Clear Sky Science · en
The potential directing role of chemokines for specific metastatic sites in breast cancer
How Cancer Cells Choose Their Next Home
When breast cancer spreads, it rarely does so at random. Tumor cells often show up in the lungs, liver, bones, or brain, and those new growths are what most threaten a patient’s life. This study asks a simple but powerful question: what if invisible chemical “scents” in the bloodstream help guide wandering cancer cells to specific organs? By tracking a small group of these signaling molecules in patients’ blood, the researchers looked for patterns that might explain—and one day help predict or block—where breast cancer will travel next. 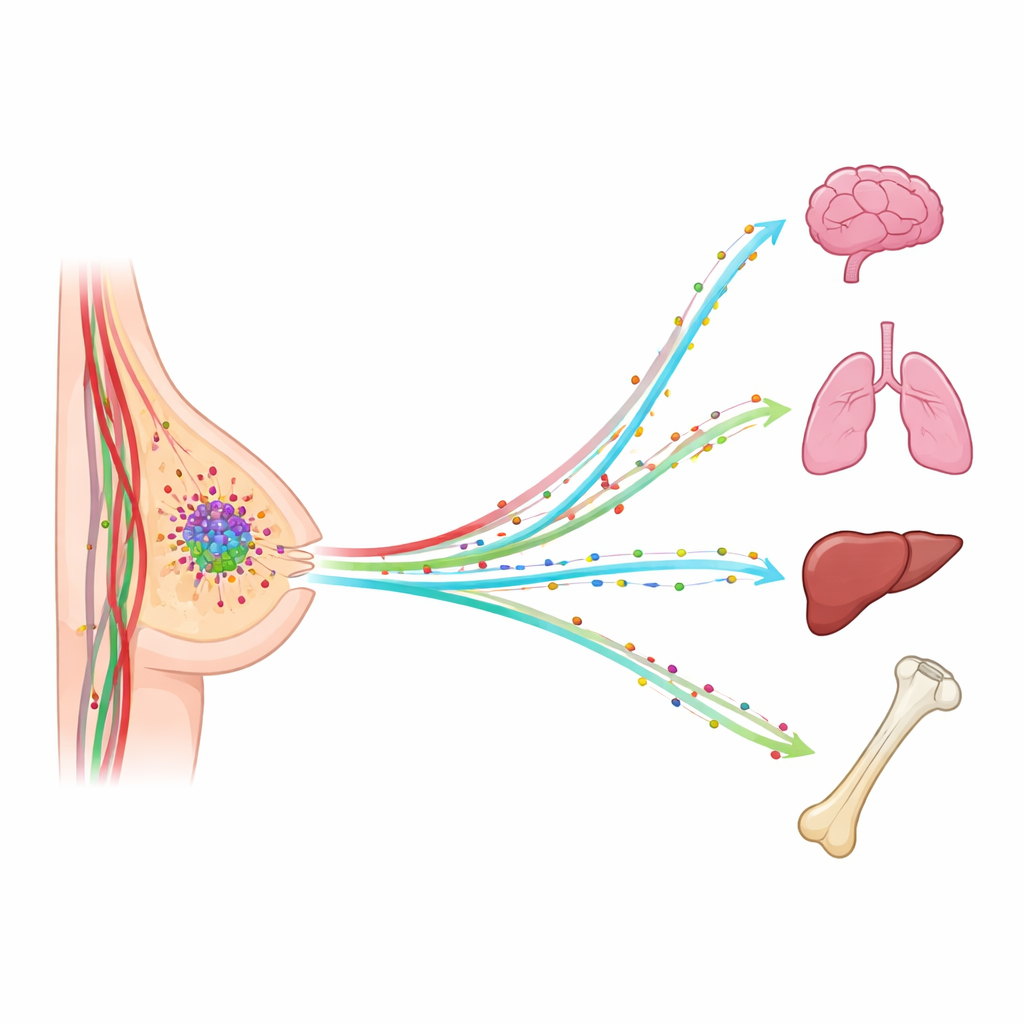
Invisible Signals That Steer Cancer Cells
Our immune system uses small proteins called cytokines and chemokines as homing beacons, telling cells where to move in the body. Tumors can hijack these signals to prepare distant organs and attract cancer cells, a bit like sending scouts ahead to set up camp. This team focused on five such molecules in the blood—IL-4, IL-11, CCL2, CCL4, and CXCL12—because they are known to influence inflammation, immune responses, and cell movement. The idea was that certain “signal profiles” might match particular metastatic sites, revealing which organs are most welcoming to incoming cancer cells.
What the Researchers Measured in Patients
The study followed 175 women with breast cancer and compared them to 50 healthy women. Some patients had disease confined to the breast area, while most had cancer that had already spread to organs such as bone, lung, liver, or brain. Using a standard laboratory test on blood samples, the researchers measured how much of each of the five molecules was circulating. They then compared levels between patients and healthy volunteers, and among patients with different tumor features, treatment responses, and sites of metastasis.
Different Signals, Different Destinations
The blood of breast cancer patients showed higher levels of IL-4, CXCL12, and CCL4 than that of healthy women, suggesting an overall “pro-metastatic” signaling environment. Beyond that, specific patterns emerged. Higher IL-11 levels were linked to metastases in the lungs. Elevated IL-4 and CXCL12 were tied to cancer that had reached the brain, especially in tumors lacking hormone receptors or carrying excess HER2, a growth-promoting protein. Bone metastases were associated with increased CCL2 and CCL4, while raised CCL2 also marked liver involvement. Statistical tests showed that these markers could moderately distinguish patients with brain, lung, or liver metastases from those without, hinting at a role in diagnosis or risk stratification. 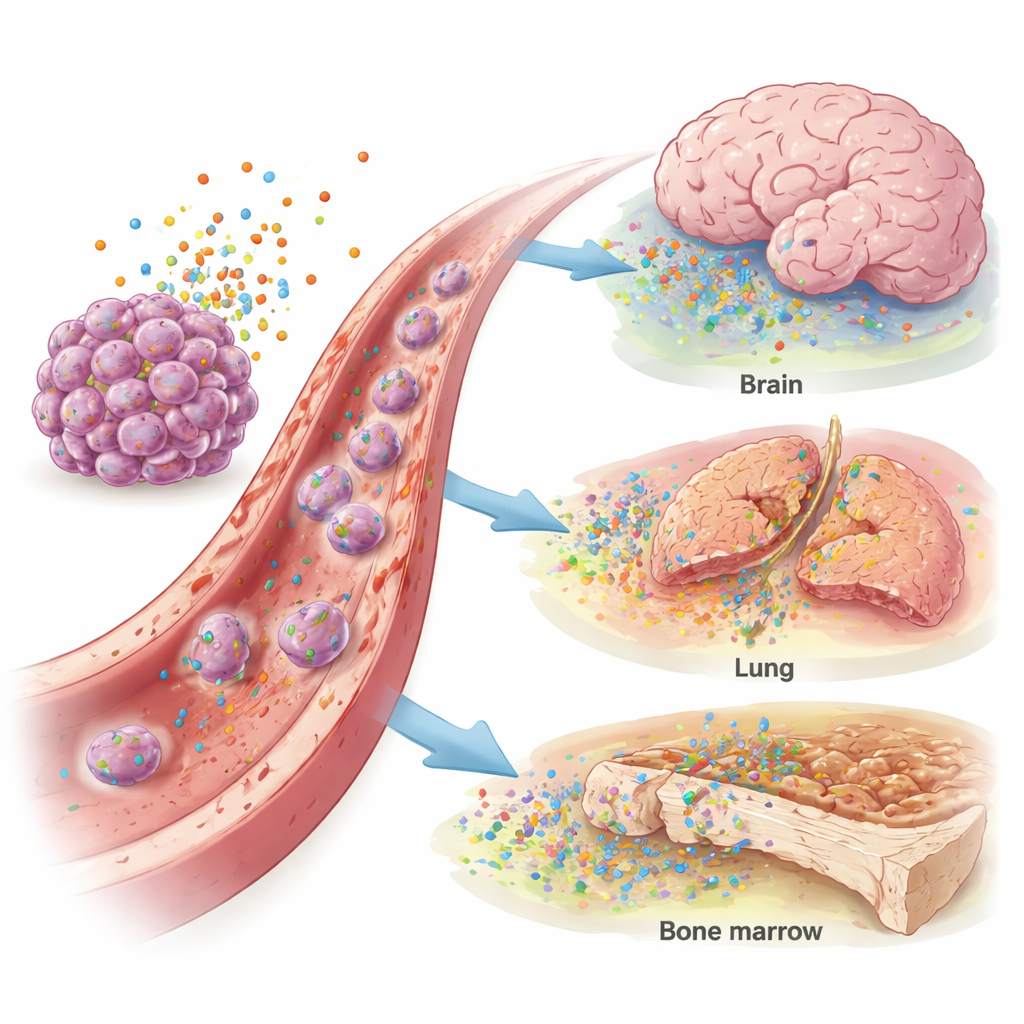
Clues to Prognosis and Treatment Response
Patients whose tumors lacked estrogen receptors, and those with spread to the brain or liver, tended to have shorter periods without the disease returning, highlighting how certain biological traits and metastatic sites go hand in hand with a worse outlook. Surprisingly, the overall levels of the five measured molecules alone did not clearly predict how long patients stayed disease-free, but higher CCL2 and CXCL12 were linked to more distant spread and poorer response to therapy. This suggests that while these signals may not be simple stand-alone prognostic tests, they are deeply involved in how tumors grow, resist treatment, and colonize specific organs.
What This Means for Patients and Future Care
For people living with breast cancer, the study’s message is that metastasis may be partly guided by recognizable chemical patterns rather than being purely random. IL-4 and CXCL12 appeared to flag a greater risk of spread to the brain, IL-11 to the lungs, and CCL2 and CCL4 to bone and liver. These findings, though based on a single, moderately sized patient group and an observational design, provide a roadmap for future research. If confirmed and refined, such blood markers could help doctors estimate where metastases are most likely to appear, monitor high-risk patients more closely, and ultimately design drugs that disrupt these “guidance cues” to keep cancer from finding new homes.
Citation: Ayoub, A.M., EL-Houseini, M.E., Tharwat, E. et al. The potential directing role of chemokines for specific metastatic sites in breast cancer. Sci Rep 16, 12015 (2026). https://doi.org/10.1038/s41598-026-45036-9
Keywords: breast cancer metastasis, chemokines, cytokines, biomarkers, tumor microenvironment