Clear Sky Science · en
Renal denervation attenuates cardiac fibrosis and improves left ventricular function in rats with myocardial infarction
Why calming kidney nerves could protect the heart
Heart attacks remain a major cause of death and disability, in part because the damaged heart muscle heals by forming a stiff scar. This scarring can weaken the heart’s pumping power and lead to heart failure. The study described here explores a surprising way to limit that damage: turning down the body’s “fight-or-flight” stress wiring by cutting nerves around the kidneys, a procedure known as renal denervation. In rats, this approach appeared to soften the heart’s long-term scarring response and preserve heart function, hinting at a new avenue for treating patients after a heart attack.
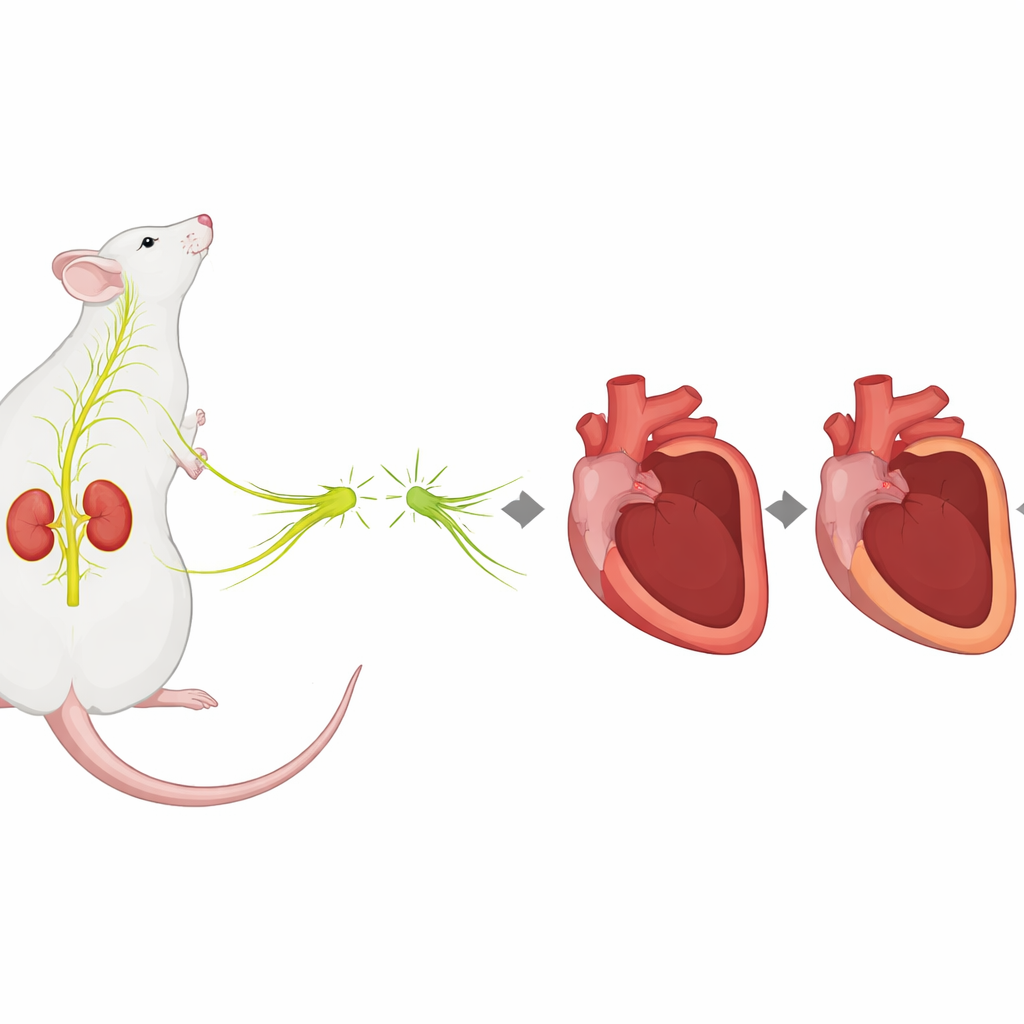
A stress pathway from kidney nerves to heart damage
After a heart attack, the body’s stress system, driven by the sympathetic nervous system, switches into high gear. This system releases chemical messengers that raise heart rate and blood pressure, but it also stirs up inflammation and helps shape how the injured heart heals. Nerves wrapped around the kidney arteries are a key hub in this stress network, sending signals that boost levels of the stress chemical norepinephrine throughout the body. Earlier work suggested that cutting or deactivating these kidney nerves can not only lower blood pressure, but also protect the heart in other disease settings. The current study asked whether performing renal denervation before a heart attack in rats could change the way the heart remodels and scars in the weeks that follow.
Testing nerve cutting in a rat heart attack model
Researchers divided rats into three groups: healthy controls, rats with an induced heart attack, and rats that underwent renal denervation two days before the same heart attack procedure. Four weeks later, they used high-resolution MRI scans to measure how well the left side of the heart pumped blood. Rats that had a heart attack without nerve cutting showed a marked drop in pumping function, while those that received renal denervation kept a much higher ejection fraction, meaning their hearts squeezed more effectively. Chemical tests of the kidneys confirmed that nerve activity and stress chemical levels were strongly reduced only in the denervated animals, showing that the procedure had successfully quieted the stress pathway.
Less scarring and quieter repair signals in the heart
To understand why heart function was preserved, the team examined the structure and molecular signals within the heart muscle. Staining heart slices for collagen, the main ingredient of scar tissue, revealed that hearts from nerve-cut rats had much smaller scar areas than those from untreated heart‑attack rats. Genetic “whole-transcriptome” profiling showed broad differences in activity of hundreds of genes. Heart attacks without nerve cutting switched on programs linked to inflammation and building of the external support mesh that becomes scar tissue. In contrast, these same gene programs were clearly dampened when kidney nerves had been cut, suggesting that the entire healing environment in the heart had shifted toward a less fibrotic, less inflamed state.
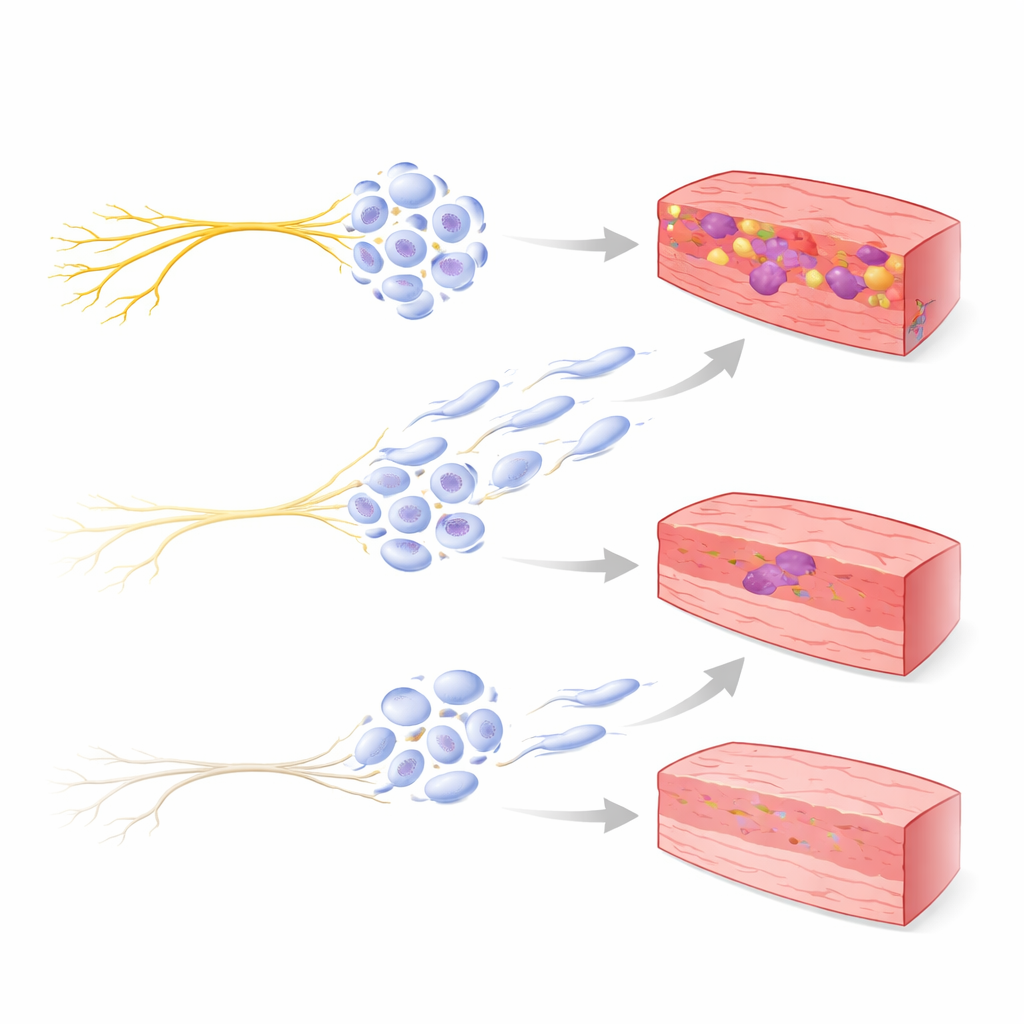
Immune cells as the link between stress signals and scarring
The study also focused on macrophages, immune cells that help clean up dead tissue and guide repair after a heart attack. In the untreated heart‑attack group, the researchers saw more macrophages and, in particular, a higher number of cells carrying a marker called CD206, which is associated with a scar‑forming, tissue‑building behavior. The more of these CD206‑positive cells present, the more fibrosis the heart showed. In the nerve‑cut group, both CD206 levels and scar burden were lower. To probe this connection, the team turned to a human cell line in the lab and exposed macrophage‑like cells to a drug that mimics stress signals acting on beta‑adrenergic receptors. These cells shifted toward a pro‑scar gene pattern, but this shift was largely blocked when a beta‑1 receptor blocker was added, pointing to a direct route from stress signaling to fibrotic immune behavior.
What this could mean for future heart attack care
Taken together, these findings suggest that dialing down kidney‑related stress nerves before a heart attack can limit long‑term damage by reducing harmful scarring and preserving heart pumping strength, at least in rats. The work highlights macrophages as key middlemen: when bathed in strong stress signals, they push the heart toward stiff, collagen‑rich scars, but when those signals are softened, they appear less likely to drive fibrosis. While renal denervation is already used in some patients with stubborn high blood pressure, its potential role after a heart attack is still untested in people. This study lays the groundwork for exploring whether targeting stress pathways and their influence on immune cells could one day become part of standard care to help hearts heal better after injury.
Citation: Therre, M., Hohl, M., Aghagolzadeh, P. et al. Renal denervation attenuates cardiac fibrosis and improves left ventricular function in rats with myocardial infarction. Sci Rep 16, 13416 (2026). https://doi.org/10.1038/s41598-026-50195-w
Keywords: renal denervation, myocardial infarction, cardiac fibrosis, macrophages, sympathetic nervous system