Clear Sky Science · en
Particle dissolution rate controls macrophage response and drug release from mesoporous silica inhalation carriers
Why tiny lung carriers matter
Breathing medicine straight into the lungs is an attractive way to treat diseases like tuberculosis while avoiding many whole-body side effects. This study explores a new class of tiny porous silica particles designed to act as "dual-scale" carriers: they enter the lungs as microscopic grains but then dissolve into much smaller nanoparticles in lung fluid. The researchers asked two practical questions: how safe are these particles for key immune cells in the lungs, and how does the speed at which they dissolve control both cell responses and how a tuberculosis drug is released over time?
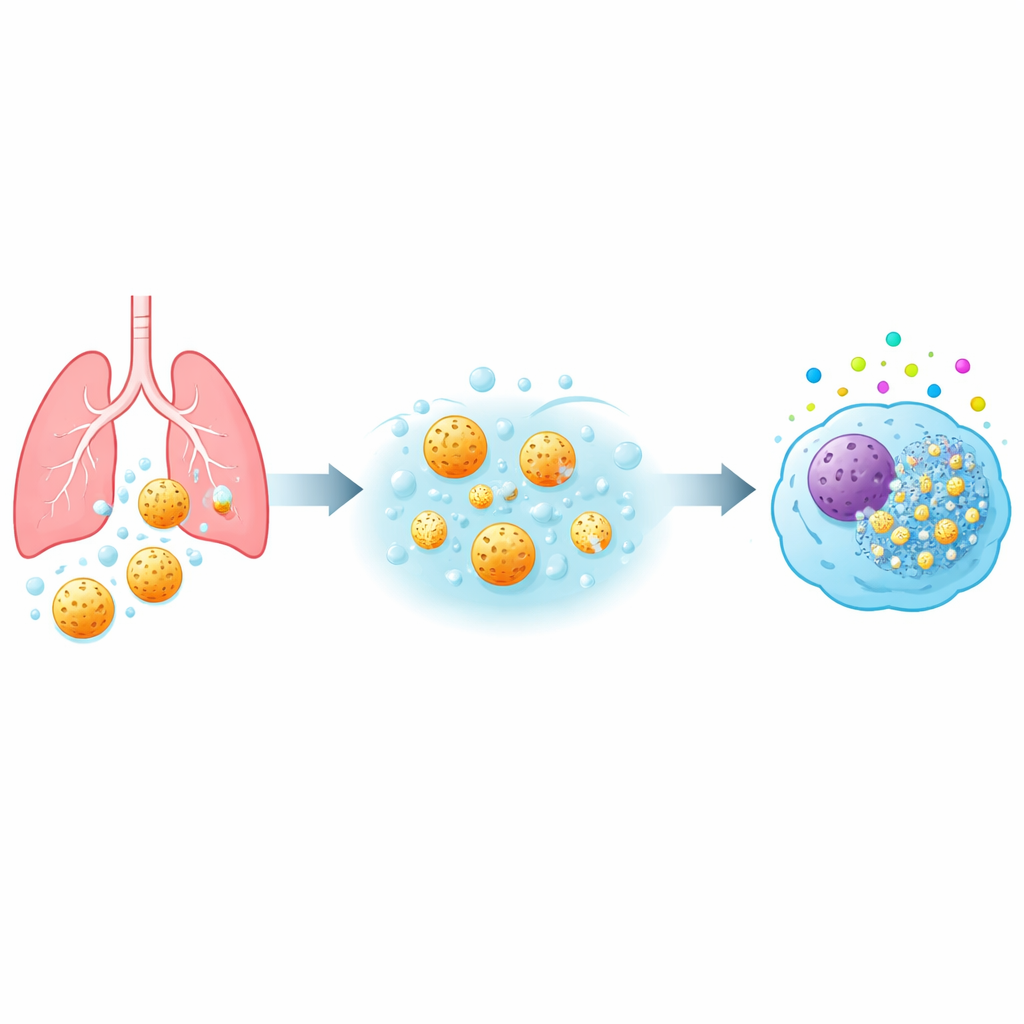
From airways to immune cells
When any dust or medicine reaches the deep lung, it first encounters alveolar macrophages—immune cells that patrol the air sacs and swallow foreign material. The team studied three types of mesoporous silica particles (called MSP-I, MSP-II, and MSP-III) that were the same overall size but had different internal pore structures and, crucially, different dissolution speeds in lung-like fluid. They exposed several human macrophage models, including primary cells from blood donors and a standard lab cell line, to a wide range of particle doses and contact times. By measuring mitochondrial activity, membrane damage, inflammatory signals, and oxidative stress, they built a detailed safety profile for each particle type.
How fast dissolving particles shape safety
Across all macrophage types, short 4-hour exposures to the particles, even at relatively high doses, kept cell mitochondrial activity above the 50% threshold often used to flag serious damage. None of the particles triggered release of the key inflammatory signal TNF-α, suggesting that they did not provoke a strong immune alarm under these conditions. However, when the particles were placed in simulated lung fluid that makes them slowly break down, an important pattern appeared: as the microparticles eroded and created clouds of nanoparticles, toxicity increased. Faster-dissolving particles were more harmful at early time points, but after 24 hours all three types converged to similar toxicity levels, closely tracking how long each particle needed to dissolve halfway. In other words, the dissolution rate effectively set the timing of peak cellular stress.
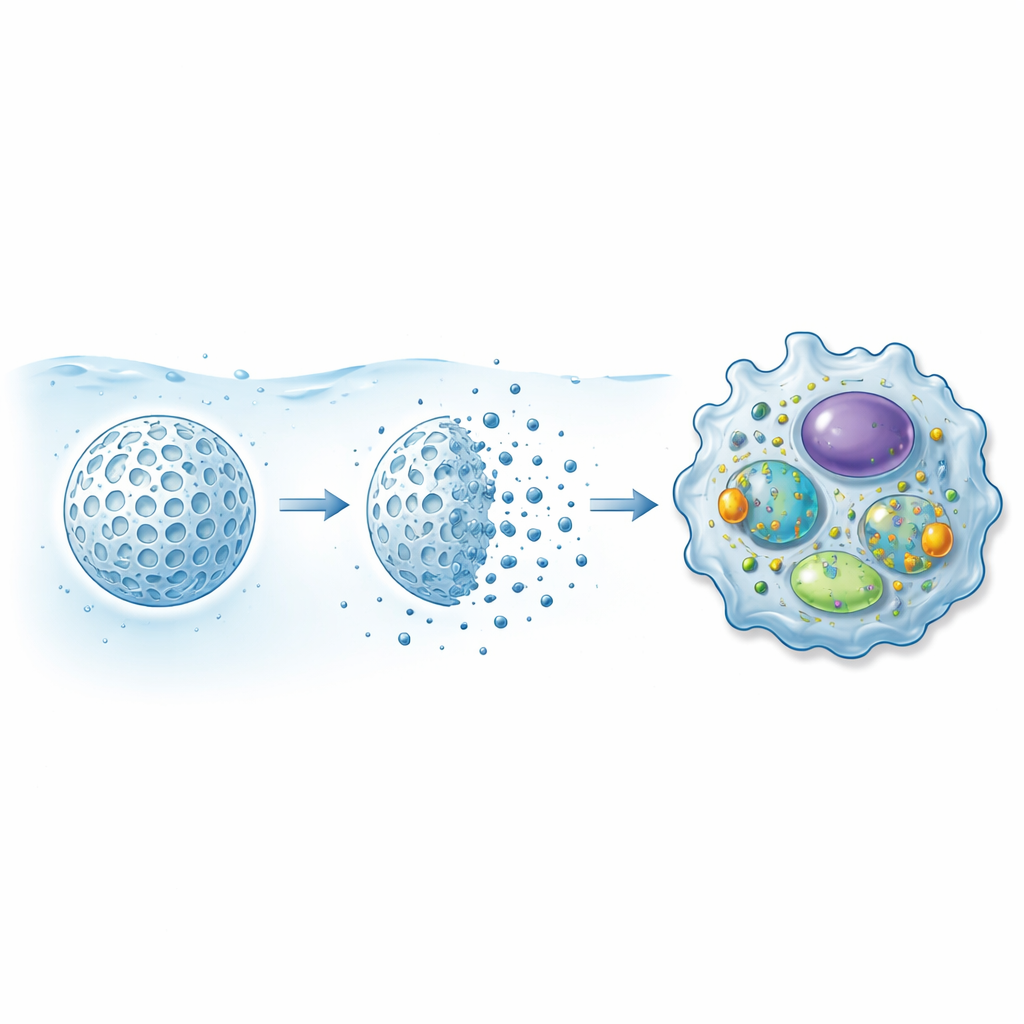
Shielding particles with lung proteins
Real lungs are not just salt water—they are rich in proteins such as albumin, the most abundant protein in lung fluid. The researchers mimicked this by adding serum proteins or purified albumin to the lung-like fluid. This protein shell, often called a "protein corona," softened the impact of the silica carriers on macrophages, especially at lower doses. At the lowest observed adverse effect level of 0.06 mg/mL, albumin was able to raise mitochondrial activity back into a range considered non-harmful in standard guidelines. At higher doses, the protection faded, but the results show that the natural protein environment in the lung can significantly improve the apparent biocompatibility of these inhaled carriers.
Linking particle breakdown to stress signals
To dig deeper into how particle behavior inside cells leads to damage, the team measured reactive oxygen species (ROS)—chemically aggressive molecules often associated with stress pathways—and checked whether lysosomes, the acidic recycling compartments inside cells, began to leak. At the low benchmark dose of 0.06 mg/mL, ROS levels and lysosomal leakage stayed close to normal. At higher doses, especially with the fastest-dissolving particle (MSP-I), both ROS and lysosomal permeabilization increased, consistent with a scenario where rapid particle erosion and nanoparticle buildup strain cellular defenses. Still, even under these more stressful conditions, the particles did not produce strong inflammatory cytokine release, pointing to a nuanced, concentration- and time-dependent response rather than a simple "on/off" toxicity.
Delivering tuberculosis drugs over time
Beyond safety, the study examined how particle properties control the release of clofazimine, an antibiotic used against tuberculosis. Both a fast-dissolving carrier (MSP-I) and a slower one (MSP-III) were loaded with small amounts of the drug and immersed in lung-like fluid containing a lung surfactant lipid. MSP-I released the drug very quickly, generating a short-lived supersaturated solution—high drug levels that then declined as the drug likely crystallized out. MSP-III, by contrast, produced slightly slower initial release but a more stable drug concentration over many hours. In both cases, dissolved clofazimine levels exceeded the concentrations known to kill tuberculosis bacteria, but the slower particle supported a more sustained exposure that may be better suited for long-lasting lung therapies.
What this means for future inhaled medicines
To a lay reader, the main message is that how fast these porous silica carriers dissolve in lung fluid acts as a master dial for both safety and performance. At or below a dose of 0.06 mg/mL, the particles caused little mitochondrial damage, modest oxidative stress, and no clear inflammatory surge in human macrophage models, especially when a realistic lung protein environment was present. Faster-dissolving particles delivered drug more rapidly but also generated sharper, earlier stress signals, while slower-dissolving ones gave a smoother, more prolonged drug release. These insights suggest that carefully tuning particle porosity and dissolution rate can create inhaled treatments that combine efficient deep-lung deposition, intracellular drug delivery, and acceptable safety margins—a promising step toward better therapies for respiratory infections like tuberculosis.
Citation: Yalovenko, T., Campos Pacheco, J.E., Sedelius, G. et al. Particle dissolution rate controls macrophage response and drug release from mesoporous silica inhalation carriers. Sci Rep 16, 11229 (2026). https://doi.org/10.1038/s41598-026-46033-8
Keywords: pulmonary drug delivery, mesoporous silica particles, alveolar macrophages, nanoparticle dissolution, clofazimine