Clear Sky Science · en
Paradoxical oncogenic effects of hepatic Brca1 through modulating Bhmt
When Cancer Protection Behaves Backwards
Most of us think of genes like BRCA1 as guardians that prevent cancer. This study turns that idea on its head for the liver. The researchers show that, under certain conditions, normal BRCA1 activity in liver cells can actually help liver tumors grow, while switching it off can protect against cancer. Understanding this surprising twist could reveal new ways to prevent or treat one of the deadliest cancers worldwide.
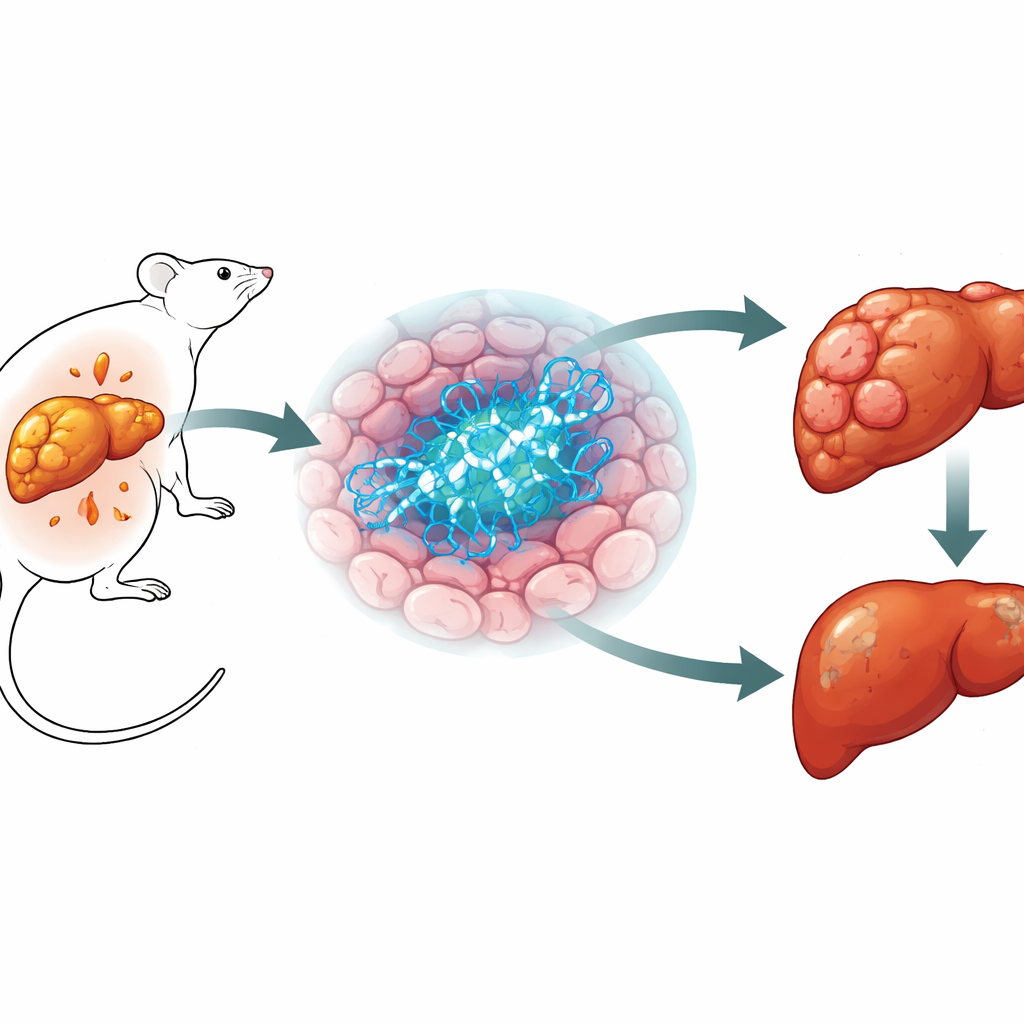
A Deadly Liver Cancer on the Rise
Liver cancer, particularly hepatocellular carcinoma, is already one of the most common and lethal cancers, and its burden is still climbing. A major driver is a condition linked to obesity and type 2 diabetes, in which fat slowly accumulates in the liver and can progress to scarring and eventually cancer. Scientists know that metabolism and cancer are tightly connected, but many of the specific molecular players remain unclear. Because BRCA1 has long been recognized as a tumor-suppressing gene in breast and ovarian cancer, and has also been tied to how cells handle fats and sugars, the authors asked whether BRCA1 might influence both liver metabolism and liver cancer.
Switching Off a Guardian in Liver Cells
The team created mice in which BRCA1 was removed only from liver cells, leaving the rest of the body untouched. As expected from BRCA1’s classic role in repairing damaged DNA, these liver cells showed more DNA breaks. But the overall health of the animals told a different story. When fed a high-fat diet, mice lacking BRCA1 in their livers accumulated less fat inside their livers and handled sugar better than normal mice, even though their body weight was similar. Most strikingly, when the animals were exposed to a cancer-causing chemical that reliably produces liver tumors, those without BRCA1 in their liver cells developed far fewer and smaller tumors than their normal littermates. This protection appeared in two different tumor models, showing that BRCA1’s presence in liver cells can, paradoxically, favor tumor growth.
Uncovering a Hidden Metabolic Pathway
To understand how loss of BRCA1 could shield the liver from cancer, the researchers examined which genes were active in thousands of individual liver cell nuclei. They discovered that one gene, Bhmt, stood out as consistently less active when BRCA1 was missing. BHMT is an enzyme almost exclusively found in the liver that sits at a crossroads of methionine and choline metabolism—pathways that supply building blocks for DNA, support chemical tagging of molecules, and feed the production of membrane lipids. Gene set analyses revealed that many small-molecule and organic acid pathways, especially those linked to one-carbon metabolism and choline use, were dialed down in BRCA1-deficient hepatocytes, and signatures associated with liver tumor formation were also reduced. The same drop in BHMT activity appeared when the team silenced BRCA1 in human liver cancer cells, suggesting a direct connection between the two genes.
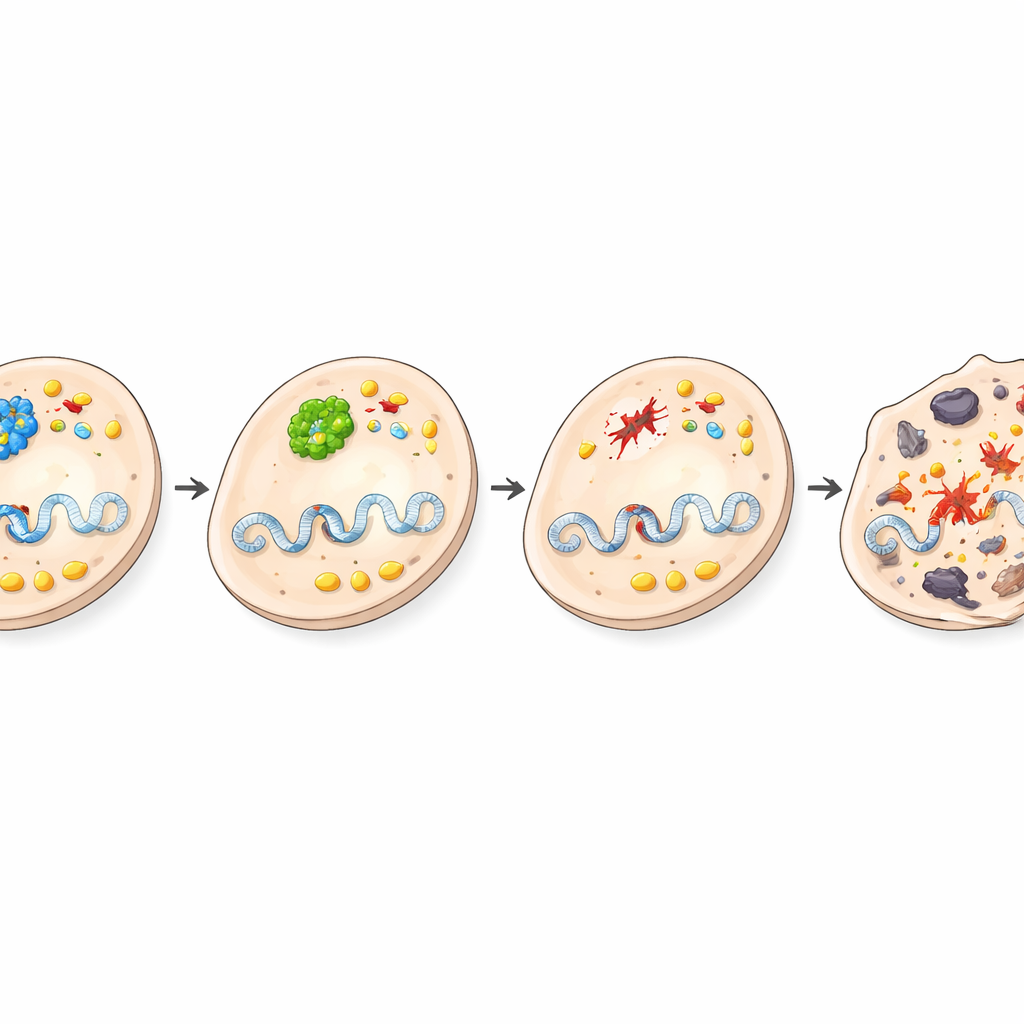
Too Much Damage, Not Enough Fuel for Tumors
The authors then zoomed in on how BRCA1 and BHMT interact functionally. In human liver cancer cells, turning off BRCA1 increased DNA breaks, and turning off BHMT did so as well. When both were suppressed together, DNA damage piled up even more. After these cells were exposed to irradiation, BRCA1 loss strongly boosted cell death, and combined loss of both genes maintained this heightened vulnerability. At the same time, the metabolic side of the story came into focus. Choline levels—a chemical whose buildup is a recognized hallmark of many cancers—rose in liver tumors from normal mice but were blunted in tumors and blood from BRCA1-deficient mice. Enzymes that channel choline into membrane lipids were also reduced, and broad metabolite profiling showed disrupted production of phosphatidylcholine, glycine, and polyamines, all important for rapidly dividing cells. Together, these changes suggest that without BRCA1, liver cells both accumulate lethal DNA damage and lose access to key metabolic fuel needed for tumor growth.
What This Twist Means for Cancer
This work reveals that BRCA1, famed as a tumor suppressor elsewhere in the body, can instead have tumor-promoting effects in the liver by supporting BHMT activity and choline-centered metabolism. When BRCA1 is removed specifically from liver cells, BHMT activity falls, DNA damage becomes overwhelming, choline-related pathways are dampened, and damaged cells die rather than evolve into tumors. The study highlights how the same gene can have opposite roles depending on tissue and metabolic context, and it points to one-carbon and choline metabolism as promising targets for new liver cancer prevention or treatment strategies.
Citation: Ghebreselassie, M., Park, Y.J., Nakib, D. et al. Paradoxical oncogenic effects of hepatic Brca1 through modulating Bhmt. Sci Rep 16, 13479 (2026). https://doi.org/10.1038/s41598-026-39581-6
Keywords: liver cancer, BRCA1, choline metabolism, one‑carbon metabolism, hepatocellular carcinoma