact on antibiotic response in living cells
To test how this plays out inside bacteria, the researchers engineered Escherichia coli strains in which a fluorescent reporter glows when an early SOS gene called recN is activated. They then treated these strains with two DNA‑damaging drugs: mitomycin C and the widely used antibiotic ciprofloxacin. Cells carrying normal RecA showed strong SOS activation. Cells lacking the tail, however, produced much weaker fluorescent signals, consistent with reduced LexA cleavage and a blunted SOS response. Mutant tails that strengthened DNA binding and filament formation, by contrast, often drove an even stronger SOS response. These in‑cell results match the biochemical findings and highlight the tail’s role in balancing repair, survival, and mutation under antibiotic stress.
Why this small tail matters
This work reveals the RecA C‑terminal tail as a subtle but powerful regulator of bacterial genome maintenance. By contacting the protein’s core and nudging DNA and partner proteins either closer or farther away, the tail slows filament growth and DNA strand exchange, helps ensure that the SOS response is triggered appropriately through LexA, and dampens excessive mutagenic repair through UmuD. For the wider public, this matters because RecA and the SOS system influence how quickly bacteria evolve resistance to antibiotics. Understanding this tiny tail’s structural and functional tricks could, in the future, guide the design of drugs that tip the balance toward repair without runaway mutation, making existing antibiotics more effective for longer.
Citation: Su, L., Li, X., Wang, F. et al. Mechanistic insights into the structure and function of the RecA C-terminal tail. Commun Biol 9, 526 (2026). https://doi.org/10.1038/s42003-026-09788-4
Keywords: RecA protein, SOS response, DNA repair, antibiotic resistance, bacterial genome stability