Clear Sky Science · en
Predictors of motor outcome with pallidal stimulation for Parkinson’s disease from the CSP468 cohort
Why this study matters for people with Parkinson’s
For people living with Parkinson’s disease, deep brain stimulation (DBS) can dramatically ease movement problems when medications are no longer enough. Yet some patients benefit a lot while others improve only modestly. This study asks a simple but crucial question: does the exact spot where surgeons place DBS electrodes inside the brain explain who does better—and can we use that knowledge to plan surgery and adjust treatment more precisely?
A closer look at brain stimulation for movement
DBS works by delivering tiny electrical pulses through implanted electrodes to specific deep brain regions involved in movement control. One common target is the globus pallidus interna (GPi), a small structure that helps regulate how smoothly we move. Large trials have already shown that GPi-DBS can ease tremor, stiffness, and slowness in Parkinson’s disease. But even in carefully run clinical studies, patients’ movement scores after surgery vary widely. Researchers have long suspected that three things might matter most: how well a person responds to the drug levodopa, their overall health and symptoms before surgery, and the exact brain area reached by the electrical current.
Finding the brain’s “sweet spot” for stimulation
The authors focused on whether stimulating a specific “sweetspot” within the GPi predicts better movement after surgery. They used advanced brain imaging and computer modeling to estimate the volume of brain tissue activated by each patient’s DBS settings—essentially, the area that actually receives stimulation. They then compared this activated volume with a previously mapped region inside the GPi that had been linked to good outcomes in earlier work. The main dataset came from a large, multicenter Veterans Affairs study (CSP #468) in which patients with Parkinson’s were randomly assigned to DBS in one of two brain targets and then evaluated by examiners who did not know which surgery they had received.
Testing the findings in two independent patient groups
To avoid building a model that simply “recognized” its own data, the team created and tested sweetspots in two separate patient cohorts. First, they analyzed 86 patients from the CSP #468 trial who had GPi-DBS and high-quality scans of their electrode positions. Second, they used imaging and follow-up exams from 51 additional patients who received GPi-DBS from a single surgeon at another center between 2014 and 2024. For each group, they generated a three-dimensional sweetspot inside the GPi and calculated, for every patient, how much their activated volume overlapped this zone. They then built statistical models that combined sweetspot overlap with clinical factors such as levodopa response, hours spent in an “off” state, age at onset, and tremor severity.
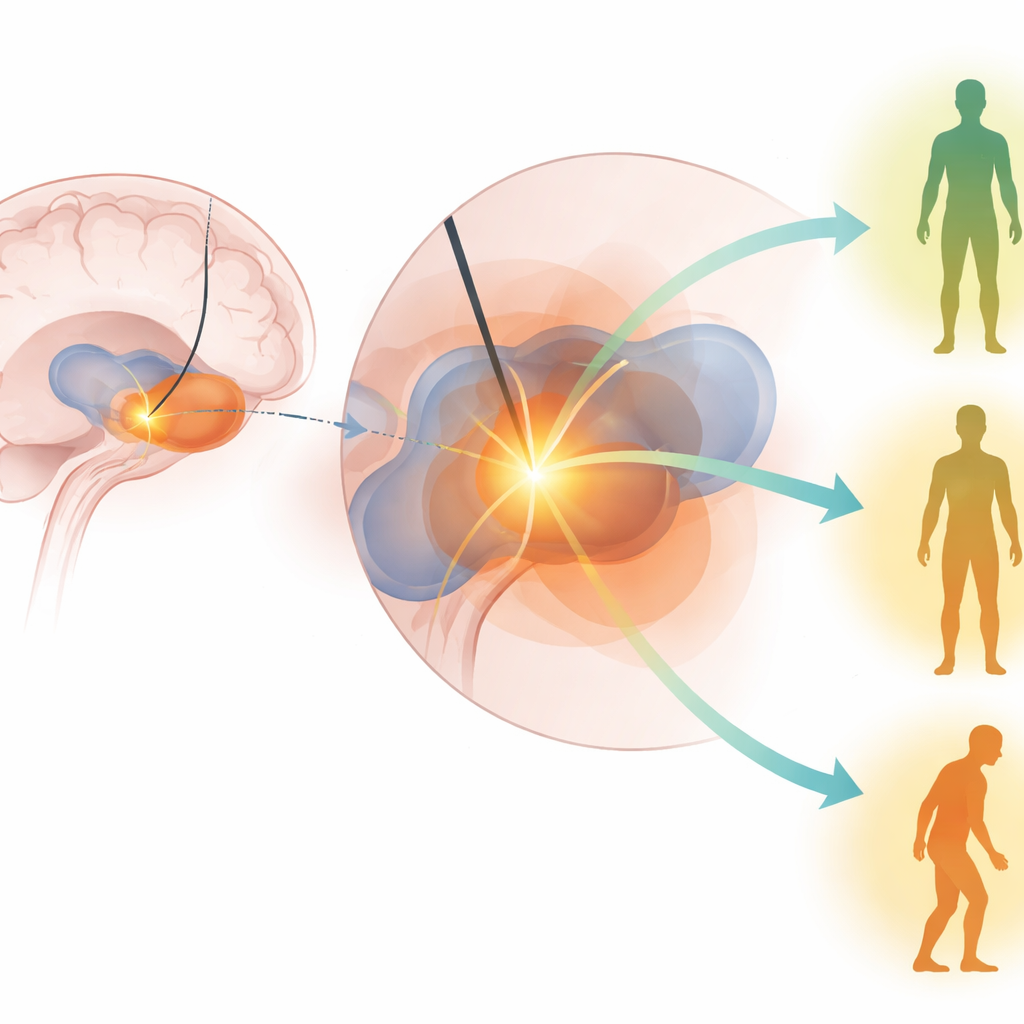
What actually predicted better movement
Across both cohorts and multiple analyses, two predictors clearly stood out. Patients who had a stronger response to levodopa before surgery tended to have better movement after GPi-DBS. Just as importantly, patients whose stimulation activated more of the GPi sweetspot showed greater improvement in their motor scores six months after surgery. Other factors—such as age, duration of disease, detailed cognitive scores, and even tremor burden—were less reliable once the key predictors were taken into account. Notably, models built from one cohort could still predict outcomes in the other group, suggesting that the identified GPi sweetspot is not a fluke of a single hospital or surgeon but a meaningful therapeutic zone shared across populations.
What this means for patients and clinicians
For people considering GPi-DBS, this study reinforces two practical messages. First, how well your symptoms respond to levodopa remains a strong indicator of how much your movement may improve after surgery. Second, the finer details of where DBS electrodes sit inside the GPi—and how closely their stimulation overlaps a specific motor control region—can significantly influence results. Although the models explain only part of the variation in outcome, they show that precise targeting of a common GPi sweetspot, combined with careful assessment of levodopa response, can help surgeons and neurologists better select candidates, plan surgery, and fine-tune stimulation settings to give more patients their best chance at smoother, more reliable movement.
Citation: D’Souza, S., Batheja, A., Chen, J. et al. Predictors of motor outcome with pallidal stimulation for Parkinson’s disease from the CSP468 cohort. npj Parkinsons Dis. 12, 107 (2026). https://doi.org/10.1038/s41531-026-01312-z
Keywords: Parkinson’s disease, deep brain stimulation, globus pallidus interna, motor outcomes, neurosurgery targeting