Clear Sky Science · en
Sex-dependent rescue of memory and synaptic deficits in AD model mice by increasing PSD-95 palmitoylation
Why this matters for everyday brain health
Alzheimer’s disease does not affect everyone equally: women are more likely than men to develop memory loss, and their symptoms often worsen faster. This study explores one possible reason why—and tests a drug strategy that directly protects the connections between nerve cells, rather than targeting the classic brain plaques. The work, done in a widely used mouse model of Alzheimer’s, suggests that shoring up fragile synapses in the memory center of the brain could selectively rescue memory in females who are at higher risk.
A tiny scaffold at the heart of memory
Our memories rely on trillions of synapses—microscopic contact points where neurons talk to each other. At each excitatory synapse, a scaffold protein called PSD-95 helps anchor receptors that receive chemical signals. For PSD-95 to stay in the right place, it must be coated with fatty chains in a reversible chemical process called palmitoylation. Earlier work showed that PSD-95 levels fall early in Alzheimer’s disease and that palmitoylation of many brain proteins is altered. The authors asked whether changes in PSD-95 palmitoylation might differ between males and females, and whether restoring this modification could protect synapses and memory.
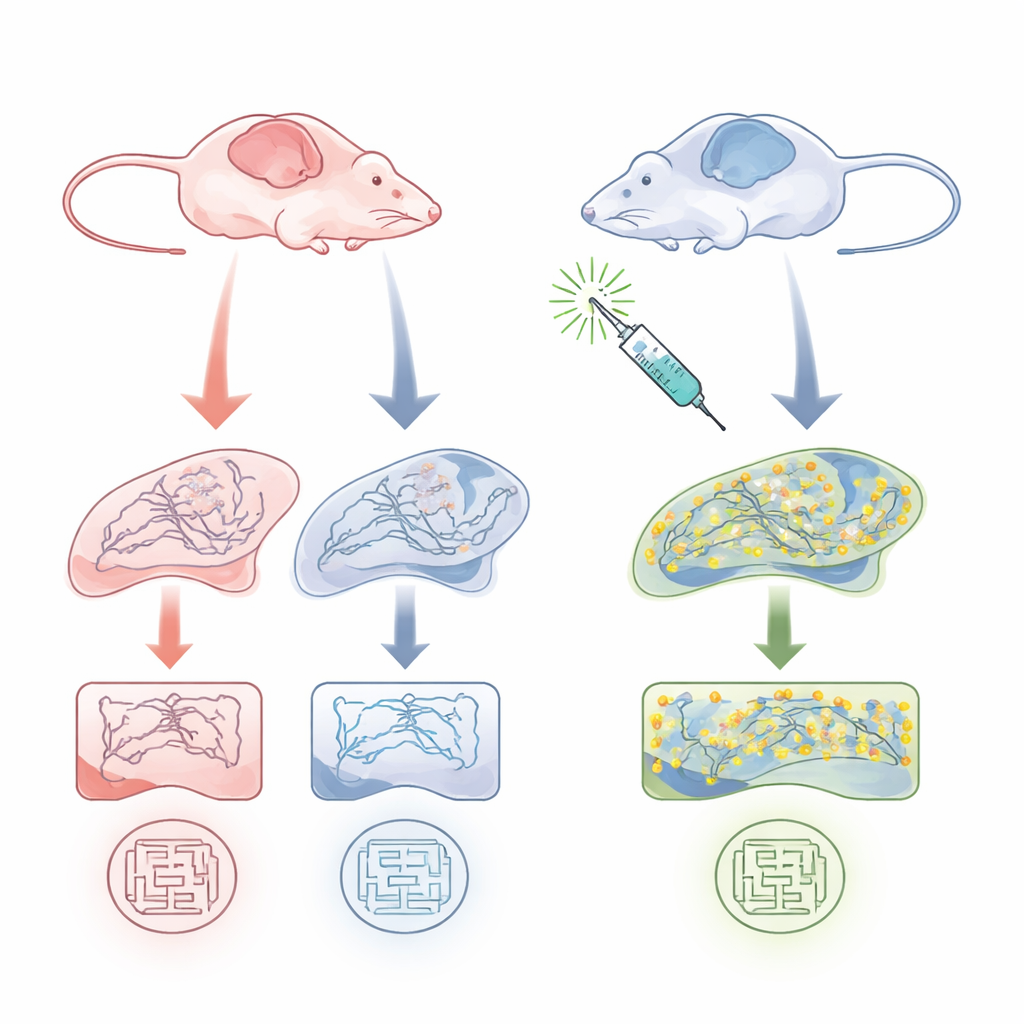
Female brains show a hidden weakness
The team studied 9–10-month-old APP/PS1 mice, a standard Alzheimer’s model that develops amyloid plaques and memory problems, and compared them with healthy littermates. They focused on the hippocampus, a key memory region. Using biochemical assays, they found that total palmitoylation of many proteins, and especially palmitoylation of PSD-95, was markedly reduced in female Alzheimer’s-model mice but not in males. Importantly, the overall amount of PSD-95 protein was similar across groups at this age—what differed was how much of it was in the palmitoylated, synapse-stabilizing form. Behaviorally, only female model mice showed clear spatial memory deficits in a water maze task, while males performed about as well as healthy controls. This pointed to a sex-specific vulnerability linking PSD-95 palmitoylation, synapse health, and memory.
A drug that locks PSD-95 in place
To test if boosting PSD-95 palmitoylation could rescue function, the researchers used Palmostatin B, a compound that blocks enzymes that strip fatty chains from proteins and had previously been shown to enlarge PSD-95 clusters in neurons. They injected a low dose of Palmostatin B into male and female Alzheimer’s-model mice before and during water maze training. In female model mice, the treatment restored performance in the memory probe test to near-normal levels, without changing how fast the animals swam or learned to find a visible platform. Biochemical analysis showed that Palmostatin B substantially increased the fraction of PSD-95 in its palmitoylated form in the female hippocampus, while leaving total palmitoylation and PSD-95 levels largely unchanged. In males, where PSD-95 palmitoylation and behavior were already normal, the drug had little effect.
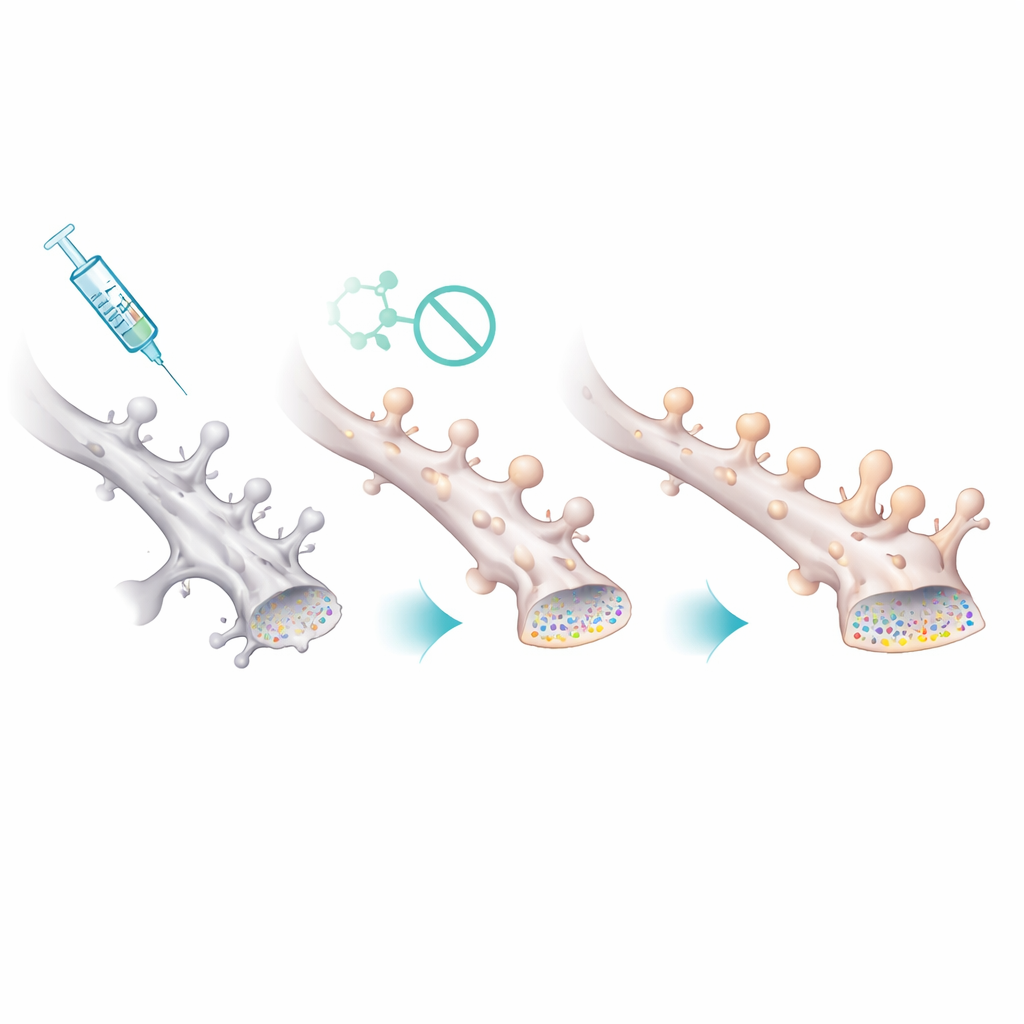
Protecting the smallest, most fragile synapses
How did this chemical tweak translate into healthier circuits? Electrophysiological recordings from hippocampal neurons revealed that female Alzheimer’s-model mice had weaker spontaneous synaptic activity, with events occurring less frequently but at slightly larger size—consistent with the loss of many small synapses and survival of fewer, larger ones. After Palmostatin B treatment, the frequency and size of these events returned toward normal. Microscopic imaging of dendritic spines—the tiny protrusions that house excitatory synapses—confirmed this picture. Female model mice had fewer spines overall, and the remaining ones were abnormally large. The drug restored spine density and shifted the distribution back toward many small, thin spines, suggesting that boosting PSD-95 palmitoylation specifically stabilizes the vulnerable, fine connections that are most important for flexible learning.
Not by clearing plaques, but by reinforcing connections
A crucial finding is what Palmostatin B did not do. The treatment did not reduce the number or size of amyloid plaques, nor did it change the activation of nearby support cells known as astrocytes. Instead, the drug altered the balance of free fatty acids and palmitoylated proteins in a sex-dependent way and improved synaptic structure and function only where PSD-95 palmitoylation was initially low. This indicates that memory rescue came from reinforcing synapses rather than from attacking classical Alzheimer’s pathology.
What this means for future Alzheimer’s therapies
For non-specialists, the key message is that Alzheimer’s can be viewed not only as a disease of plaques, but as a disease of failing connections—especially in women. This study shows that chemically “gluing” a critical synaptic scaffold into place by boosting its fatty coating can restore memory and synaptic health in female Alzheimer’s-model mice, even after substantial pathology has developed. While Palmostatin B itself is not ready for human use and affects more than one target, the work highlights PSD-95 palmitoylation and its regulators as promising leads for therapies that strengthen synapses and may one day help protect those at greatest risk.
Citation: Du, Y., Prinkey, K., Pham, A.Q. et al. Sex-dependent rescue of memory and synaptic deficits in AD model mice by increasing PSD-95 palmitoylation. Commun Biol 9, 451 (2026). https://doi.org/10.1038/s42003-026-09702-y
Keywords: Alzheimer’s disease, synapse loss, PSD-95, sex differences, palmitoylation