Clear Sky Science · en
Crystalline nitrogen chain radical anions
Why unstable nitrogen chains matter
Nitrogen gas makes up most of the air we breathe, but in certain extreme environments—like the upper atmosphere or the hearts of explosives—nitrogen can link into energetic chains that store tremendous chemical power. These chains usually fall apart almost instantly under everyday conditions, so they have been studied only fleetingly in space-like plasmas or under crushing pressures. This article reports something long thought out of reach: solid, room‑temperature crystals that safely contain a fragile four‑atom nitrogen chain, opening a new window on how such high‑energy forms of nitrogen behave and how they might one day be used as controllable sources of reactive nitrogen in synthesis and materials. 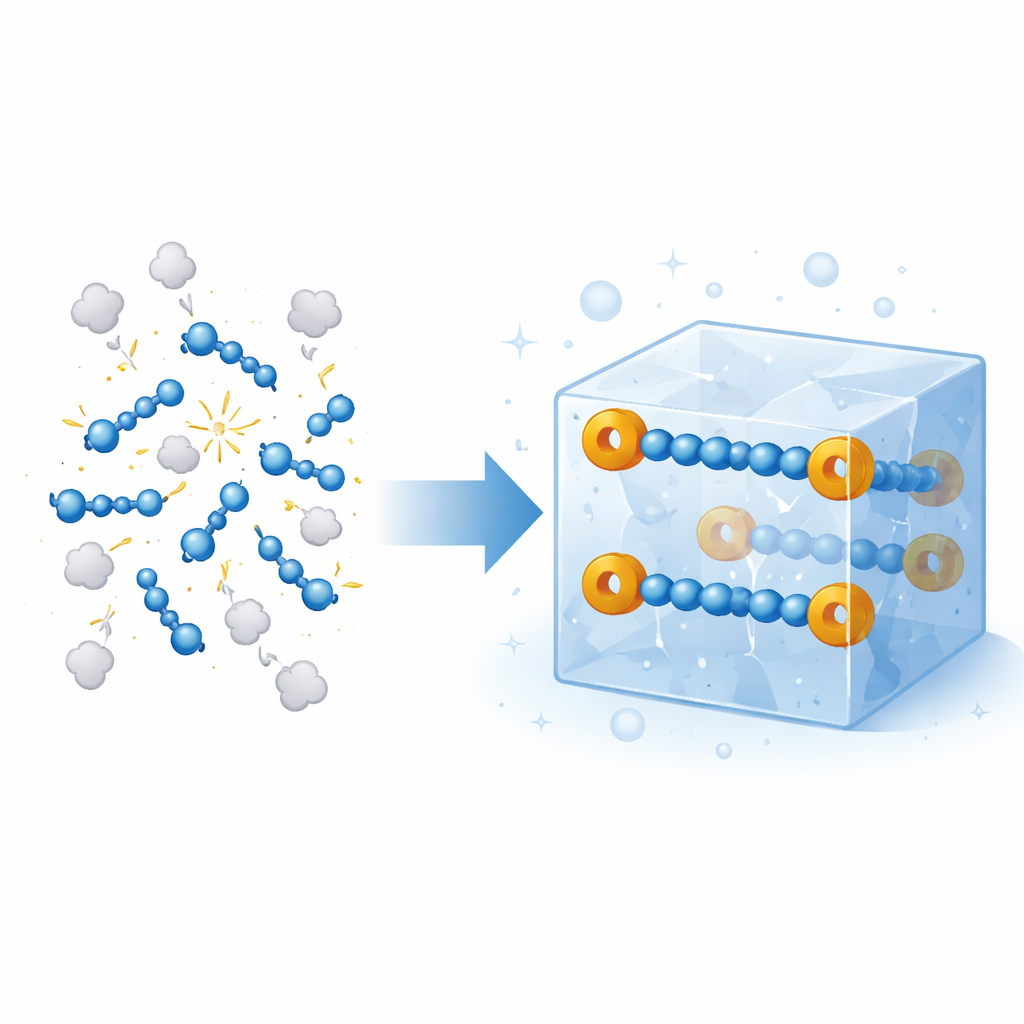
From sky chemistry to the lab bench
Long nitrogen chains containing more than three nitrogen atoms are known from the Earth’s ionosphere and from the hazy atmosphere of Saturn’s moon Titan, where intense radiation and charged particles briefly knit N2 molecules into exotic ions and radicals. Similar nitrogen chains have been spotted inside dense solids squeezed at enormous pressures, and they are attractive candidates for advanced propellants and explosives because they tend to "snap back" to very stable N2 gas, releasing energy. The catch is that this same tendency makes them extremely short‑lived and dangerous under normal conditions. Chemists have tried to tame them by binding them to metal atoms or by burying them inside bulky organic groups, but truly "free" nitrogen chains carrying an extra electron—a radical anion—have remained too reactive to isolate in a bottle.
Building a stable four‑nitrogen chain
The authors set out to create a robust version of a four‑nitrogen radical anion, written {N4}•−, without relying on metal atoms to hold it in place. They started from simple aryl azides, common laboratory compounds that already contain a three‑nitrogen fragment. By reducing these azides in the presence of a potassium source and a crown‑like ligand that wraps the metal ion, they encourage two azide units to couple head‑to‑head, forming a linear N–N–N–N chain capped at each end by a brominated benzene ring. The resulting anion, noted as [(4‑BrC6H4)2N4]•−, crystallizes as striking black needles that are stable for weeks in the solid state if kept away from air. X‑ray diffraction shows a nearly straight chain of four nitrogen atoms with bond lengths between typical single and double bonds, indicating that the extra electron is shared (delocalized) rather than trapped on a single atom.
Seeing where the unpaired electron lives
To understand why this chain is so unusually persistent, the team combined multiple spectroscopic methods with quantum‑chemical calculations. Electron paramagnetic resonance, a technique that detects unpaired electrons, revealed that the radical character is spread across the whole molecule but is strongest at the two terminal nitrogens, which sit next to the aromatic rings. Calculated spin‑density maps confirm that most of the unpaired electron resides on the N4 unit, with the benzene rings acting like "safety valves" that help smear out and stabilize the electron, similar to how they stabilize classic benzylic radicals. Ultraviolet–visible spectroscopy and theoretical analyses further show a network of electron‑sharing orbitals that resembles that of a simple carbon chain, but built entirely from nitrogen atoms. 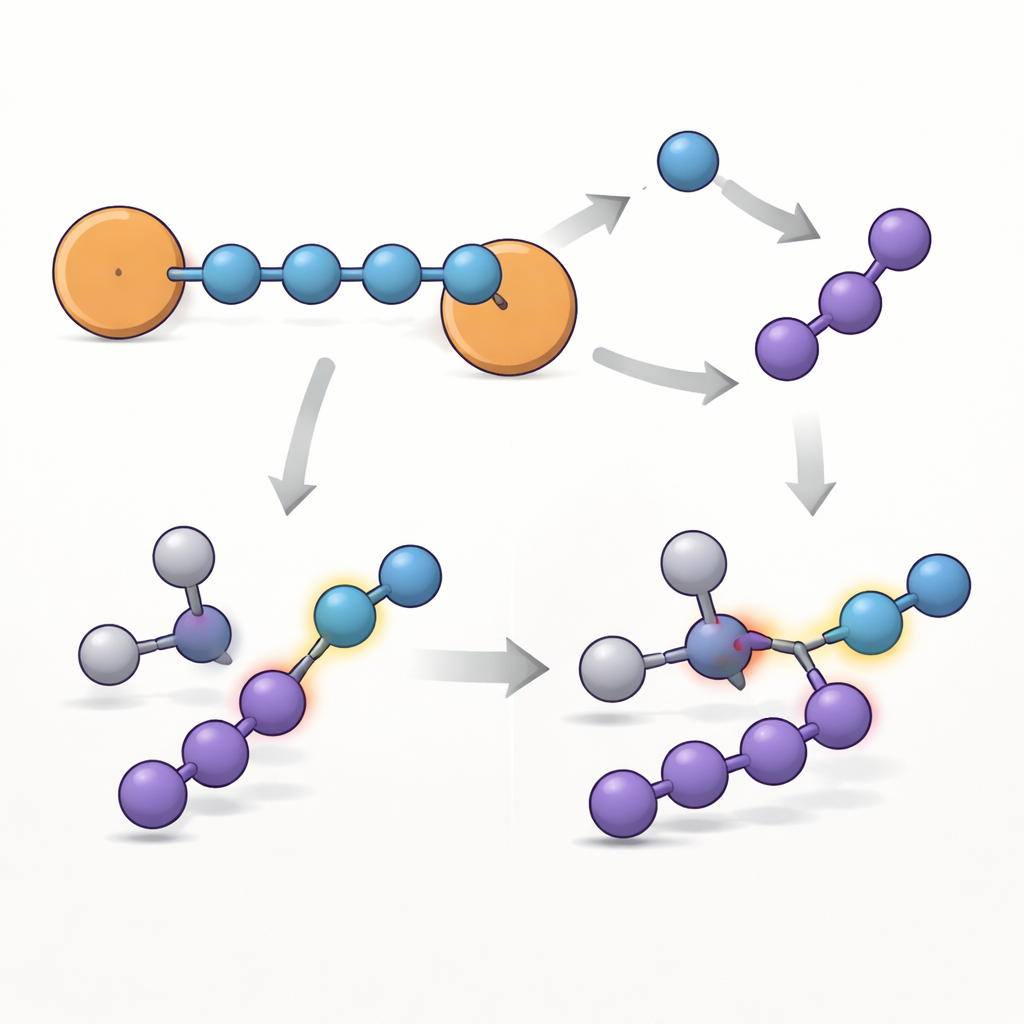
A family of chains and a new nitrogen source
The approach proved general: by changing the substituent on the aromatic rings (fluoro, chloro, methyl, or unsubstituted phenyl), the authors generated a small family of related {N4}•− molecules. All keep the same basic chain structure, but their lifetimes and the exact distribution of the unpaired electron vary with the ring substituent, showing that chemists can fine‑tune stability by simple organic modifications. The team then explored how the flagship brominated compound reacts. Under certain conditions, the four‑nitrogen chain breaks into a single‑nitrogen unit (N1) and a three‑nitrogen fragment (N3). Evidence from product analysis and calculations indicates that one pathway produces a nitrene radical anion—a highly reactive nitrogen species capable of inserting into carbon–hydrogen bonds. When reacted with an aromatic aldehyde, the system converts a carbonyl C–H bond into an amide linkage, demonstrating that this crystalline {N4}•− can serve as a storable, bench‑top source of reactive nitrogen for building more complex molecules.
What this means for future chemistry
By trapping a delicate four‑nitrogen radical chain in an organic framework that remains crystalline and stable at room temperature, this work turns an elusive atmospheric curiosity into a tangible laboratory reagent. The study shows that carefully chosen aromatic groups can delocalize charge and spin enough to suppress the usual explosive loss of N2, yet still allow the chain to fall apart in a controlled way when triggered, releasing powerful single‑nitrogen units. For non‑specialists, the key message is that chemists are learning to domesticate one of nitrogen’s wildest forms, opening the door to new high‑energy materials and to convenient, solid reagents that transfer nitrogen fragments into pharmaceuticals and other valuable products with far greater control than was previously possible.
Citation: Lister-Roberts, R., Galano, D., van IJzendoorn, B. et al. Crystalline nitrogen chain radical anions. Nat. Chem. 18, 686–694 (2026). https://doi.org/10.1038/s41557-025-02040-2
Keywords: nitrogen chains, radical anions, high-energy materials, nitrene chemistry, organo-nitrogen synthesis