Clear Sky Science · en
Collagen-specific molecular chaperone Hsp47 in inguinal white adipose tissue promotes high-fat diet-induced inflammatory gene expression in male mice
Why fat tissue’s hidden scaffolding matters
Obesity is often described as “extra fat,” but what happens inside our fat tissue is far more complex. Fat cells live within a flexible protein scaffold, and when we eat a long-term high-fat diet, this scaffold can stiffen, inflame, and eventually damage the body. This study zeroes in on a lesser-known molecule, Hsp47, that helps build that scaffold by guiding collagen production. By studying mice on a high-fat diet, the researchers asked a simple question with big implications: does turning down Hsp47 in fat cells protect the body from diet-induced damage?
A closer look at fat depots
Not all body fat is the same. The researchers focused on two major fat depots in male mice: subcutaneous fat under the skin in the groin area (inguinal white adipose tissue, or ingWAT) and deeper belly fat surrounding organs (epididymal white adipose tissue, or epiWAT). Earlier work showed that a high-fat diet ramps up genes for collagen and other scaffold components in fat, hinting that structural remodeling is central to obesity. Hsp47 is a helper protein inside cells that ensures collagen is folded correctly before it is secreted. It is known to drive scarring in organs like the liver, but its long-term role in different fat depots under chronic overfeeding was unclear. 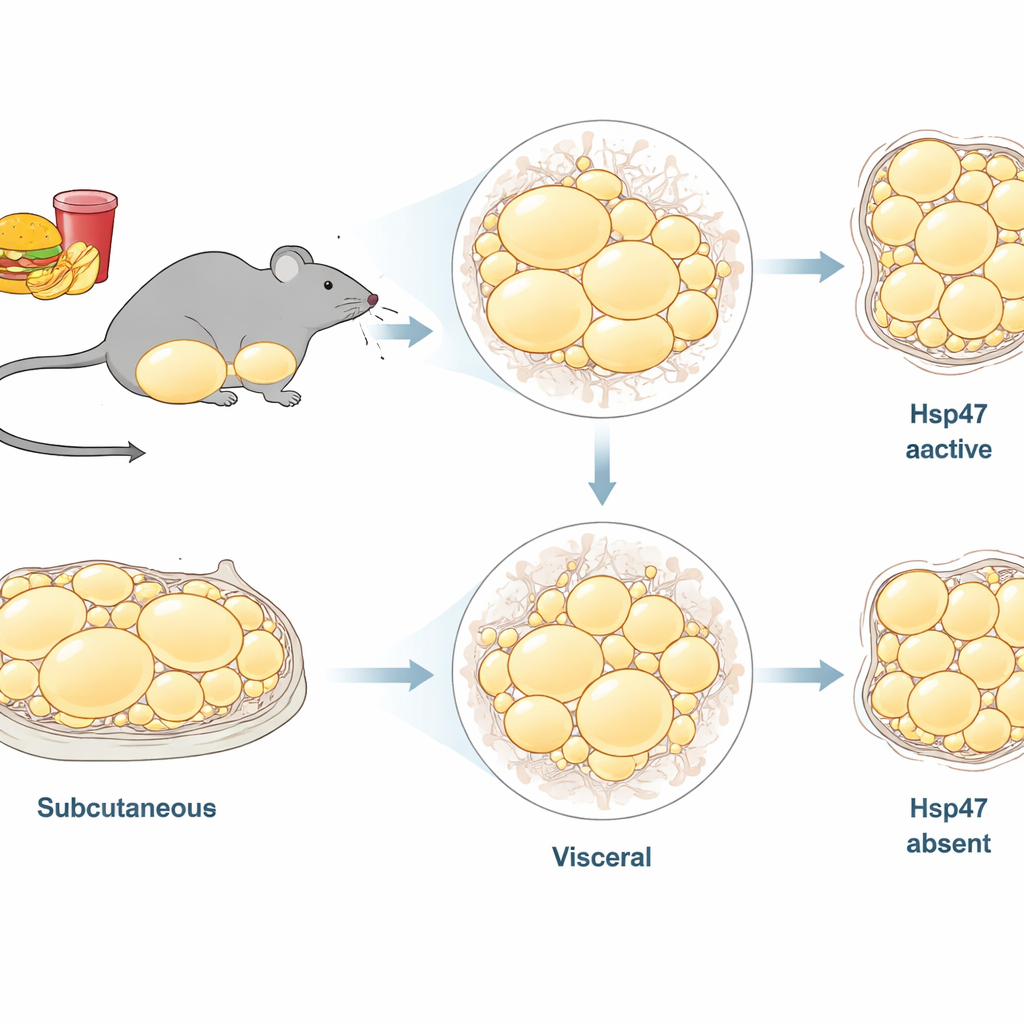
Switching off Hsp47 in fat cells
To test Hsp47’s role, the team engineered mice in which Hsp47 could be removed only from mature fat cells, leaving other tissues intact. These adipocyte-specific knockout (aKO) mice and their normal (flox) littermates were fed either a standard diet or a high-fat diet for 12 weeks, long enough to trigger early inflammation and collagen changes. Under the high-fat diet, Hsp47 levels rose specifically in fat tissues, but not in the kidney or lung. Importantly, deleting Hsp47 in fat cells did not cause signs of stress inside those cells, suggesting that fat tissue can tolerate reduced collagen chaperoning, in contrast to collagen-heavy tissues like cartilage or bone.
Where Hsp47 does the most harm
The most striking results appeared in the subcutaneous ingWAT. In normal mice, a high-fat diet made fat cells in both depots swell (hypertrophy). In aKO mice, however, ingWAT cells resisted this expansion, while epiWAT still grew larger. When the researchers stained tissue sections to visualize collagen, they found that high-fat feeding did not yet cause massive collagen buildup overall, but within ingWAT the aKO animals had less collagen-positive area than controls. Gene-expression analyses reinforced this depot-specific pattern: in ingWAT, loss of Hsp47 modestly reduced collagen genes and clearly lowered inflammatory and matrix-related genes such as interleukin-6, tumor necrosis factor, Ccl2, and fibronectin. In epiWAT, by contrast, Hsp47 deletion had little impact on these transcripts. 
From local fat changes to whole-body stress
Because fat tissue communicates with the rest of the body through hormones and inflammatory signals, the team next looked at blood and urine chemistry. As expected, high-fat feeding in control mice disturbed multiple blood markers, including glucose and mineral levels linked to early kidney strain. In the aKO mice, some of these disruptions were softened: inorganic phosphate and calcium levels shifted back toward normal, and several urine markers suggested less kidney-related stress. The data did not yet show clear structural protection in the kidney itself, but they point to a chain of events in which Hsp47-driven remodeling and inflammation in ingWAT contribute disproportionately to whole-body metabolic and mineral imbalance.
What this could mean for treating obesity
Overall, the study reveals Hsp47 as a key amplifier of unhealthy changes in the fat scaffold under a high-fat diet, particularly in subcutaneous ingWAT. When Hsp47 is active, collagen around fat cells becomes more abundant and likely stiffer, fat cells enlarge, and inflammatory genes turn on, nudging the body toward systemic trouble. When Hsp47 is removed just from fat cells, these processes are muted, without obvious problems under normal diet conditions. Because small-molecule drugs that block Hsp47 already exist in experimental form, these findings suggest a promising strategy: selectively dampening Hsp47 activity could ease harmful fat-tissue remodeling and inflammation in obesity while leaving everyday fat function largely intact.
Citation: Ito, S., Kamei, R., Kasai, A. et al. Collagen-specific molecular chaperone Hsp47 in inguinal white adipose tissue promotes high-fat diet-induced inflammatory gene expression in male mice. Sci Rep 16, 14301 (2026). https://doi.org/10.1038/s41598-026-45003-4
Keywords: obesity, adipose tissue, collagen, inflammation, metabolic disease