Clear Sky Science · en
Intracranial LPS injection induces cerebral palsy-like motor and behavioral deficits in immunodeficient mice
Why this research matters
Cerebral palsy is one of the most common causes of life‑long movement difficulties in children, yet doctors still lack treatments that can truly repair the injured brain. Many promising approaches, such as stem cell therapies, must first be tested in animals. However, standard laboratory animals often reject human cells, making it hard to predict whether a therapy will work in children. This study introduces a new mouse model that closely mimics key movement problems seen in cerebral palsy while also being friendly to human cells, opening the door to more realistic testing of future treatments.
Building a special kind of mouse model
The team worked with newborn immunodeficient mice that lack key parts of the immune system which normally attack foreign cells. When the pups were six days old, the researchers carefully injected a tiny amount of lipopolysaccharide, or LPS—a molecule found on certain bacteria—directly into a white‑matter region of the brain. LPS is widely used in research because it reliably triggers inflammation, similar to what might occur during a serious infection before or shortly after birth, a known risk factor for cerebral palsy. A second group of pups underwent the same surgery but received only saltwater, serving as healthy controls. The mice were then allowed to grow for six weeks, roughly corresponding to a juvenile stage, before their health and behavior were examined in detail. 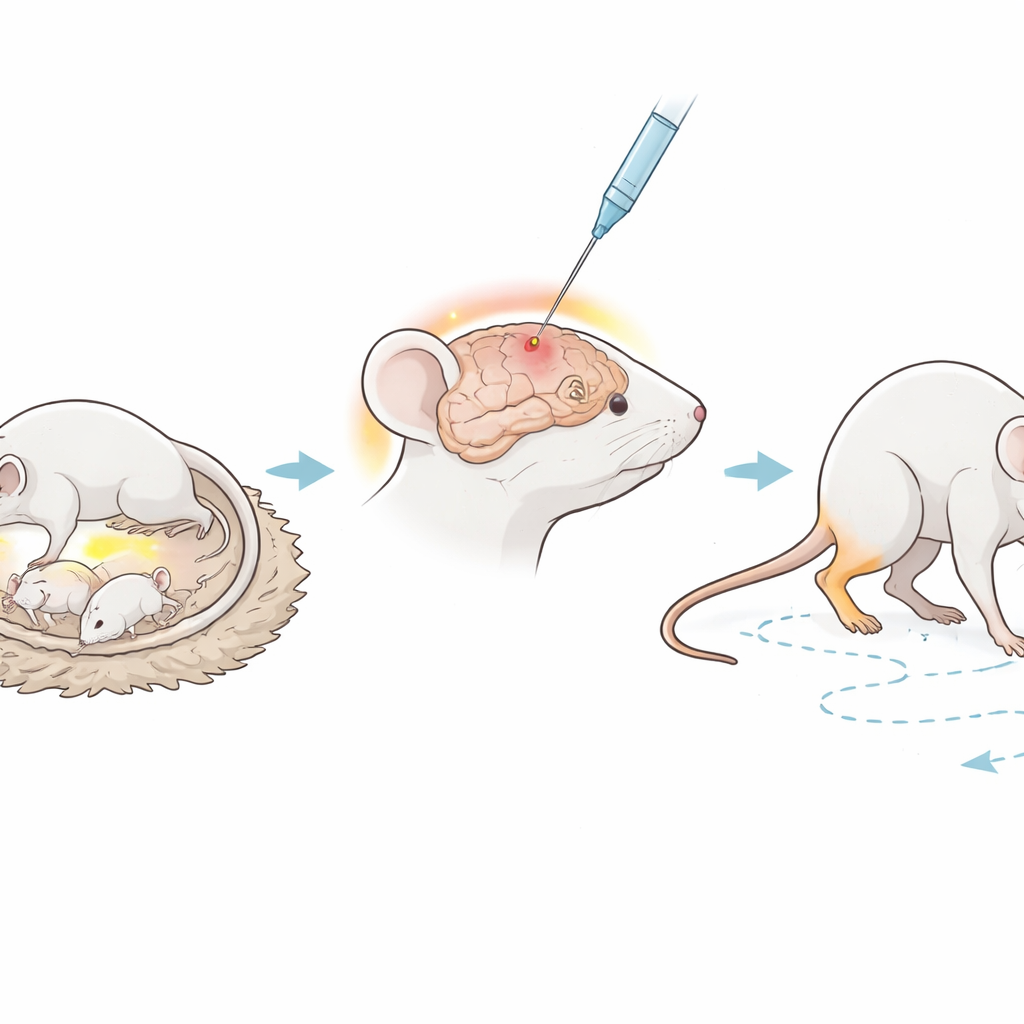
Signs of poorer growth and uneven movement
Compared with control animals, fewer LPS‑treated mice survived to the end of the study, and those that did weighed less, suggesting that the early brain insult affected overall development. When researchers tested basic movement, they found that LPS‑treated mice showed a clear imbalance: when held gently by the tail, their bodies swung more often to one side, and on a rotating rod they fell off sooner, indicating reduced balance and endurance. In an open arena, these mice walked more slowly and covered less distance overall, a pattern known as hypolocomotion. Interestingly, although they did not spend more time in the center of the arena, a greater share of the little movement they did make occurred there, hinting at altered exploration patterns rather than simple changes in anxiety.
Gait changes that echo children’s walking problems
The most striking findings came from computerized gait analysis, which measures how each paw moves while the mouse walks on a transparent treadmill. LPS‑treated mice walked more slowly, with longer step cycles and fewer steps per second. Their hindlimbs were especially affected: they spent more time in contact with the belt and pushed off for longer, but their base of support became narrower. The angle of the hind paws increased, resembling the inward‑turning, scissoring‑like foot positions often seen in children with spastic diplegic cerebral palsy. At the same time, the front paws showed different changes, underscoring a complex reshaping of coordination between front and back limbs. Together, these patterns indicate a multifaceted movement disorder dominated by hindlimb abnormalities, closely paralleling the lower‑limb‑focused problems in many children with cerebral palsy. 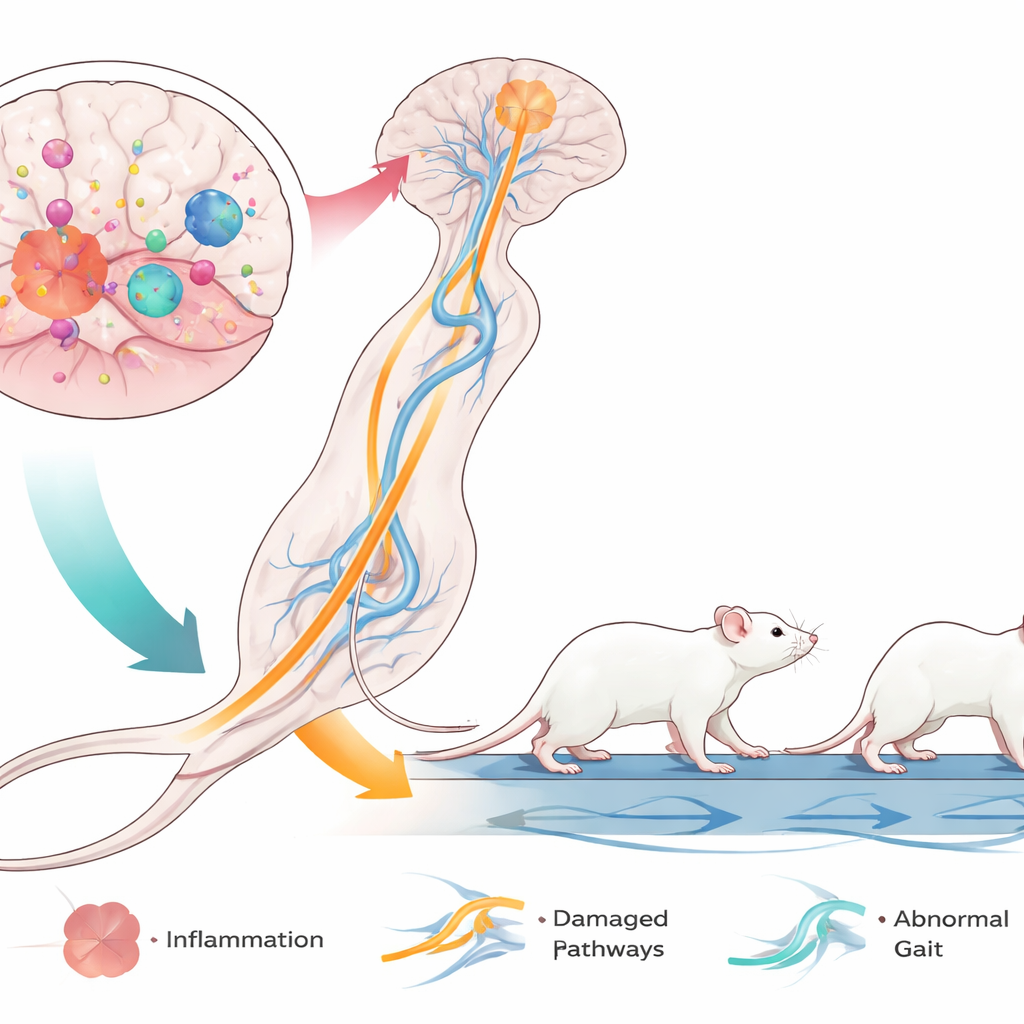
What may be happening inside the brain
Although this study did not directly measure brain cells, it draws on earlier work suggesting that LPS sparks an aggressive inflammatory response in the brain’s resident immune cells, called microglia. These activated cells can damage the insulation around nerve fibers—especially in white‑matter regions that carry signals from the brain’s movement centers to the spinal cord and legs. Because the mice lack adaptive immune cells but still have innate immune responses, the model isolates how early inflammation alone can disrupt motor pathways while remaining compatible with human cell transplants. The hindlimb‑focused deficits hint that specific brain regions and wiring for the rear legs are particularly vulnerable to this kind of early‑life insult.
What this means for future therapies
In simple terms, the researchers created young mice whose movement problems look and behave much like a form of cerebral palsy in children, especially affecting the legs, and did so in animals that can safely host human cells. This new model does not capture every aspect of the human condition, but it provides a realistic and ethically refined platform for exploring how early inflammation injures the developing brain and for testing whether experimental treatments—especially human stem cell and other cell‑based therapies—can restore more normal movement. If successful therapies emerge in this setting, they are more likely to translate into meaningful improvements for children living with cerebral palsy.
Citation: Yang, J., Li, Y., Shi, C. et al. Intracranial LPS injection induces cerebral palsy-like motor and behavioral deficits in immunodeficient mice. Sci Rep 16, 10105 (2026). https://doi.org/10.1038/s41598-026-40909-5
Keywords: cerebral palsy, neuroinflammation, animal models, gait abnormalities, cell therapy