Clear Sky Science · en
Association between bone marrow donor origin and gut microbiota composition following fecal microbiota transplantation in mice
Why the Gut’s Tiny Tenants Matter
Doctors increasingly use fecal microbiota transplantation (FMT)—transferring stool from a healthy donor to a patient—to reset the gut’s community of microbes and treat stubborn diseases. But not every transplant “takes” equally well. This mouse study asks a surprisingly practical question: if a patient has previously received a bone marrow transplant, and thus a different immune system, does the origin of that bone marrow change how well donor gut microbes can settle in? The answer could help design safer, more reliable microbiome-based therapies.
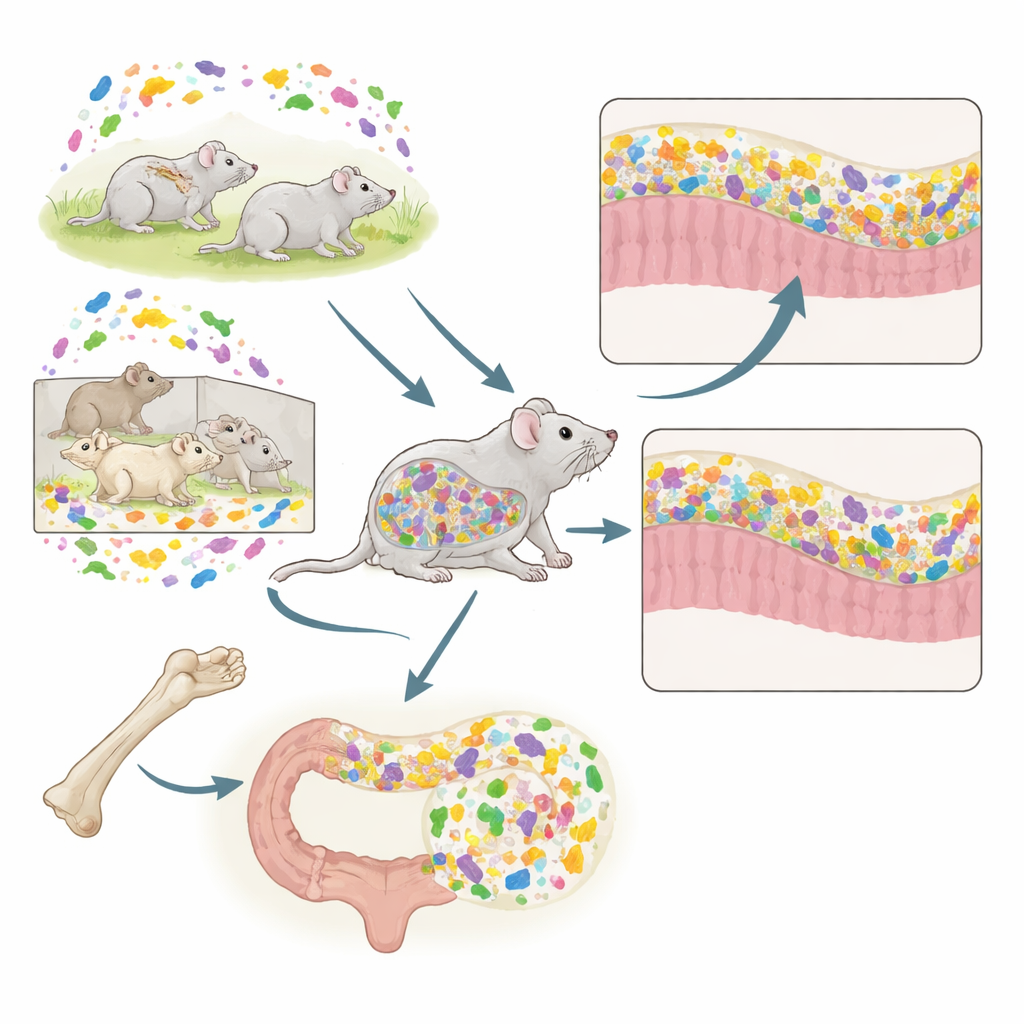
Two Transplants, One Mouse
The researchers combined two powerful tools in mice: bone marrow transplantation (BMT), which replaces immune cells, and FMT, which replaces gut microbes. Donor mice came from two distinct living conditions. Conventional (CV) mice live in standard open cages and harbor a rich, varied microbiota. Specific-pathogen-free (SPF) mice live in protected barrier facilities and carry a simpler, less diverse microbial community and a less mature immune system. Recipient mice always lived in conventional conditions but received bone marrow and/or feces from CV or SPF donors in different combinations. This design let the team test how the immune background shaped by bone marrow origin interacted with the incoming fecal microbes.
Checking Who Moves into the Gut
One week after FMT, the scientists collected fecal samples and analyzed the bacterial DNA, focusing on which types of microbes were present and how abundant they were. Overall diversity—the sheer variety and balance of microbes—did not differ much between groups. However, the detailed community structure did. Using distance measures that capture which species are present (rather than how dominant each is), they found that the origin of the bone marrow donor significantly changed the pattern of microbes that established after FMT. These effects were most obvious when the fecal material came from conventional donors, suggesting that a richer, more complex microbial mix is particularly sensitive to the immune environment set by bone marrow.
Hidden Functional Shifts in Microbial Metabolism
To look beyond “who is there” and ask “what might they be doing,” the team used computational tools to predict metabolic pathways from the bacterial DNA profiles. When bone marrow and feces both came from donors with the same type of microbiota (CV with CV, or SPF with SPF), the predicted functions of the gut community shifted in consistent ways. In mice receiving matched CV material, pathways linked to building amino acids such as arginine and polyamines were more prominent. In mice with matched SPF material, pathways related to aromatic amino acids like tyrosine and phenylalanine were predicted to increase. These forecasts are indirect and need confirmation with deeper molecular analyses, but they hint that matching donor sources may subtly steer what the transplanted microbes can produce.
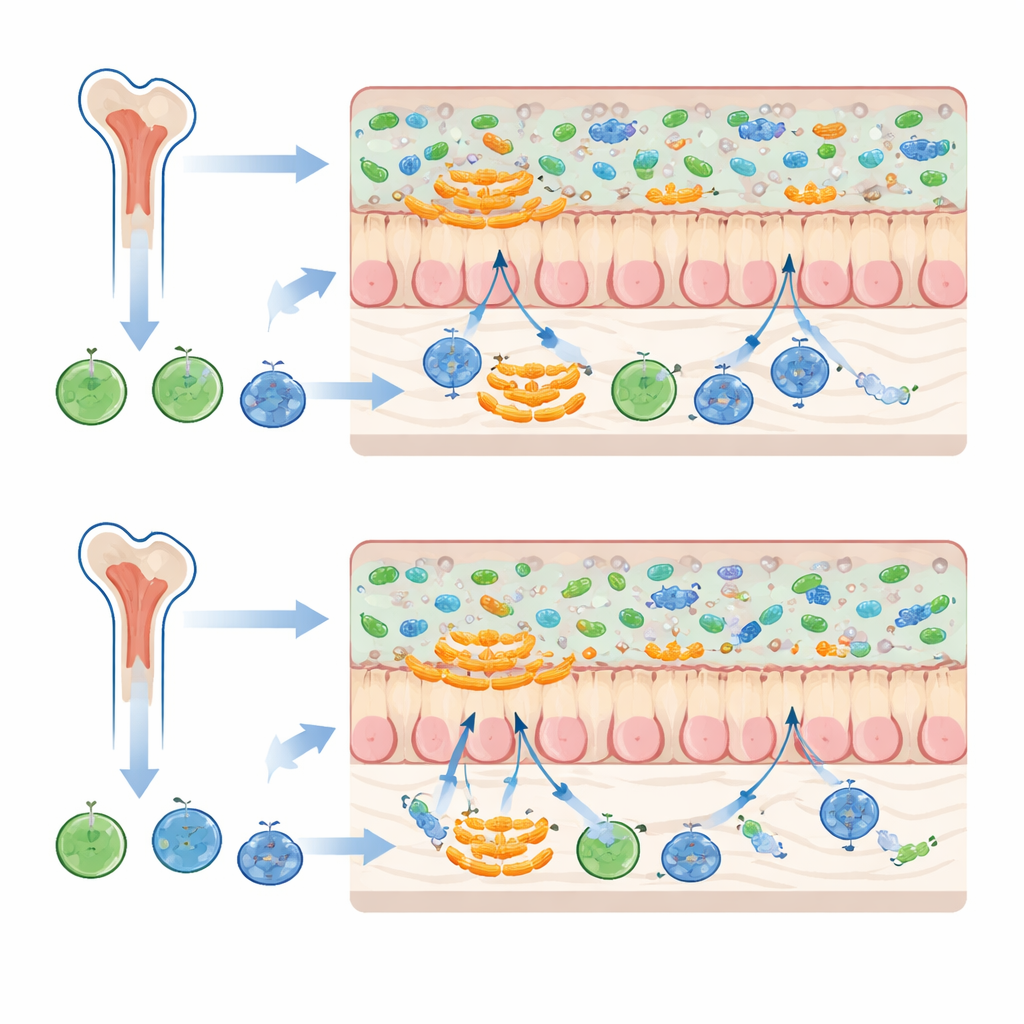
A Special Group of Mucus-Loving Bacteria
The most striking changes involved a family of bacteria called Muribaculaceae, which tend to live close to the intestinal mucus layer rather than floating freely in the gut contents. When mice received both bone marrow and feces from donors with the same microbiota background, certain Muribaculaceae became more abundant, and the overall microbial community in recipients more closely resembled that of the fecal donors. When bone marrow and feces came from different microbiota backgrounds, these bacteria expanded less, and donor-like communities were less faithfully reproduced. Because mucus-associated microbes sit at the interface between gut contents and the intestinal lining, they may be especially sensitive to the immune tone set by donor-derived bone marrow cells.
What This Means for Future Treatments
In plain terms, the study suggests that the “match” between a person’s immune system and their incoming gut microbes can influence which bacteria stick around after FMT, especially those living close to the gut wall. Matching bone marrow and fecal donor backgrounds in mice favored the growth of certain mucus-dwelling bacteria and helped recipient microbiotas more closely mirror their donors, even though overall diversity did not dramatically change. While radiation and other pre-transplant treatments also affect the gut and were not fully separated here, these results highlight that immune history matters for microbiome therapies. As FMT and related approaches move forward in patients, especially those who are immunocompromised, carefully considering donor–recipient pairings could make these treatments more predictable and effective.
Citation: Ichimura, R., Tanaka, K., Song, I. et al. Association between bone marrow donor origin and gut microbiota composition following fecal microbiota transplantation in mice. Sci Rep 16, 13314 (2026). https://doi.org/10.1038/s41598-026-36933-0
Keywords: fecal microbiota transplantation, bone marrow transplantation, gut microbiome, immune system, mucus-associated bacteria