Clear Sky Science · en
Antiviral innate immunity induces alpha synuclein phosphorylation at serine129 in neurons independent of aggregation
Why Viruses and Parkinson’s Might Be Linked
Many people who recover from serious viral infections later develop problems with memory, movement, or mood. At the same time, brain diseases such as Parkinson’s are marked by abnormal clumps of a protein called alpha‑synuclein inside nerve cells. This study asks a pressing question for patients and families: when the brain fights off a virus, does that very immune response nudge alpha‑synuclein toward the kinds of changes that can, over years, set the stage for Parkinson’s‑like disorders?
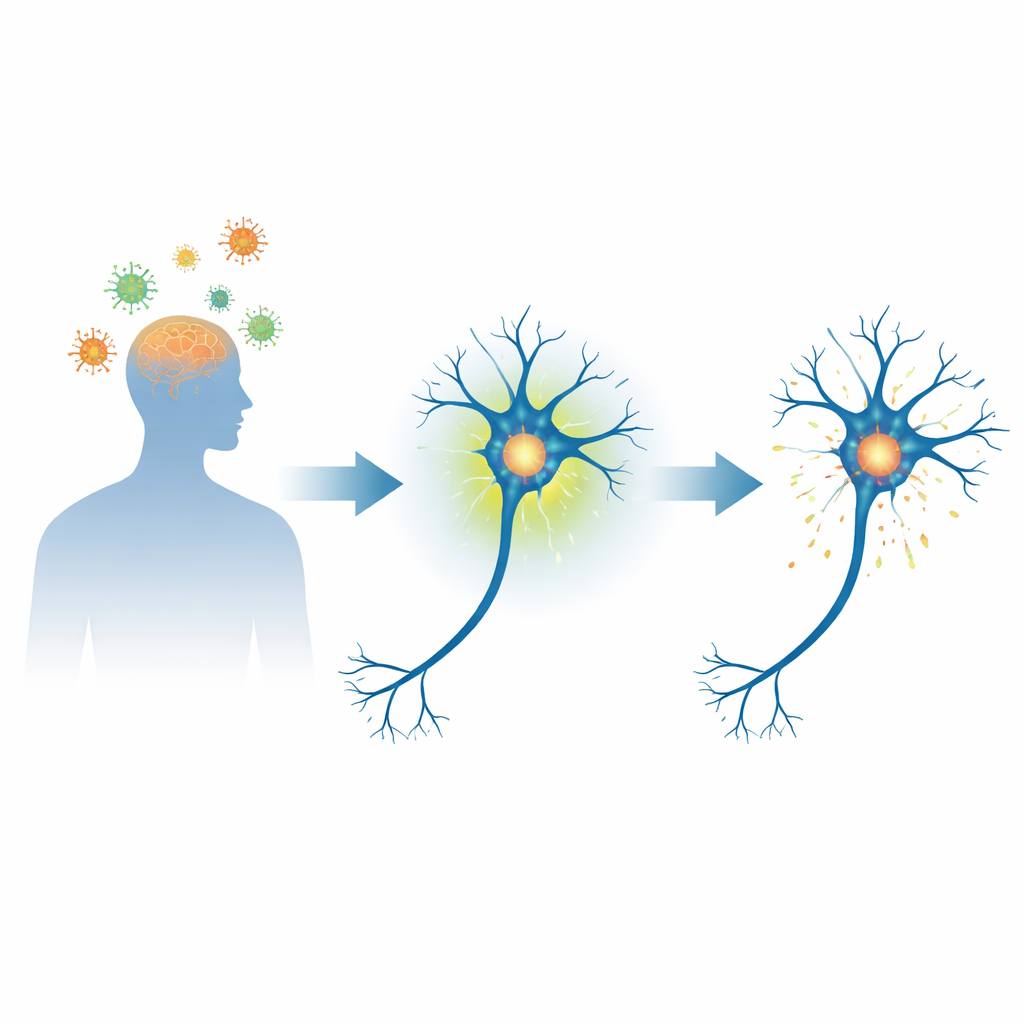
A Nerve Cell Protein at the Center of Brain Disease
Alpha‑synuclein is a small, flexible protein that is abundant in nerve cells and helps regulate how signals are passed between them. In Parkinson’s disease and related conditions, this protein can misfold and pile up into dense deposits known as Lewy bodies, which are closely linked to nerve cell death. A key chemical tag on this protein, called phosphorylation at a specific site (serine 129), is found on most of the alpha‑synuclein inside these deposits. That has made scientists wonder whether this tag is merely a marker of damage or an early step on the road to disease.
Clues from People and Animals with Brain Infections
The researchers first examined brain tissue from people who had died with severe West Nile virus brain infection. Compared with control brains, the infected brains showed more alpha‑synuclein carrying the serine‑129 tag inside neurons, especially in areas important for movement, and these same cells showed signs of active antiviral signaling. To test cause and effect, the team then developed a mouse model in which West Nile virus travels from the nose into the brain along the smell pathways. As the virus spread, the animals lost weight, switched on strong antiviral genes, and—crucially—showed a surge of tagged alpha‑synuclein in the olfactory bulbs, even though the protein had not yet formed insoluble clumps.
Viral Triggers Without Lasting Clumps
To see whether this was a general response, the scientists looked at other viruses. A DNA virus that causes a shingles‑like illness in monkeys, and herpes simplex virus in mice, also boosted tagged alpha‑synuclein in nerve clusters serving the skin and face. In dishes of mouse brain cells, West Nile virus infection rapidly raised levels of tagged alpha‑synuclein within hours, but these levels then fell back down, and sensitive tests detected no new aggregates. Even mimicking viral genetic material with a synthetic molecule, without using real virus, was enough to temporarily increase the tagged form of the protein in neurons, again without creating clumps.
Interferon: The Messenger that Switches the Tag On
What tied all these observations together was the brain’s frontline alarm system: type I interferon, a family of proteins nerve cells release when they sense viral genetic material. In cultured neurons, both actual infection and the viral mimic caused the cells to secrete this kind of interferon, and direct treatment with interferon alone quickly doubled tagged alpha‑synuclein levels within 30 minutes. This spike was short‑lived and still not accompanied by aggregates. Strikingly, when the team repeated the experiments in neurons genetically lacking the interferon receptor, neither virus nor the RNA mimic could raise tagged alpha‑synuclein. That shows the chemical tag is added downstream of interferon signaling, not by the virus itself.
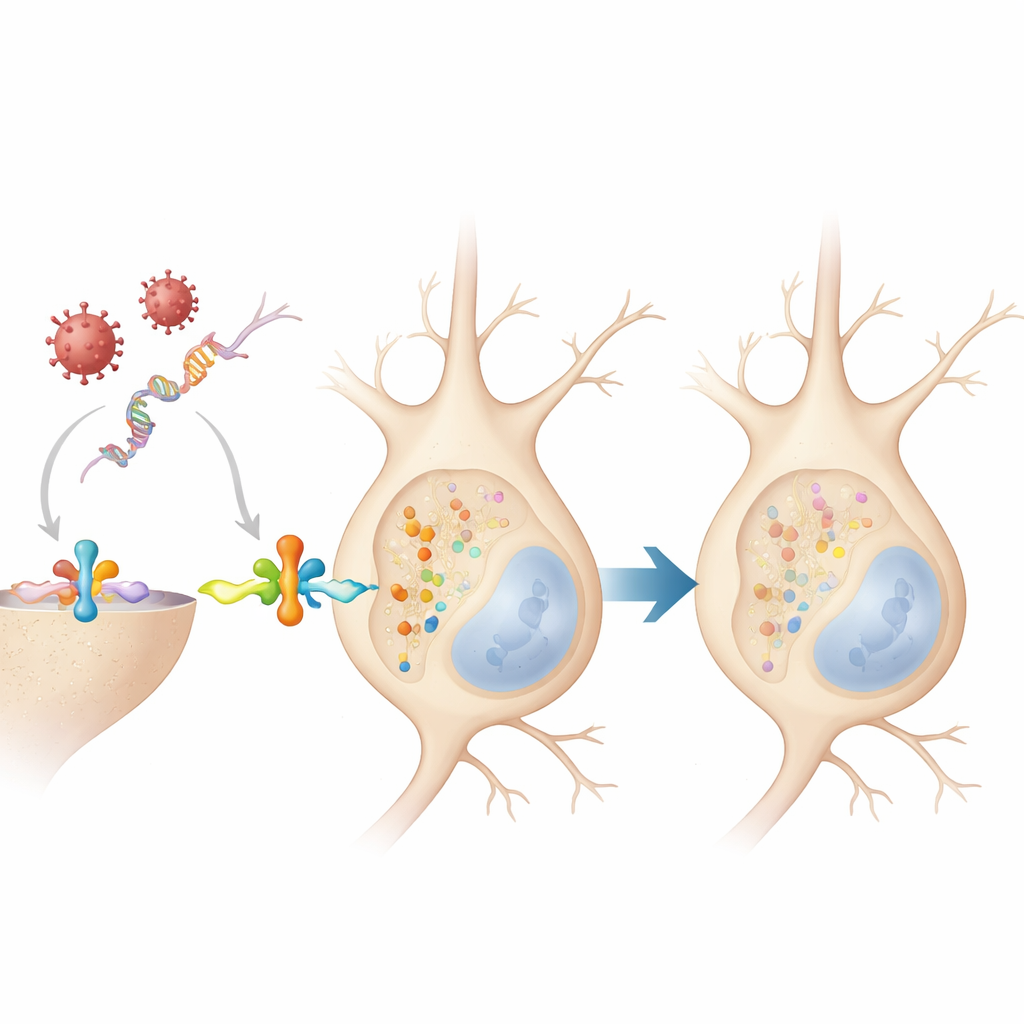
What This Means for Long‑Term Brain Health
Taken together, the work suggests that when neurons mount an antiviral defense, they briefly modify alpha‑synuclein in a way that is normally reversible and does not immediately create toxic clumps. However, because this response appears across different species and diverse viruses, repeated infections or chronic inflammation over a lifetime could repeatedly flip this molecular switch. If the systems that normally clear tagged alpha‑synuclein become overloaded or fail with age, these harmless, fleeting changes might start to accumulate, nudging the protein toward the aggregates seen in Parkinson’s and related diseases. In this view, alpha‑synuclein tagging is not just a sign of damage but part of the brain’s early response to infection—a response that, under the wrong conditions, may slowly turn from protective to harmful.
Citation: Heiden, D.L., Merrick, C., Evans, R.C. et al. Antiviral innate immunity induces alpha synuclein phosphorylation at serine129 in neurons independent of aggregation. npj Parkinsons Dis. 12, 80 (2026). https://doi.org/10.1038/s41531-026-01297-9
Keywords: Parkinson’s disease, alpha-synuclein, viral infection, interferon, neuroinflammation