Clear Sky Science · en
Extramedullary hematopoietic niches support the initiation and progression of myeloid malignancy in the murine spleen
Why the Spleen Matters in Blood Cancer
When we think about where blood is made, the bone marrow usually comes to mind. Yet many patients with blood cancers such as leukemia also develop enlarged spleens, and doctors have long noticed that this can signal more severe disease. This study in mice asks a deceptively simple question with big implications: can the spleen itself become a kind of “greenhouse” that helps dangerous blood cells grow and spread? By uncovering how a specific group of spleen cells shapes this hidden environment, the work suggests new ways to slow or reroute the course of myeloid blood cancers.
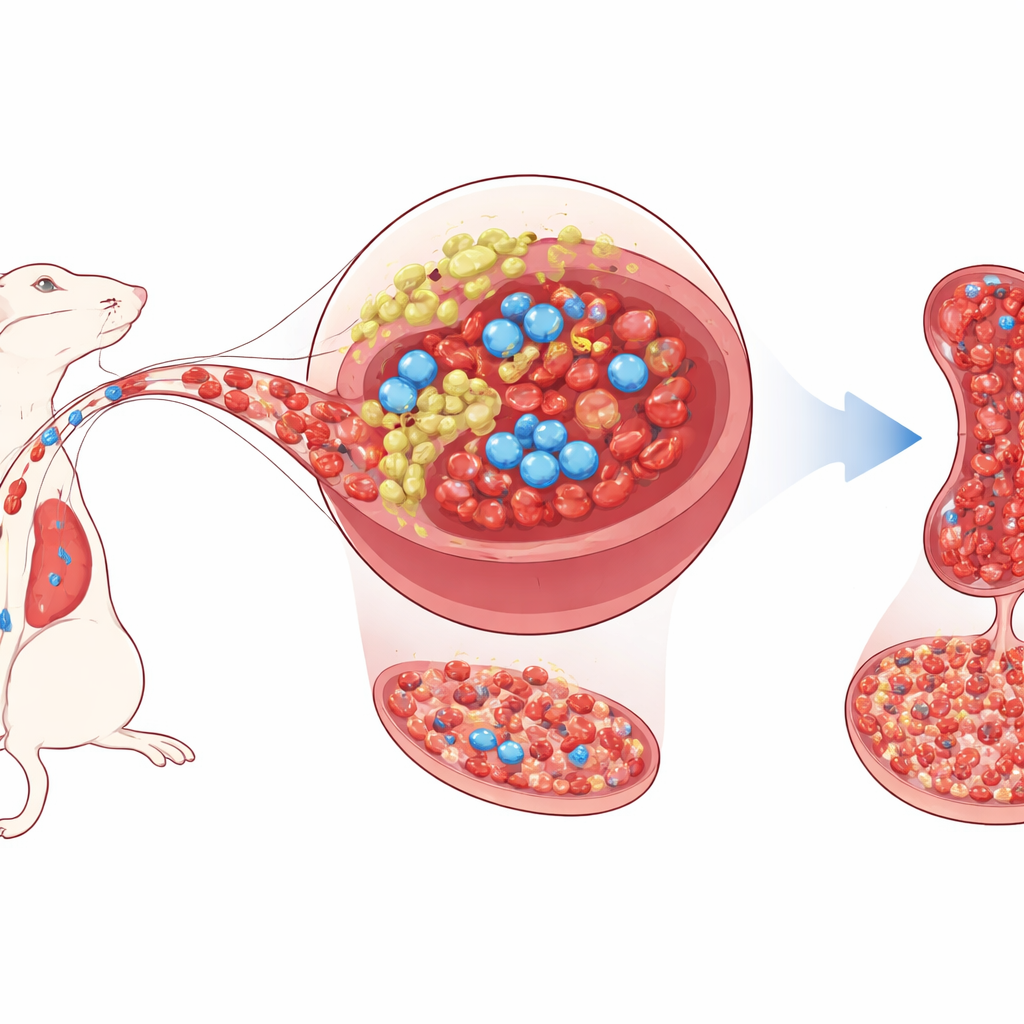
A Second Home for Blood-Making Cells
Healthy blood-forming stem cells usually live deep inside the bone marrow, where neighboring support cells feed them with growth signals and keep their behavior in check. The spleen, best known for filtering blood and fighting infections, can also switch on backup blood production under stress, a process called extramedullary hematopoiesis. The authors focus on a small population of support cells in the mouse spleen—mesenchymal stromal/progenitor cells—that carry a gene called Tlx1. In adult mice, Tlx1 is found almost exclusively in these splenic cells. Earlier work showed that turning Tlx1 on more strongly mobilizes stem cells from the bone marrow into the spleen and kick-starts extra blood production there.
How a Single Switch Rewires the Spleen
To understand how these Tlx1-bearing cells influence blood formation, the researchers first recreated the spleen’s support niche in a dish. They engineered splenic stromal cells so that adding a drug would boost Tlx1. When normal blood stem and progenitor cells were grown on top of these “switched-on” spleen cells, the blood cells multiplied more and died less, compared with cultures on unaltered spleen cells or on unrelated fibroblasts. The Tlx1-high stromal cells also made more of several key growth factors known to nourish stem cells. In other words, raising Tlx1 converted splenic support cells into a powerful cradle that expands immature blood cells.
When the Spleen Becomes a Leukemia Haven
The team then asked what happens during acute myeloid leukemia, a fast-growing cancer of myeloid blood cells. Using a well-established mouse leukemia cell line, they transplanted cancer cells into animals and watched how organs changed over time. As leukemia progressed, the spleen enlarged and its Tlx1-positive stromal cells ramped up Tlx1 and stem-cell-supporting molecules. Microscopy showed leukemia cells settling in close contact with Tlx1-rich niches in the spleen’s red pulp regions. Removing the spleen delayed the appearance of leukemia cells in the bloodstream and modestly extended survival, suggesting that this organ actively helps fuel the disease.
Turning the Niche Up or Down
To test cause and effect more directly, the authors created two types of genetically altered mice. In one, they could artificially overactivate Tlx1 in splenic stromal cells, exaggerating the extramedullary blood-forming niche. In these mice, transplanted leukemia cells homed to and accumulated in the spleen much more readily, spilled earlier into the blood, and shortened survival compared with normal littermates. In the second model, they could delete Tlx1 specifically from splenic stromal cells. Here, leukemia cells barely managed to take hold in the spleen, although the bone marrow still became heavily infiltrated and overall survival did not dramatically improve. These experiments reveal that Tlx1-driven spleen niches are crucial docking stations for leukemia, but they are not the only sites that sustain the disease.
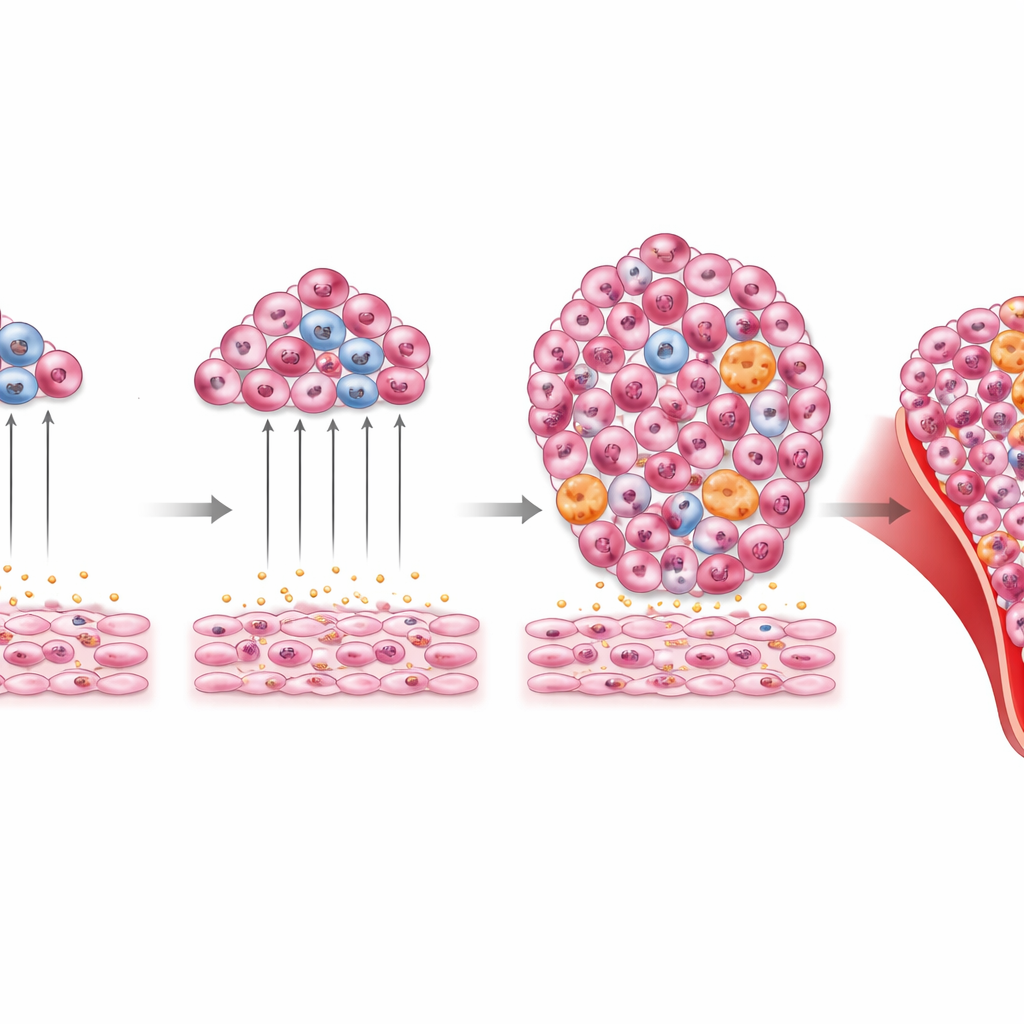
When Backup Blood Production Goes Too Far
The story does not end with short-term leukemia growth. The researchers also looked at what happens when Tlx1 stays high in splenic stromal cells for many months, even without injecting leukemia cells. Mice with chronic Tlx1 overexpression developed large spleens, anemia, excess white blood cells, and distorted bone marrow packed with myeloid cells and their precursors. Their livers became infiltrated by these cells, and blood smears showed abnormal, immature forms. Importantly, when bone marrow or spleen cells from these animals were transplanted into new recipients, the recipients developed similar myeloid abnormalities. This indicates that the long-lasting, Tlx1-driven spleen niche can help generate or maintain unhealthy myeloid cell populations that behave like a myeloid malignancy.
What This Means for Patients
Altogether, the work portrays the spleen not as a passive bystander but as an active partner in certain blood cancers. By flipping the Tlx1 switch in a specialized group of support cells, the spleen can recruit normal and malignant precursors, boost their growth, and, over time, encourage the rise of abnormal myeloid cells. For patients, this suggests that therapies aimed at reshaping or dampening the spleen’s leukemia-friendly niche—rather than removing the organ outright—could one day complement existing treatments. While humans are more complex than mice and many questions remain, the study highlights how the places where cancer cells live can be as important a target as the cancer cells themselves.
Citation: Oda, A., Tezuka, T., Nakamura, T. et al. Extramedullary hematopoietic niches support the initiation and progression of myeloid malignancy in the murine spleen. Sci Rep 16, 11211 (2026). https://doi.org/10.1038/s41598-026-41461-y
Keywords: acute myeloid leukemia, spleen niche, extramedullary hematopoiesis, hematopoietic stem cells, myeloid malignancy