Clear Sky Science · en
Study on the effects and mechanisms of M2 macrophages on PYCR1-promoted biological behavior of hepatocellular carcinoma cells
Why this liver cancer study matters
Primary liver cancer, mostly hepatocellular carcinoma, is one of the deadliest cancers worldwide, in part because it often spreads early and resists treatment. This study looks at a little-known metabolic enzyme, PYCR1, and a group of immune cells called macrophages to understand how liver tumors grow, spread, and dodge cell death. By uncovering how these players cooperate inside the tumor’s neighborhood, the work points to a new weak spot that future drugs might target to slow or stop liver cancer.
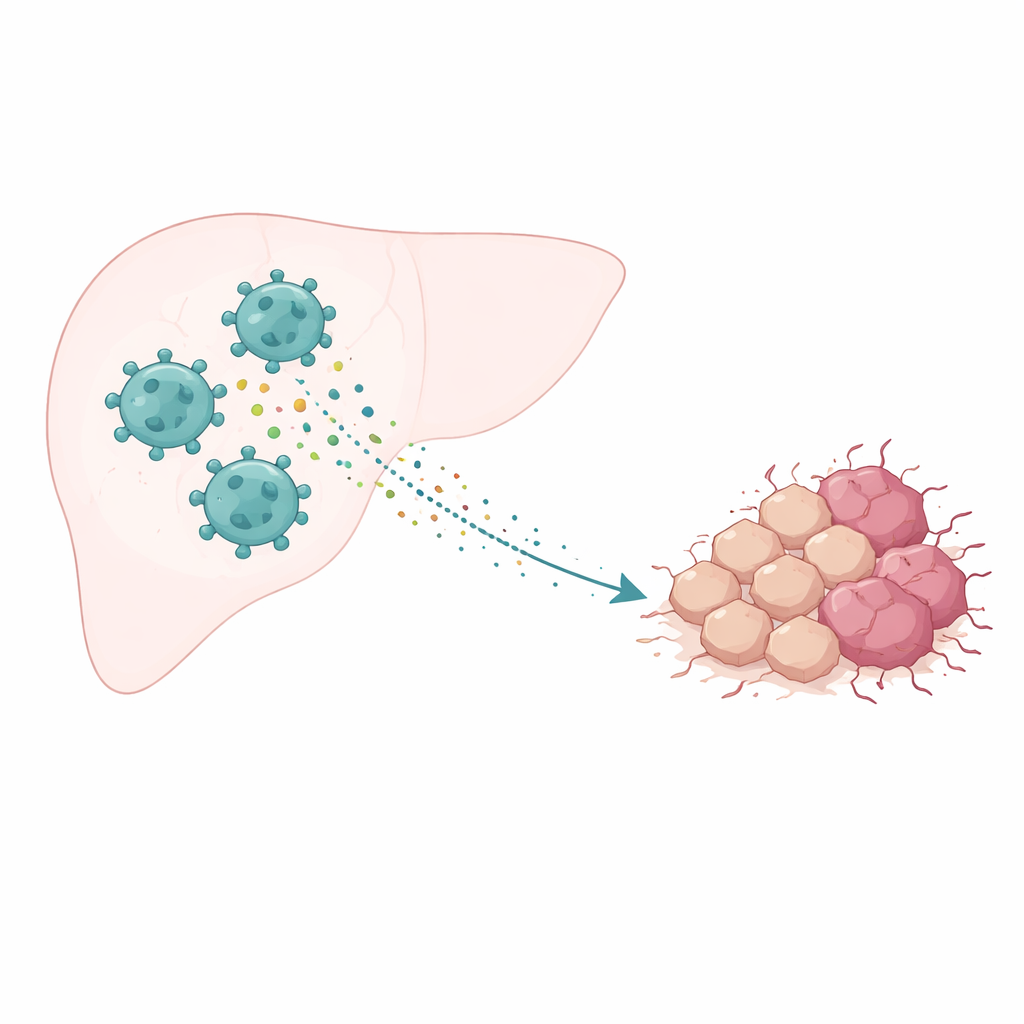
The neighborhood around a tumor
Cancer does not grow in isolation; it lives in a complex “neighborhood” of blood vessels, support cells, and immune cells known as the tumor microenvironment. In liver cancer, one of the most abundant immune cell types is the tumor‑associated macrophage. These cells can adopt different personalities: some attack tumors, while others, called M2‑type macrophages, soothe inflammation and can end up helping the cancer. Many studies have shown that when M2‑type cells dominate, tumors tend to grow faster, spread more easily, and respond poorly to treatment. This raised an important question: how exactly do these helper‑type macrophages push liver cancer cells toward more malignant behavior?
An enzyme that fuels dangerous growth
The researchers focused on PYCR1, an enzyme that helps cells make the amino acid proline, a key component of structural proteins like collagen and an important fuel for rapidly dividing cells. By analyzing patient data, they found that PYCR1 levels are higher in liver tumors than in normal liver tissue, and that patients whose tumors show more PYCR1 and more M2‑type macrophage infiltration tend to have worse survival. This suggested that PYCR1 might be a crucial link between the tumor cells’ altered metabolism and the harmful influence of their immune surroundings.
How macrophages push tumor cells to spread
To test this idea, the team grew liver cancer cell lines in dishes with liquid collected from M2‑type macrophages. Under these conditions, the cancer cells became more aggressive: they moved across artificial “wounds” faster, invaded through membranes more readily, and showed protein changes that signal a shift from a stable, cobblestone‑like epithelial state to a more flexible, travel‑ready mesenchymal state. This switch, known as epithelial–mesenchymal transition, is a key step in metastasis. When the scientists used genetic tools to lower PYCR1 levels in the cancer cells, these M2‑driven effects were largely reversed, and the cells became less mobile and less invasive, indicating that PYCR1 is required for macrophage signals to fully empower the tumor cells.
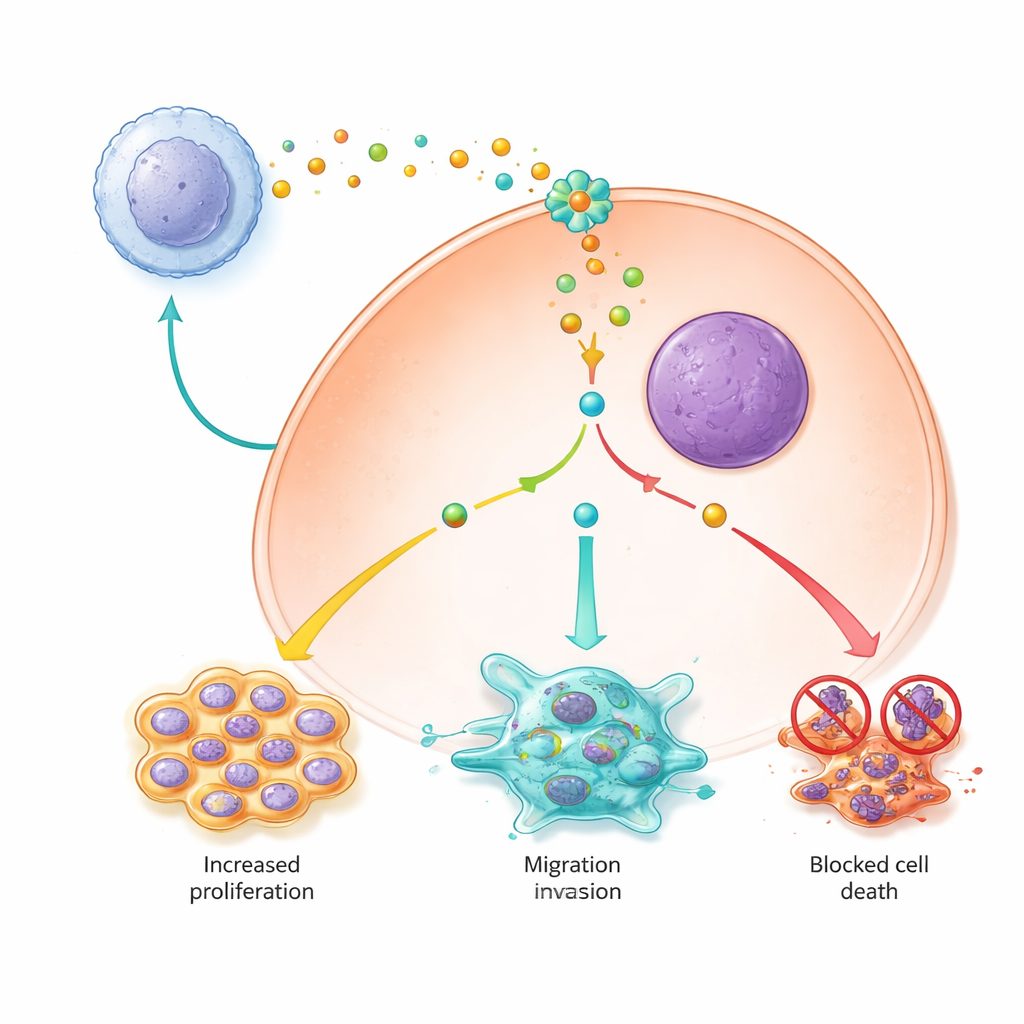
Keeping cancer cells alive and dividing
The study also showed that M2‑type macrophage signals help liver cancer cells stay alive and multiply by altering key survival pathways. Cancer cells exposed to M2‑conditioned medium divided more rapidly, were less likely to undergo programmed cell death, and were less prone to a newer form of iron‑dependent cell death called ferroptosis. Inside the cells, PYCR1 turned out to be a central switch: when it was active, a major signaling route called JAK2/STAT3 was more strongly turned on, pro‑survival proteins increased, and proteins that block ferroptosis were elevated while damaging iron and reactive molecules were kept low. Knocking down PYCR1 or blocking JAK signaling with a small‑molecule inhibitor weakened this cascade, slowed proliferation, and restored both apoptosis and ferroptosis.
What this means for future treatments
Taken together, the findings paint PYCR1 as a master helper that allows liver cancer cells to fully exploit the support of M2‑type macrophages: it boosts growth, enables shape‑shifting and spread, and shields cells from two forms of death. For patients, this suggests that drugs designed to block PYCR1, especially when combined with treatments that reprogram or deplete tumor‑supporting macrophages, could attack liver cancer on several fronts at once. While these experiments were done in cell cultures and still need to be confirmed in animal models and clinical studies, they highlight PYCR1 and the tumor microenvironment as promising targets in the quest for more effective liver cancer therapies.
Citation: Jin, X., Hou, Y., Guo, J. et al. Study on the effects and mechanisms of M2 macrophages on PYCR1-promoted biological behavior of hepatocellular carcinoma cells. Sci Rep 16, 13716 (2026). https://doi.org/10.1038/s41598-026-40817-8
Keywords: hepatocellular carcinoma, tumor microenvironment, macrophages, PYCR1, JAK2 STAT3