Clear Sky Science · en
Riboflavin metabolism shapes FSP1-driven ferroptosis resistance
Why This Vitamin Story Matters
Most people know vitamins as something you take in the morning for general health. This study shows that one familiar vitamin, riboflavin (vitamin B2), may do something far more dramatic inside our cells: it helps decide whether stressed cells live or die through a form of iron‑driven damage called ferroptosis. Because ferroptosis is linked to cancer, neurodegeneration and heart attacks, uncovering how a common nutrient tunes this process could eventually influence how we design diets and drugs for these diseases.
A Fragile Shield Around Our Cells
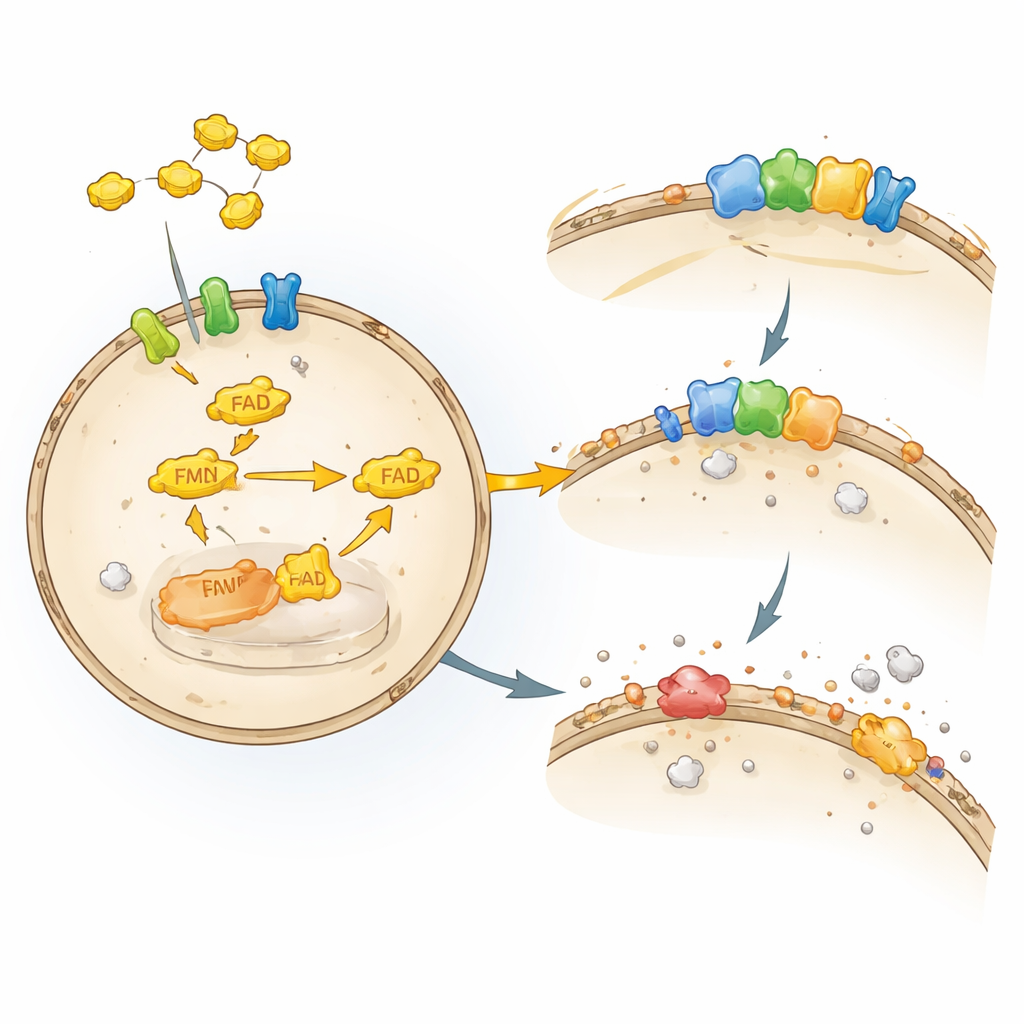
Every cell is wrapped in a thin fatty membrane that works like a combination wall and power strip. That fatty layer is easily damaged by certain forms of oxygen, in a process known as lipid peroxidation. When this damage spirals out of control, cells undergo ferroptosis, a regulated form of cell death that depends on iron and oxidized fats. Our cells deploy several defense systems to stop this cascade. One key guardian, the enzyme GPX4, uses the antioxidant glutathione to detoxify harmful lipid peroxides. Another guardian, FSP1, sits at the membrane and keeps fat‑soluble antioxidants such as coenzyme Q and vitamin K in their active, protective form, acting like a recycling station for these molecules.
Finding the Hidden Helpers of a Cell Guardian
Although FSP1 was known to be a powerful brake on ferroptosis, researchers did not know what cellular factors kept it running smoothly. The authors engineered human cancer cells so that they relied entirely on FSP1 to survive when GPX4 was disabled. They then used a focused CRISPR–Cas9 gene‑editing screen, knocking out about 3,000 drug‑relevant genes to see which losses made the cells suddenly vulnerable. Among the strongest hits were enzymes and transporters involved in handling riboflavin, especially riboflavin kinase (RFK) and flavin adenine dinucleotide synthase (FADS), which build the cofactor FAD from riboflavin. When these genes were disrupted, FSP1 protein levels dropped, lipid damage surged, and cells died by ferroptosis, revealing a direct upstream dependence of FSP1 on riboflavin‑derived cofactors.
Vitamin Supply as a Dial for Cell Survival
To test whether the nutrient itself, not just the enzymes, mattered, the team grew cells in media lacking riboflavin or containing amounts closer to those in human blood, which are far lower than in standard lab solutions. Under riboflavin‑poor conditions, many flavin‑dependent proteins decreased, but FSP1 was among the most strongly depleted, without a similar drop in its messenger RNA. The cells became far more sensitive to compounds that block GPX4, and their membranes accumulated oxidized phospholipids, a hallmark of ferroptosis. Importantly, simply restoring riboflavin brought FSP1 protein back and reduced lipid damage. Even modest shifts in riboflavin within a physiologic range tuned how much FSP1 protection cells could muster, suggesting that everyday variations in vitamin B2 status may quietly influence how tissues cope with oxidative stress.
Turning a Helpful Vitamin Against Cancer
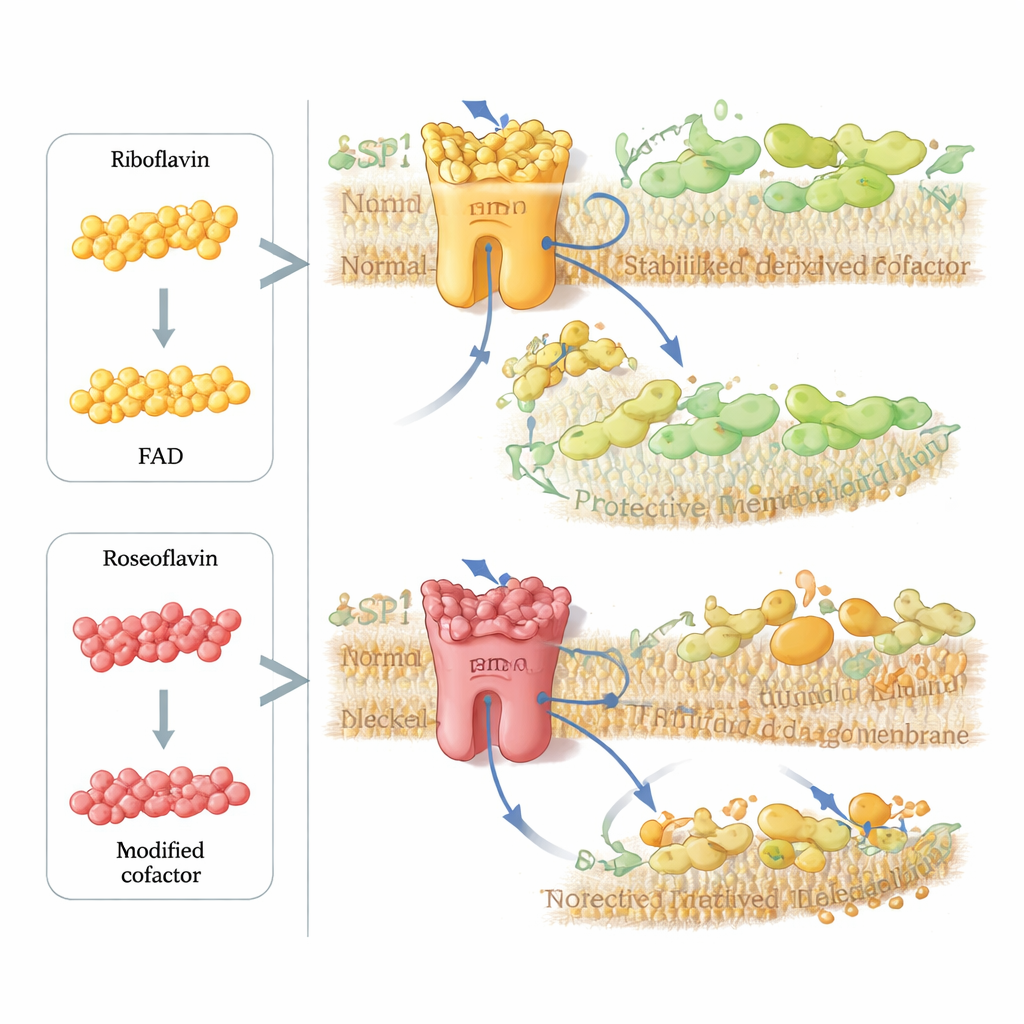
The researchers then asked whether they could deliberately sabotage this protective pathway in cancer cells. They turned to roseoflavin, a natural antibiotic made by certain bacteria that closely resembles riboflavin but carries a small chemical change. Cells take up roseoflavin and convert it into a modified version of FAD that can fit into FSP1 and stabilize the enzyme’s structure, but cannot carry electrons properly. In purified enzyme tests and computer simulations, FSP1 loaded with roseoflavin’s cofactor was essentially powerless. In multiple cancer cell lines, tiny, nanomolar doses of roseoflavin sharply boosted the killing power of GPX4 inhibitors, but only when FSP1 was present, showing that the drug specifically cripples this membrane‑repair circuit.
What This Means for Health and Therapy
Taken together, the work recasts riboflavin from a simple nutrient into a master regulator of a critical cell‑death switch. By feeding into the FAD cofactor, riboflavin controls the stability and activity of FSP1, which in turn governs how effectively cells recycle lipid‑soluble antioxidants and resist membrane damage. Too little riboflavin weakens this shield and promotes ferroptosis; cleverly designed analogues like roseoflavin can hijack the same pathway to disable FSP1 on demand. For a layperson, the takeaway is that micronutrients do more than “support metabolism”—they can hard‑wire whether cells survive oxidative stress. This insight opens new avenues to sensitize tumors to therapy, refine antioxidant strategies, and better understand how diet might influence diseases where ferroptosis plays a central role.
Citation: Skafar, V., de Souza, I., Ghosh, B. et al. Riboflavin metabolism shapes FSP1-driven ferroptosis resistance. Nat Cell Biol 28, 696–706 (2026). https://doi.org/10.1038/s41556-025-01856-x
Keywords: ferroptosis, riboflavin, FSP1, lipid peroxidation, cancer therapy