Clear Sky Science · en
Blood vessel-resident macrophages safeguard blood and vessel integrity in zebrafish
Hidden Guardians in Our Bloodstream
Every second, blood rushes through an immense network of vessels that, laid end to end, would stretch for miles in even a tiny animal. We rely on this hidden highway to carry oxygen, nutrients and immune cells. Yet one basic question has remained surprisingly unclear: who constantly checks the quality of the blood itself and keeps delicate vessel walls safe? This study, done in transparent zebrafish embryos and extended to mice, uncovers a previously unrecognized type of immune cell that lives inside blood vessels and acts as an on‑the‑spot inspector and cleaner for the circulation.
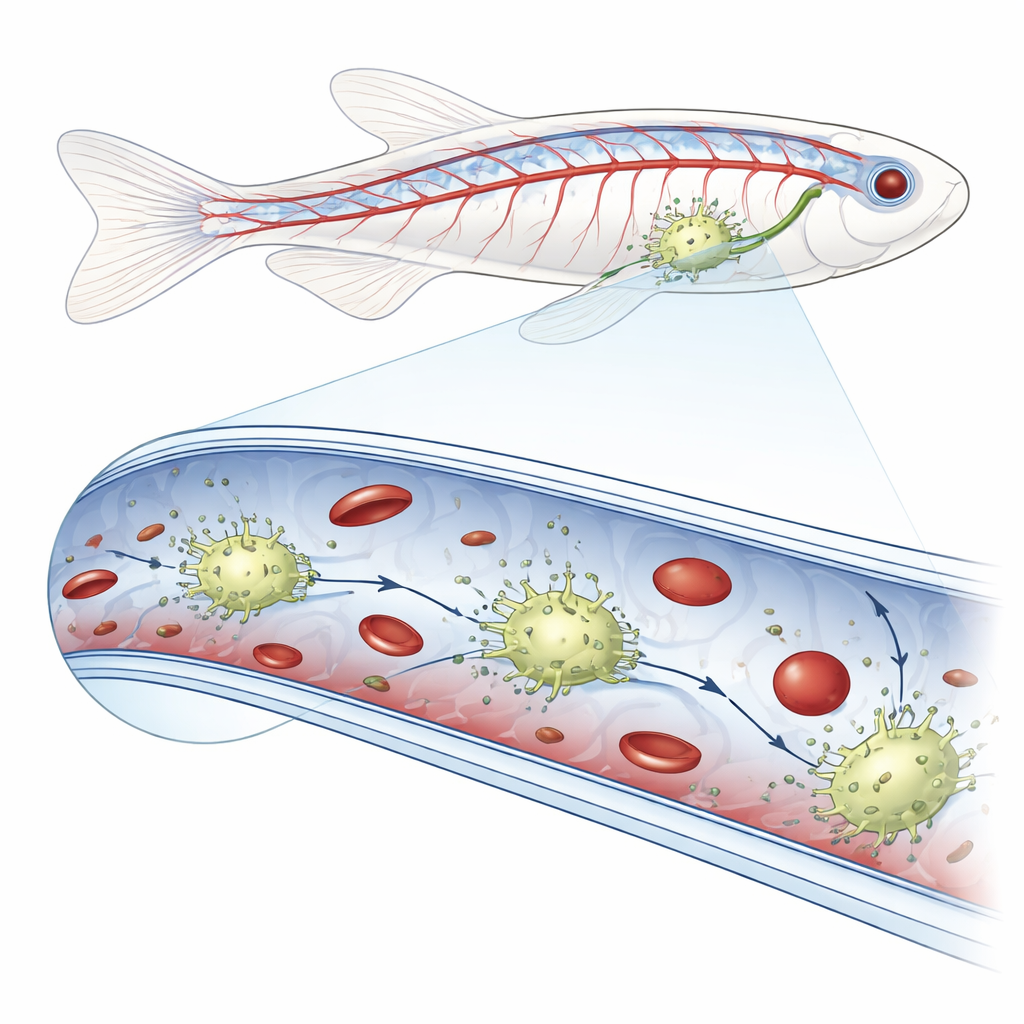
New Patrol Cells Inside Blood Vessels
Using high‑resolution live imaging in zebrafish embryos, the researchers noticed unusually bright cells crawling along the inner surfaces of veins in the tail region. When they injected fluorescent bacteria, fungal particles, synthetic beads or donor red blood cells into the bloodstream, these intravascular cells were the only ones that swallowed the foreign material. Careful genetic labeling showed that these cells carried hallmarks of both vessel‑lining cells and classic immune eaters known as macrophages. Because they reside within blood vessels rather than in surrounding tissues, the team named them blood vessel‑resident macrophages, or bMΦs.
How These Cells Watch and Clean the Blood
Close‑up time‑lapse movies revealed that bMΦs crawl in an amoeba‑like fashion along the vessel lining, sometimes briefly blocking flow, a sign of strong attachment. Their surfaces bristle with thin, flexible protrusions that help them latch onto the wall and snare passing material. When the team disrupted the cells’ internal scaffolding with a drug, these protrusions disappeared, bMΦs detached from the vessel surface and could no longer efficiently clear injected particles, which then drifted freely in the circulation. The cells also interacted with the animal’s own blood cells: they grabbed passing red blood cells and blood‑forming stem and progenitor cells, examined them at the surface and then either released them unharmed or engulfed those that looked unusually large or otherwise abnormal, helping maintain blood quality.
First Responders to Vessel Injury
Beyond everyday cleaning, bMΦs also act as rapid first responders when vessels are damaged. The researchers used an optogenetic tool—essentially a light‑activated molecular switch—to trigger the death of a few selected endothelial cells, which form the vessel wall. Immediately after this targeted injury, bMΦs rushed to the site and efficiently cleared away the dying cell fragments. Traditional tissue macrophages and fast‑moving white blood cells called neutrophils arrived less often, spent less time at the damage site and performed little cleanup. These observations suggest that bMΦs are specially positioned and equipped to sense trouble from within the vessel and to preserve the cleanliness and integrity of the blood–vessel interface.
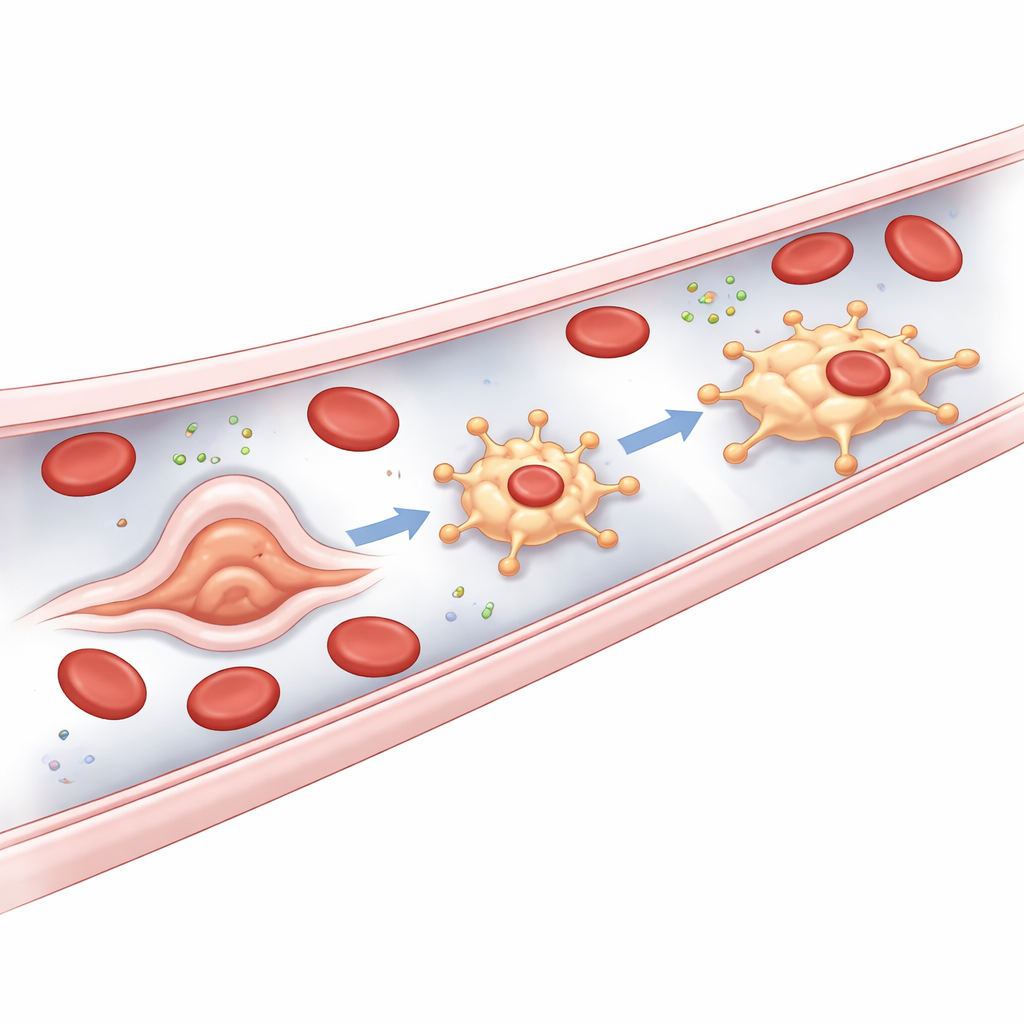
Where These Guardians Come From
A key surprise of the study is the origin of bMΦs. Most tissue‑resident macrophages arise from early blood precursors or from later bone marrow cells. In contrast, bMΦs appear to bud directly from the vessel lining itself. In zebrafish embryos, soon after blood flow begins, certain endothelial cells in the main trunk vessels and especially in the tail vein plexus undergo a rapid shape change, bulging into the vessel and then detaching as free‑moving bMΦs. The authors call this unusual process an endothelial‑to‑macrophage transition. It depends on well‑known regulators of the myeloid immune lineage but, strikingly, does not require Runx1, a master switch normally essential for turning vessel cells into blood‑forming stem cells. This marks bMΦs as a distinct, fourth developmental route for macrophages in zebrafish.
Conserved Role Across Species and Future Promise
To see whether similar cells exist in mammals, the team injected fungal particles into the blood of mice and examined which cells engulfed them. They found a rare group of circulating cells that, like zebrafish bMΦs, were efficient eaters, carried a mixture of immune and vessel‑associated surface markers and displayed tentacle‑like extensions under the microscope. These cells were distinct from known monocyte and neutrophil populations, suggesting that bMΦ‑like guardians are evolutionarily conserved. Intriguingly, they also develop and work even when a key receptor normally required for most other macrophages is missing, which may explain why they escaped attention for so long. Together, the findings redraw our picture of how blood and vessels are surveilled, hinting that one could one day harness or engineer these resident patrol cells to treat infections traveling in the blood or diseases that damage vessel linings.
What This Means for Our Health
In everyday terms, this work shows that our blood vessels host their own built‑in security guards. These guards arise in an unexpected way from the vessel wall, settle on the inside surface, and continuously inspect both the blood and the lining for trouble—removing germs and faulty cells and cleaning up after damage. By revealing this overlooked layer of protection in both fish and mice, the study opens the door to future therapies that might boost or mimic these cells to keep blood cleaner and vessels healthier in conditions ranging from bloodstream infections to vascular injury and cardiovascular disease.
Citation: Weijts, B., Demmers, J.A.A. & Robin, C. Blood vessel-resident macrophages safeguard blood and vessel integrity in zebrafish. Nat Immunol 27, 975–984 (2026). https://doi.org/10.1038/s41590-026-02481-y
Keywords: macrophages, blood vessels, immune surveillance, zebrafish, vascular biology