Clear Sky Science · en
Transfer of the synechan biosynthesis and regulatory pathway enables sulfated polysaccharide production in Synechococcus elongatus PCC 7942
Turning Sunlight into Helpful Sugars
Sulfated polysaccharides are long, sugar-based molecules with attached sulfate groups that give them remarkable properties: they can hold lots of water, form gels, and interact with our cells in ways that make them useful in medicines and cosmetics. Today, many of these compounds come from animals such as pigs and sharks, or from seaweeds, raising cost, sustainability, and ethical concerns. This study explores whether simple photosynthetic microbes—cyanobacteria—can be rewired to make these valuable molecules directly from sunlight and carbon dioxide, pointing toward more sustainable ways to produce high‑value ingredients for health and industry.
Why Special Sugars Matter
Sulfated polysaccharides already play quiet but important roles in daily life. Variants like heparin help prevent blood clots, and related molecules are added to skin creams and eye drops for their soothing and water‑binding abilities. Industrially, however, they are still mostly harvested from livestock and marine organisms, tying their supply to animal farming and fishing. Chemically attaching sulfate groups to sugars in a factory is possible but expensive, mainly because the activated sulfate donor molecule is costly. A living cell that can assemble both the sugar chain and the sulfate decorations on its own, using sunlight as its energy source, could offer a cleaner, cheaper alternative.
Cyanobacteria as Tiny Sugar Factories
Cyanobacteria are microscopic, sunlight‑powered organisms often called “blue‑green algae.” Many species naturally surround themselves with complex, slimy sugar coats that can include sulfated polysaccharides. The authors previously discovered a new sulfated exopolysaccharide, dubbed synechan, made by one model cyanobacterium, Synechocystis sp. PCC 6803. They mapped the large set of genes—called the xss cluster—that builds synechan and controls when it is produced. In the new study, they asked whether this entire genetic program could be transferred into a different, well‑studied cyanobacterium, Synechococcus elongatus PCC 7942, which does not normally make sulfated polysaccharides. If so, it would be a proof of concept that such pathways can be moved between microbes like modular toolkits. 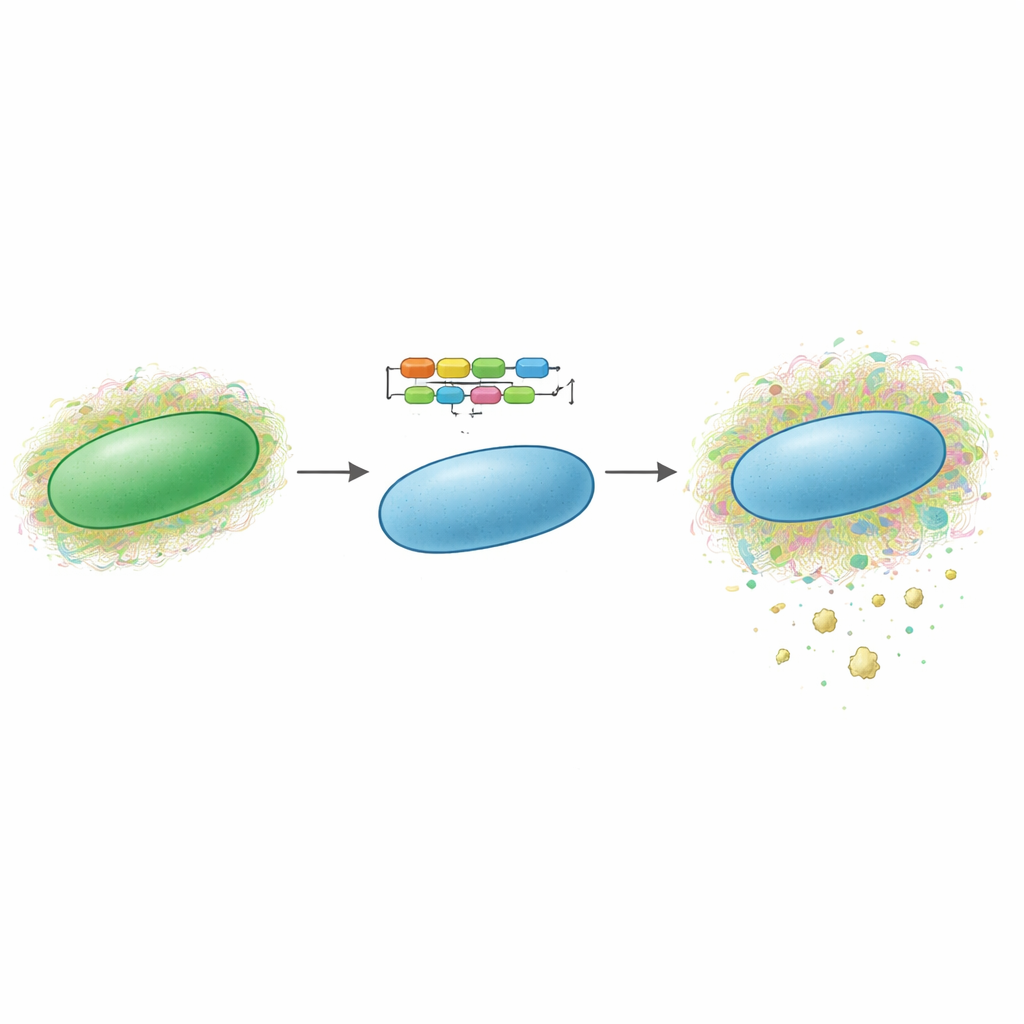
Borrowing and Installing a Complex Pathway
To achieve this, the team divided the synechan system into two main parts: the genes that build and secrete the sugar polymer, and the genes that act as on–off switches for these builders. They inserted the biosynthesis genes into a shuttle plasmid and placed one key gene under the control of a strong, inducible promoter, so that an added chemical signal (IPTG) would trigger production. They then integrated regulatory genes into a neutral spot in the host chromosome, again under inducible control. When they switched the system on, growth of the engineered strain slowed sharply and the cells began to clump together. Electron microscopy revealed long fibrous material coating the cell surfaces. Special staining that selectively highlights sulfated polysaccharides showed clear blue aggregates outside the cells, and chemical assays confirmed that both cell‑bound and released extracellular sugars increased compared with control strains.
What the New Sugar Looks Like Inside and Out
The researchers next asked whether the new material truly resembled synechan. By breaking down the polymers and analyzing their building blocks, they found that the engineered cells produced a sulfated polysaccharide made of glucose, galactose, and mannose, with a sulfate content similar to native synechan but a different ratio of sugars. Unlike synechan, which is mostly released into the surrounding liquid, much of the new polymer stayed associated with the cell surface. This suggests that, although the transferred gene set worked, it did not recreate the original structure perfectly; instead, the host’s own metabolism and transport systems shaped a “synechan‑like” product. Genome‑wide RNA sequencing showed that turning on the xss genes globally rewired cell behavior: stress‑related factors rose, photosynthetic and nutrient‑uptake genes dropped, and pathways for sugar and sulfur handling were adjusted, all consistent with cells diverting resources into making an energy‑demanding extracellular coat. 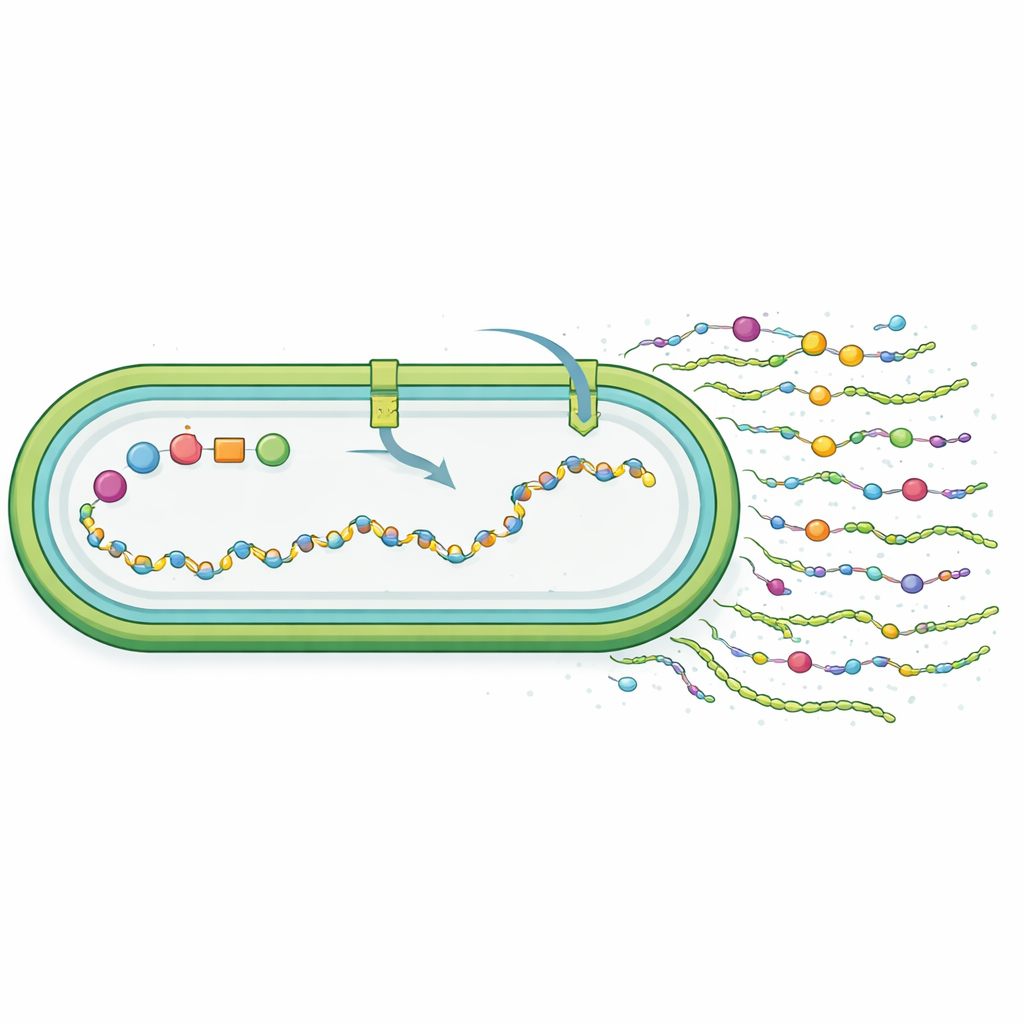
Building Toward Green Molecular Design
From a lay perspective, the central message is that scientists successfully moved a large, membrane‑bound biosynthetic machine—from one microbe into another—and got the recipient to make a new kind of sulfated sugar coating. The product is not an exact copy of the original polymer, but that difference is actually promising: it shows that the fine details of these molecules can be tuned by changing the host cell and the genetic design. In the long term, this approach could enable “solar‑powered” microbial factories that turn carbon dioxide, light, and simple nutrients into libraries of tailor‑made sulfated polysaccharides with specific textures and biological activities, reducing dependence on animal sources and opening new paths for sustainable pharmaceuticals, cosmetics, and functional foods.
Citation: Maeda, K., Ohdate, K., Sakamaki, Y. et al. Transfer of the synechan biosynthesis and regulatory pathway enables sulfated polysaccharide production in Synechococcus elongatus PCC 7942. Sci Rep 16, 13012 (2026). https://doi.org/10.1038/s41598-026-46439-4
Keywords: sulfated polysaccharides, cyanobacteria engineering, exopolysaccharides, photosynthetic biomanufacturing, synthetic biology