Clear Sky Science · en
Dynamics of a Cu/ZnO/Al2O3 catalyst revealed by operando transmission electron microscopy during CO2 hydrogenation
Turning a Climate Problem into a Useful Liquid
Carbon dioxide is often viewed only as a waste gas warming our planet, but industry can also turn it into methanol, a liquid that stores energy and serves as a building block for fuels and chemicals. This article peeks inside one of the workhorse catalysts that makes this conversion possible, watching its atoms rearrange in real time under realistic conditions to understand what keeps it fast, efficient and long‑lived.
The Workhorse of Methanol Production
For decades, factories have relied on a mixture of copper, zinc oxide and alumina to turn mixtures of hydrogen and carbon oxides into methanol on a massive scale. Engineers know that copper does the main chemical work, while zinc dramatically boosts performance, but exactly how these ingredients cooperate on the tiny surfaces where reactions happen has remained surprisingly elusive. Traditional X‑ray methods average over billions of particles, blurring out the local changes that occur on individual nanoparticles as the catalyst is heated, cooled and exposed to different gas mixtures.
Watching Nanoparticles at Work
The researchers used operando transmission electron microscopy, a technique that lets them heat the real industrial catalyst in a tiny gas‑filled cell while simultaneously imaging it at the nanoscale and monitoring the reaction products. Starting from an oxidized precursor, they tracked how tiny copper oxide and zinc oxide crystals emerge as the material is activated in hydrogen, and then how copper is gradually reduced to metallic form. At the same time, partially reduced zinc species become mobile and spread out, forming a thin skin that wets the copper nanoparticles. This nanoscale reshaping depends strongly on temperature and gas composition, and it cannot be captured by examining the catalyst only before or after the reaction.
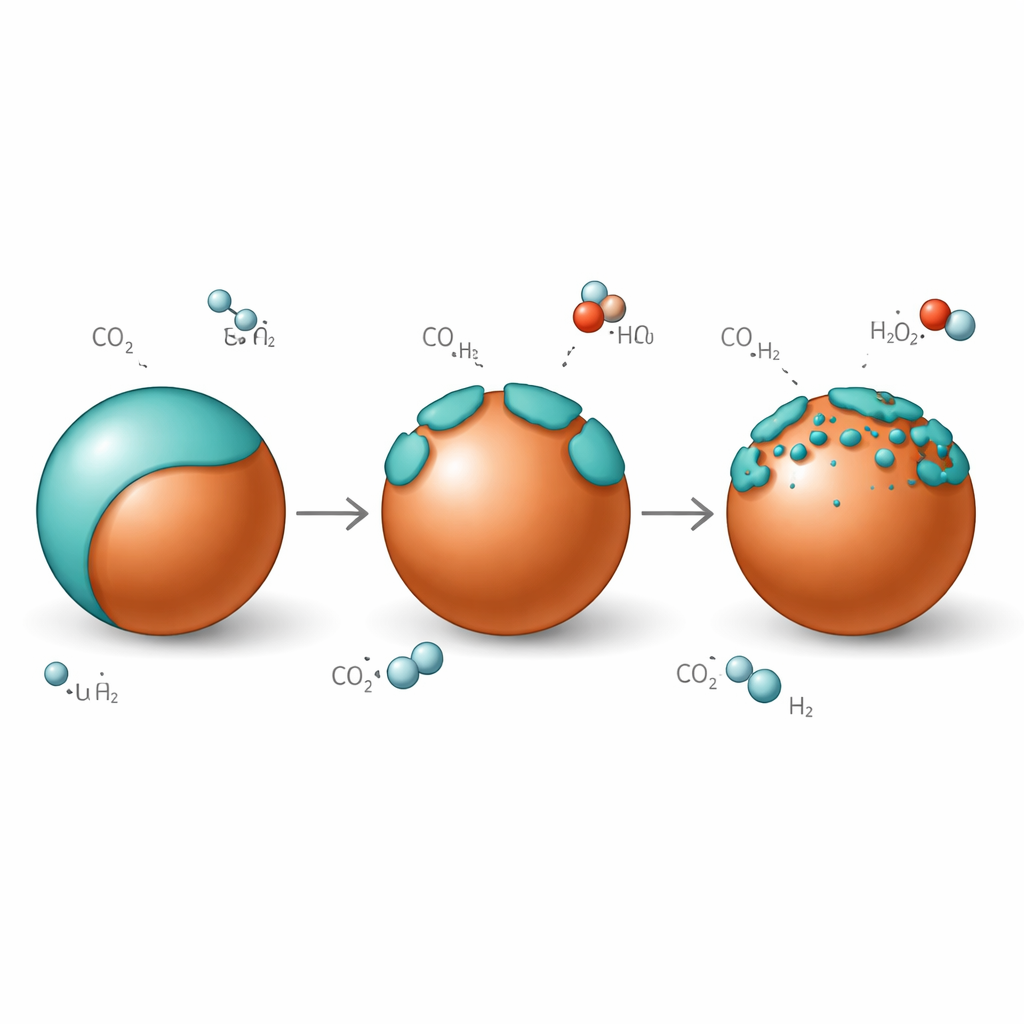
A Breathing Skin on Copper
Under reaction conditions where carbon dioxide and hydrogen flow over the catalyst, the zinc‑rich skin does not stay fixed. At low temperature, copper particles are almost fully wrapped in a graphitic zinc‑oxide overlayer. When the temperature is raised into the working range for methanol synthesis, this overlayer “opens up”: the continuous skin breaks into islands, exposing copper–zinc‑oxide edges that are especially good at activating carbon dioxide. When the system is cooled again, the overlayer closes back over the copper with nearly the same thickness, showing that this wetting is reversible rather than a one‑way degradation process. The thickness of the zinc‑rich shell is also tuned by how much carbon dioxide is present in the gas feed, with more carbon dioxide driving thicker coverage.
A Delicate Shuffle Between Alloy and Oxide
Beyond the surface skin, zinc can also dissolve into copper to form a copper–zinc alloy, another state believed to be catalytically active. By tracking tiny changes in the spacing between copper atoms via electron diffraction while simultaneously measuring water and carbon monoxide leaving the reactor, the team discovered that this alloy is transient. At higher temperatures the alloy begins to form, slightly expanding the copper lattice, but as water appears from the reverse water–gas‑shift side reaction, zinc is rapidly re‑oxidized back into zinc oxide at the surface. The catalyst thus shuttles zinc atoms back and forth between metallic and oxidized states, creating a continual cycle of alloy formation and overlayer growth and opening that depends sensitively on the local balance of hydrogen, carbon dioxide and water.
Why This Nanoscale Dance Matters
To a lay observer, the conclusion is that the catalyst works best not when it sits in a single, perfectly ordered state, but when it hovers between two: copper–zinc alloy and copper coated with zinc oxide. Reaction conditions keep pushing the system one way, while the water it produces pulls it back, creating a kind of frustrated phase transition where both states coexist and interconvert. This dynamic balance appears to generate and renew the special edge sites that are highly active for turning carbon dioxide and hydrogen into methanol. Over time, some mobile zinc locks into more stable crystalline forms that no longer participate in this dance, which may help explain why catalysts slowly lose activity. Understanding and controlling this delicate reshuffling could guide the design of longer‑lived, more efficient materials for recycling carbon dioxide into useful products and fuels.
Citation: Boniface, M., Götsch, T., Dong, J. et al. Dynamics of a Cu/ZnO/Al2O3 catalyst revealed by operando transmission electron microscopy during CO2 hydrogenation. Nat Catal 9, 404–413 (2026). https://doi.org/10.1038/s41929-026-01514-x
Keywords: CO2 hydrogenation, methanol synthesis, copper zinc catalyst, operando TEM, catalyst dynamics