Clear Sky Science · en
Atomistic and electronic insights into Ca2+ and Li+ intercalation in TiS2: a first-principles approach supported by electrochemical validation
Why new battery chemistry matters
From smartphones to electric cars, today’s rechargeable batteries mostly rely on lithium. But lithium is relatively scarce and expensive, and pushing current technology to higher performance is getting harder. This study explores an alternative: batteries that shuttle calcium ions instead of lithium ions. By zooming down to the atomic and electronic structure of a classic battery material called titanium disulfide (TiS2), the researchers ask a simple question with big implications: can calcium, which carries twice the charge per ion, move easily through TiS2 and store energy efficiently without tearing the material apart?
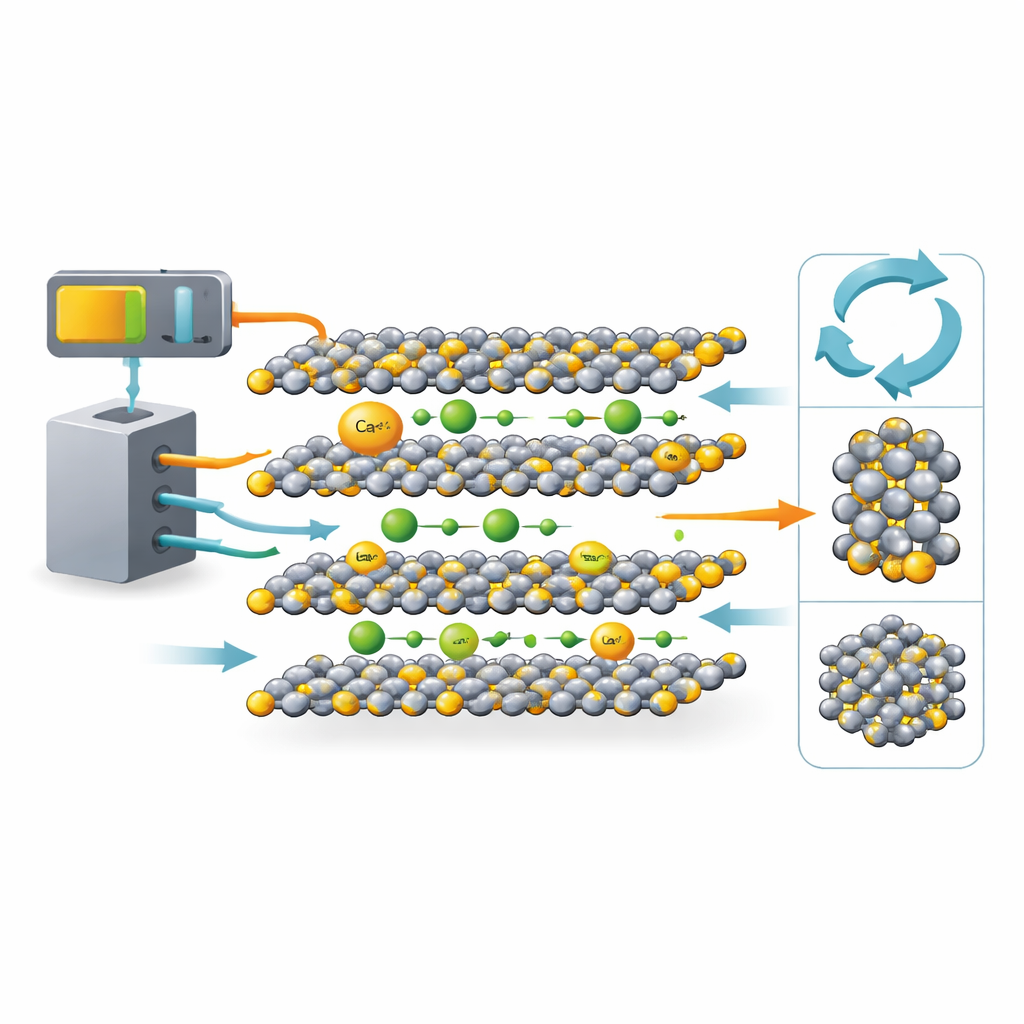
A familiar battery material under a new light
TiS2 has a long history in lithium-ion research. Its crystal structure is made of flat layers stacked like sheets of paper, with open spaces between them where ions can slip in and out. That architecture has made TiS2 a textbook host for lithium ions. The twist in this work is to compare lithium and calcium inside the very same TiS2 framework, side by side. Calcium ions are larger and carry double the charge, so many scientists have worried they would move sluggishly or damage the host lattice. By choosing TiS2, a well-understood material, the team can separate which behaviors come from the ions themselves and which come from the host.
Looking at atoms and electrons together
The researchers combined advanced computer simulations with real battery tests. Using first-principles calculations, they optimized the atomic positions in TiS2 when it is filled with either lithium or calcium and tracked how the layers expand. They then calculated how easily each ion hops from one site to another inside the material and examined how the electrons rearrange as ions move in. A second set of calculations focused on local chemical bonds—how strongly the ions interact with nearby sulfur atoms and how the titanium–sulfur framework responds. In parallel, they built actual coin cells using TiS2 electrodes and electrolytes carrying either lithium or calcium ions, measuring capacity, voltage, ion diffusion rates, and cycling stability.
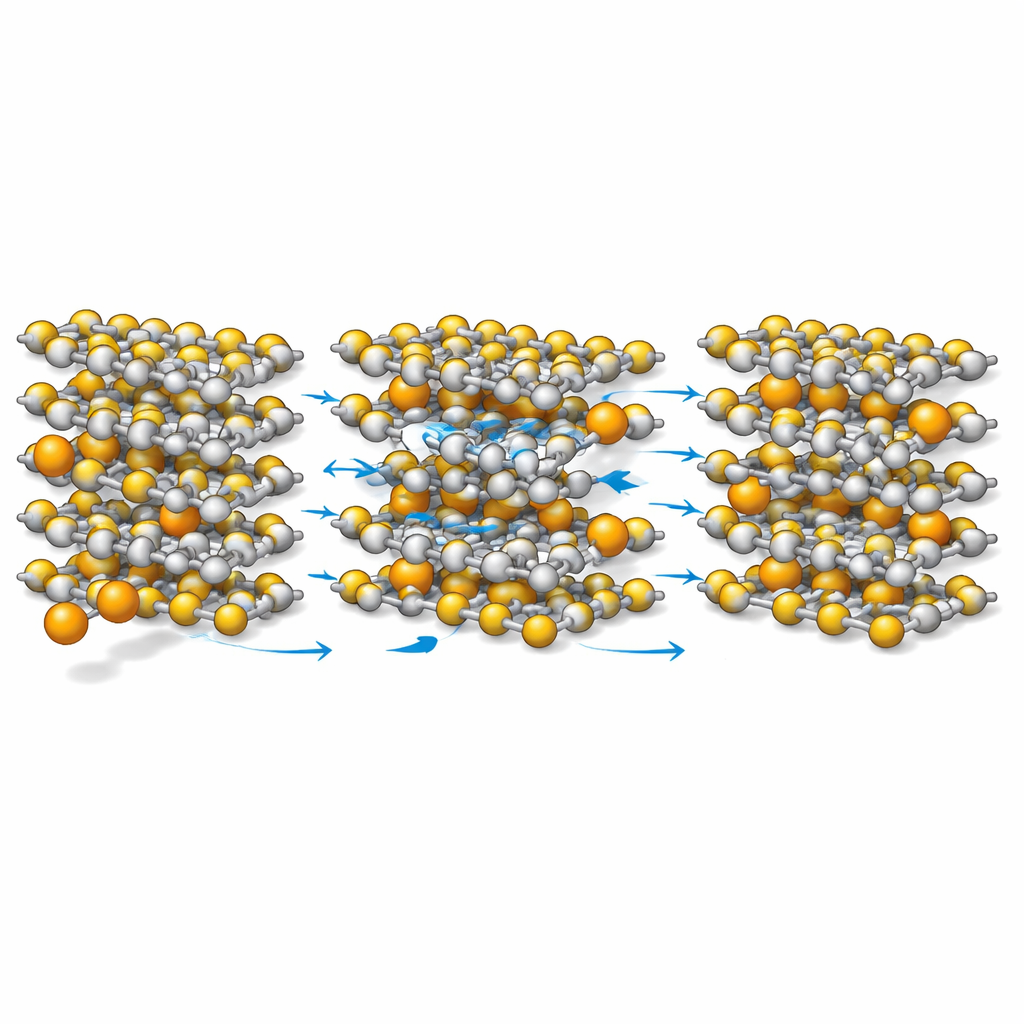
Calcium opens the channels but preserves the frame
The atomic-scale picture that emerges is counterintuitive. When calcium enters TiS2, it pushes the layers apart more than lithium does, widening the channels for ion motion. At the same time, calcium interacts more weakly with the sulfur atoms than lithium does, which means the ions are less tightly “anchored” and can move more freely. Yet the titanium–sulfur bonds that hold the layers together actually become slightly stronger in the calcium case, so the overall framework remains robust. Calculations show that the energy barrier for calcium ions to diffuse is lower than for lithium ions, and the electronic states near the material’s operating energy level become denser and more connected, which favors electronic conduction.
What this means for real batteries
Electrochemical tests echo the theoretical predictions. In TiS2-based cells, calcium intercalation delivers a first discharge capacity of about 201 mAh per gram—substantially higher than the roughly 134 mAh per gram seen with lithium under similar conditions. Calcium also shows faster ion diffusion, as inferred from cyclic voltammetry, and better rate performance: when the charge–discharge current is ramped up and then brought back down, the calcium cells retain over 96% of their original capacity, compared with about 89% for lithium cells. Over 100 cycles, both chemistries gradually lose capacity, but calcium maintains slightly higher values while keeping the charge–discharge process highly reversible.
Balancing energy, stability, and practicality
The study does find one trade-off: the average voltage of calcium in TiS2 is lower than that of lithium in the same host, meaning each unit of charge yields somewhat less energy. However, because each calcium ion moves two electrons instead of one, and because the structure stays stable and ions move quickly, the overall energy storage remains competitive. More importantly for future technology, calcium is abundant, low-cost, and environmentally benign. By showing in microscopic detail how calcium can move easily through TiS2 while the host lattice stays intact, this work lays out design rules for next-generation multivalent batteries: keep the host framework strong, let the ions bind just weakly enough to move fast, and tune the electronic structure so that charge flows smoothly. Those principles can now be applied to other layered materials in the search for practical calcium-ion batteries.
Citation: Yang, S., Lee, S., Nogales, P.M. et al. Atomistic and electronic insights into Ca2+ and Li+ intercalation in TiS2: a first-principles approach supported by electrochemical validation. Sci Rep 16, 14605 (2026). https://doi.org/10.1038/s41598-026-42087-w
Keywords: calcium-ion batteries, titanium disulfide, multivalent ions, battery materials, ion diffusion