Clear Sky Science · en
Regulatory mechanism of cholesterol transport via LtmA in Mycobacterium smegmatis
Why this study matters
Tuberculosis remains one of the world’s deadliest infectious diseases, in part because its causative bacterium, Mycobacterium tuberculosis, is so good at surviving inside our immune cells. One of its key tricks is to feed on our cholesterol and use it to fortify its own defenses. This study, using a safe model cousin of the tuberculosis bacterium called Mycobacterium smegmatis, uncovers how a single regulatory protein helps switch cholesterol uptake on or off—offering clues to new ways of starving these microbes inside the body.
The bacterium that eats our cholesterol
When tuberculosis bacteria are swallowed up by immune cells called macrophages, they do not get destroyed as many other microbes do. Instead, they settle into a compartment inside the cell and reshape the surrounding membrane to attract cholesterol. This fatty molecule is not just a building block for the bacterial cell wall; it also becomes a major source of carbon and energy, especially during long-lasting infections when other nutrients are scarce. Specialized transporter systems in the bacterial envelope, including one called the Mce4 complex, act like molecular doors that pull cholesterol from the host environment into the microbe.
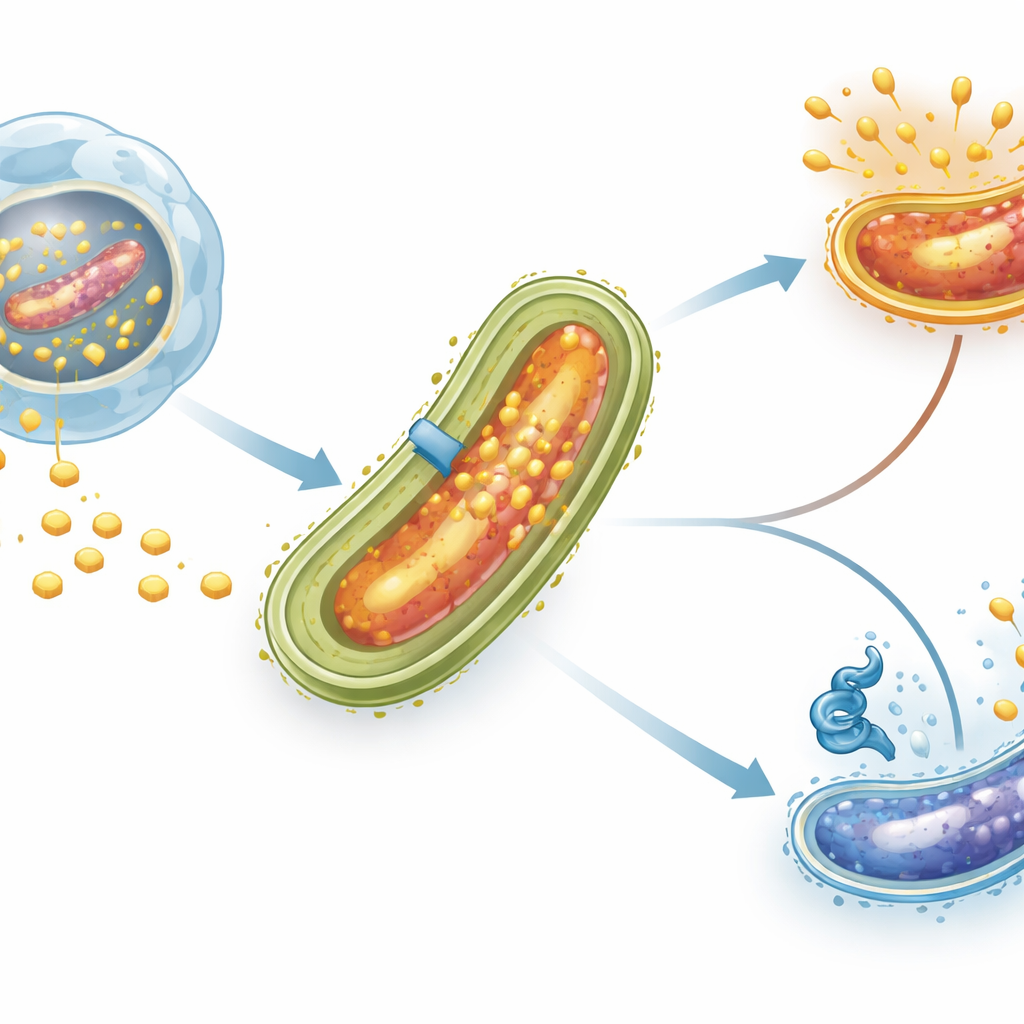
A traffic light for cholesterol entry
The researchers focused on a regulatory protein named LtmA, previously known to influence many lipid-related genes. Proteins like LtmA act as “traffic lights” on the bacterial DNA, deciding which genes should be active and which should be paused. Using computer analysis, the team identified a short DNA sequence in front of the mce4 gene cluster that looked like a likely docking site for LtmA. They then purified LtmA in the lab and performed a technique called an electrophoretic mobility shift assay, which essentially tests whether a protein physically grabs onto a DNA fragment. LtmA bound tightly and specifically to the normal mce4 control region but not to a version whose key bases were altered, confirming that this regulator directly recognizes the mce4 promoter.
Turning the cholesterol tap up or down
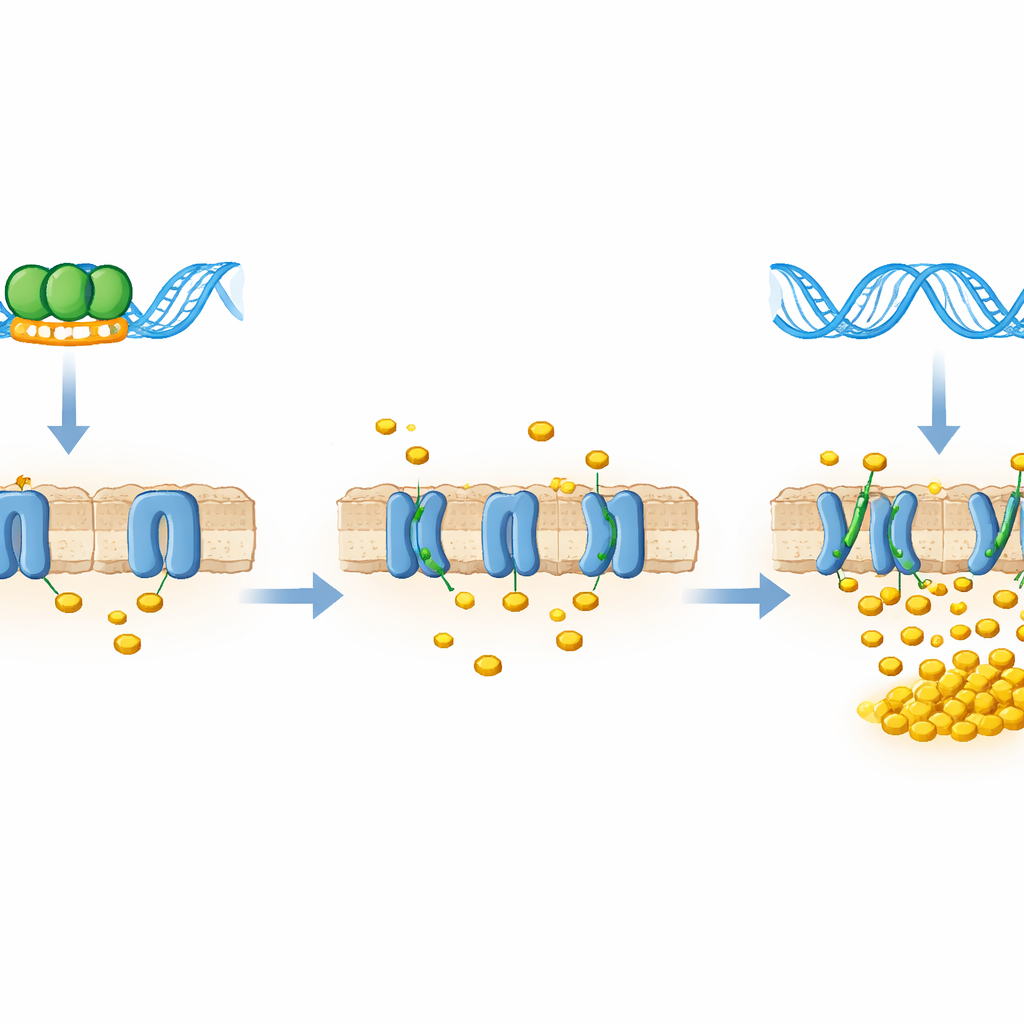
To see what this binding means for the living bacterium, the team engineered strains that either produced less LtmA (knockdown) or extra LtmA (overexpression). They measured the activity of the mce4 genes and found a mirror-image pattern: when LtmA levels were low, transcripts of the mce4 transporter genes rose several-fold; when LtmA was abundant, those same genes were sharply repressed. Functional tests supported this picture. In a cholesterol uptake assay, bacteria with reduced LtmA pulled in cholesterol faster and in greater amounts than normal cells, while those with excess LtmA lagged behind. LtmA, in other words, acts as a brake on the cholesterol-import machinery.
Growth only when cholesterol is on the menu
The consequences of this regulatory brake became clear when the scientists grew the different bacterial strains on defined carbon sources. When glucose served as food, all strains—whether short on LtmA, normal, or overloaded with it—grew equally well and showed similar metabolic activity. But when cholesterol was the only carbon source, the picture flipped. Strains with extra LtmA, and therefore less active mce4 transporters, grew poorly and showed low metabolic activity. Strains with diminished LtmA, whose Mce4 doors were more open, grew more robustly and metabolized cholesterol more vigorously. This shows that LtmA specifically limits growth under cholesterol-dependent conditions by restricting how much of this host-derived nutrient can enter the cell.
What this means for fighting tuberculosis
Taken together, the findings reveal LtmA as a direct repressor of the mce4 cholesterol transporter: when LtmA is bound to the mce4 control region, the molecular doors for cholesterol stay partly shut; when LtmA is reduced or absent, those doors open and the bacterium can feast. Although the work was carried out in a non-pathogenic model species, LtmA is closely related to a protein in Mycobacterium tuberculosis, suggesting that a similar control switch may influence how the pathogen exploits host cholesterol during infection. If so, drugs that interfere with this regulatory node—or with the transporter it controls—could one day help starve tuberculosis bacteria inside macrophages, adding a new angle to the global effort to control this enduring disease.
Citation: Feng, YW., Yin, BC., Li, HB. et al. Regulatory mechanism of cholesterol transport via LtmA in Mycobacterium smegmatis. Sci Rep 16, 14235 (2026). https://doi.org/10.1038/s41598-026-43871-4
Keywords: tuberculosis, cholesterol uptake, bacterial regulators, mycobacteria, host–pathogen interaction