Clear Sky Science · en
Turn-on quinoline probe for selective sensing of hypochlorite in live cells
A New Way to See a Hidden Chemical in Our Cells
Our cells constantly produce and break down highly reactive molecules that help fight infections but can also damage our tissues when they build up. One of these, hypochlorite, is closely linked to inflammation, immune defense, and diseases tied to oxidative stress. This study describes a newly designed small dye molecule that stays dark until it meets hypochlorite, then switches on with a bright green glow. That simple light switch makes it much easier for researchers to watch this elusive chemical in real time inside living cells.
Why Tracking a Harsh Oxidant Matters
Reactive oxygen species are chemical by-products formed when our cells use oxygen. At the right levels, they help white blood cells kill microbes and act as signals that guide normal cell behavior. But when their levels get too high, they attack DNA, proteins, and fats, contributing to conditions ranging from neurodegeneration to heart disease. Hypochlorite is one such reactive species produced by immune cells, and it is unusually stable inside living tissues. Because it is both useful and potentially harmful, scientists want tools that can accurately reveal when, where, and how much hypochlorite is present in cells.
Designing a Small, Smart Light Switch Molecule
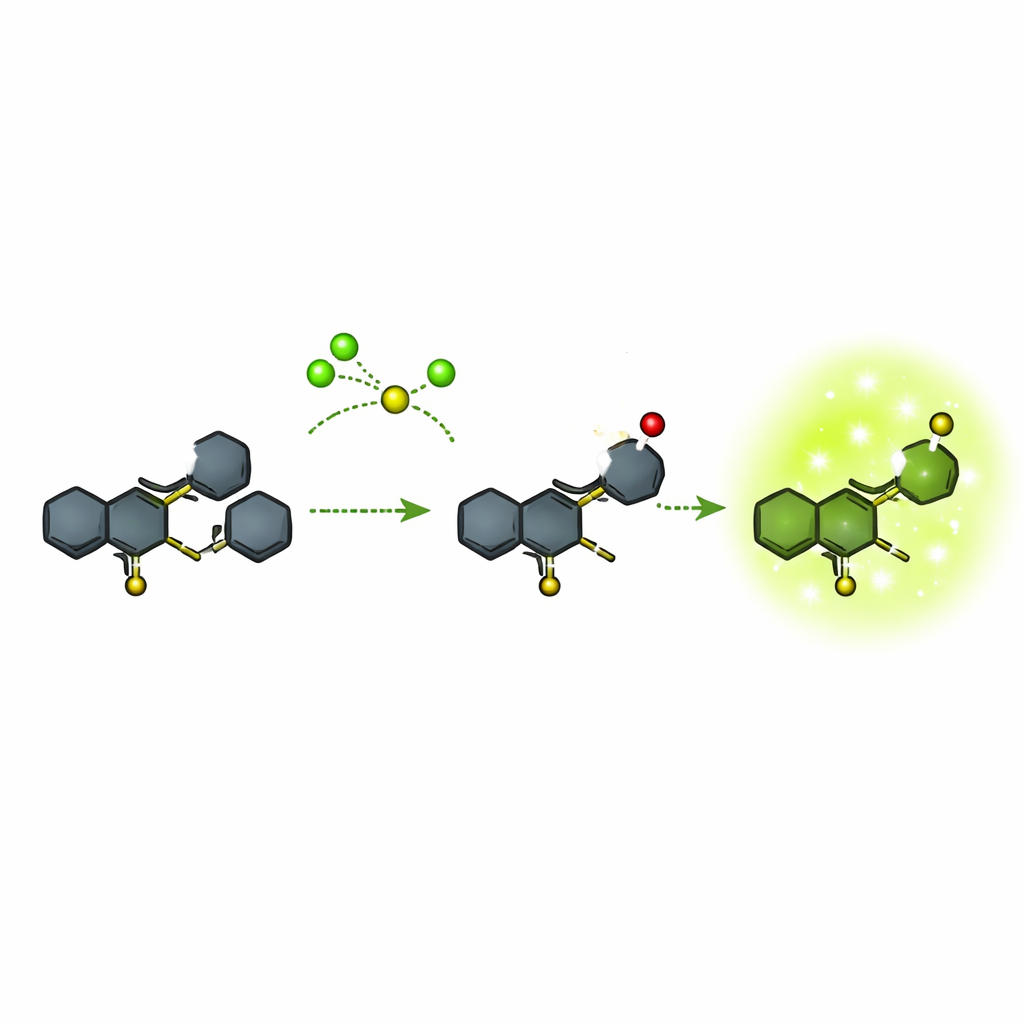
Many existing fluorescent probes for hypochlorite are relatively bulky or require complicated color changes that make precise measurements difficult. The authors focused on a compact chemical framework based on a ring system called quinoline, joined to another ring system known as phenothiazine. They built this structure in a few steps from commercially available starting materials, rigorously checking each stage with nuclear magnetic resonance, mass spectrometry, and optical measurements. The final probe, called compound 5, is almost non-glowing on its own but is designed so that a particular sulfur atom can be selectively attacked by hypochlorite. That reaction transforms the molecule into a new form (compound 6) that glows strongly in the green region of the spectrum.
From Dark to Bright in Seconds, and Only for the Right Target
The team tested how specifically and how quickly the probe responds to different chemicals. In solution, compound 5 showed essentially no fluorescence in a wide range of solvents. When exposed to many common ions, small sulfur-containing molecules, and other reactive oxygen species, it stayed dark. Only hypochlorite caused a strong, sharp green emission around 523 nanometers, and the brightness increased in step with hypochlorite concentration over a useful range. The reaction was rapid: measurable light appeared within about 10 seconds and continued to rise over minutes. At very high hypochlorite levels, the probe could be pushed one step further to an over-oxidized form (a sulfone) that was less bright, but this occurred outside the main working range. Overall, the detection limit was extremely low, well below one micromolar, and the signal was intense enough to be seen by eye.
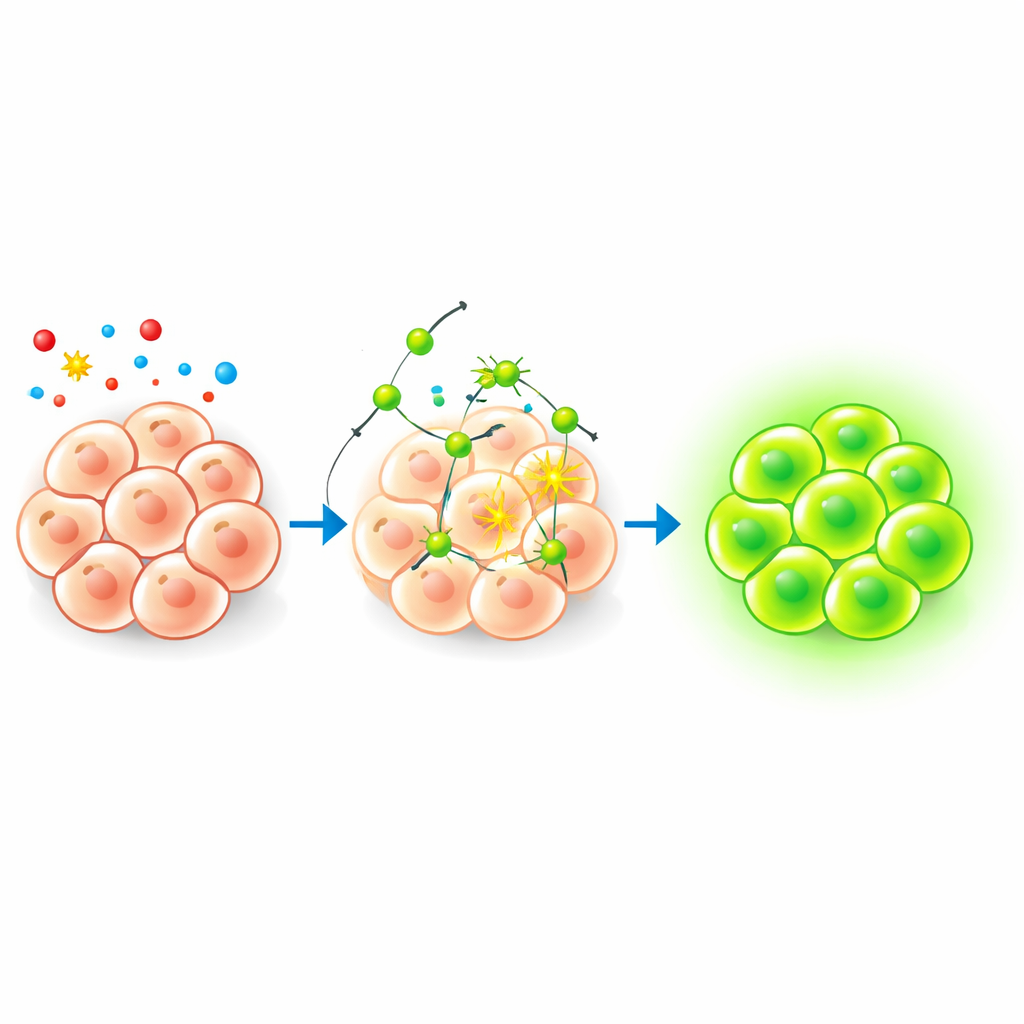
Watching Hypochlorite Inside Living Cells
To see if this chemistry worked in a biological setting, the researchers tested the probe in freshly isolated mouse bone marrow cells. At a low probe dose that did not harm the cells, they used flow cytometry and fluorescence microscopy to track its behavior. Cells treated with the probe alone showed only a slight increase in green light, suggesting that normal internal hypochlorite levels are low but detectable. When the same cells were then exposed to increasing amounts of added hypochlorite, a large fraction of cells lit up brightly, with the signal scaling with the dose. Follow-up tests showed that the glowing product was stable under continuous illumination both in solution and in cells, and that its optical properties were especially favorable in water-based, biologically relevant conditions.
What This Means for Studying Health and Disease
Together, these results introduce a compact, fast-acting, and selective "turn-on" probe for hypochlorite that operates under gentle conditions and works reliably in live cells. Rather than relying on subtle color shifts or bulky dye structures, this design uses a simple switch from dark to bright green light tied directly to a specific chemical change at one atom in the molecule. Because the probe can highlight cells experiencing elevated hypochlorite, it offers a practical tool for exploring how oxidative stress arises during infection, inflammation, and other disease states, and may guide the development of more refined sensors for other reactive chemicals in the future.
Citation: Olubiyo, F.F., Klu, S.Y.S., Burgess, R.J. et al. Turn-on quinoline probe for selective sensing of hypochlorite in live cells. Sci Rep 16, 10715 (2026). https://doi.org/10.1038/s41598-026-45327-1
Keywords: hypochlorite sensing, fluorescent probes, reactive oxygen species, cell imaging, oxidative stress