Clear Sky Science · en
Chronic adaptive deep brain stimulation in Parkinson’s disease: ADAPT-START findings and programming principles
Why Smarter Brain Pulses Matter
For many people with Parkinson’s disease, tiny electrical pulses delivered deep inside the brain can dramatically ease stiffness and shaking. Yet this treatment, called deep brain stimulation, is still something of a blunt tool: the current is usually kept constant all day long, even though symptoms and brain activity change from hour to hour. This study reports on a more flexible, “adaptive” form of stimulation that listens to brain signals in real time and automatically adjusts the intensity of the pulses. The authors describe how they set up this technology in everyday clinical practice and what it has meant so far for patients’ movement, especially walking.
From Always-On to Just-Right Stimulation
Traditional deep brain stimulation works like leaving a light on at a fixed brightness. Electrodes placed in a tiny region called the subthalamic nucleus are connected to a pacemaker-like device in the chest, which continuously sends electrical pulses using preset settings. This method can greatly improve tremor and stiffness, but some symptoms, particularly problems with balance and walking, often remain. Patients’ movements also vary with medication, sleep, and daily activities, while the stimulation does not. As a result, the “always-on” approach can sometimes under-treat symptoms or cause side effects such as involuntary movements.
Listening to the Brain’s Rhythms
Newer implants do more than send electricity—they can also record local brain activity. In Parkinson’s disease, a particular rhythm known as beta activity becomes stronger when patients are slow and rigid, and typically weakens when they move more freely or take medication. The idea behind adaptive stimulation is to use this rhythm as a guide: when beta activity rises, the device increases current; when it falls, the device eases off. The system tested here uses a “dual threshold” design. Clinicians define a lower and an upper band of acceptable beta power. If the brain signal moves above or below these bands for long enough, the device slowly ramps the stimulation up or down within safe limits that have been set for each patient.
Trying Adaptive Stimulation in Real Life
Between January and April 2025, the team offered this dual-threshold approach to 20 people with Parkinson’s disease who already had conventional stimulation using a sensing-enabled device. Not all could use the new mode: several had noisy recordings, lacked a clear beta rhythm, or required settings incompatible with sensing. Nine patients were ultimately switched to adaptive stimulation, and five completed at least a month of stable treatment. Setting things up involved three structured clinic visits. First, doctors mapped each patient’s brain signals and current settings, then sent them home for several days while the implant passively recorded beta activity during normal life. In the next visit, they defined safe minimum and maximum current levels and chose beta thresholds based on these recordings. A follow-up visit two weeks later was used to fine-tune the system, using both symptom checks and data from a belt-worn motion sensor that tracked walking and periods of good or poor movement across the day.
What Changed for Patients
After one month on adaptive stimulation, the five patients showed, on average, about 35 percent more improvement in a standard motor score compared with their previous optimized conventional settings, even though those original settings were already working well. Gait-related items and a questionnaire focused on freezing of gait showed the clearest gains. Wearable sensors suggested that patients spent over 40 extra minutes per day in a good movement state without troublesome involuntary movements, and up to 160 fewer minutes per day in poor “OFF” periods. Most measures of non-motor symptoms also improved modestly. Interestingly, an overall quality-of-life questionnaire sometimes worsened, even though patients and caregivers strongly preferred the adaptive mode. The authors suspect this may reflect people becoming more active—and thus more aware of remaining limitations—rather than a true decline.
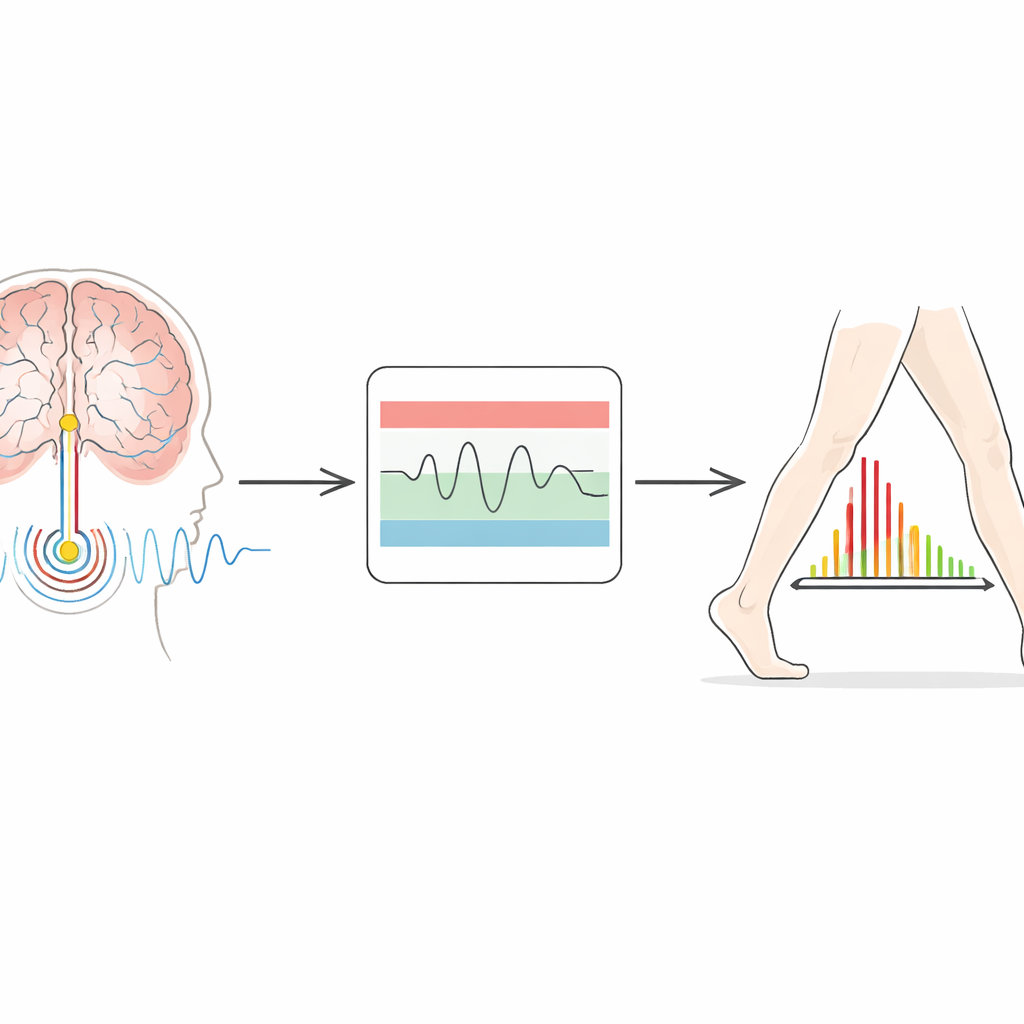
Challenges and Next Steps
Despite encouraging results, the authors stress that adaptive stimulation is not plug-and-play. Only about half of the screened patients could use the dual-threshold mode because of technical constraints, such as signal contamination by heartbeats or the need for certain electrode configurations. Even for eligible patients, repeated visits were often needed to find a stable brain rhythm to track and to adjust the thresholds so that stimulation truly adapted instead of behaving like a simple two-level switch. On average, adaptive therapy delivered slightly more electrical energy than conventional therapy, but not continuously; instead, current rose mainly when symptoms were likely to be worse. Future improvements, such as better signal processing, more precise brain biomarkers for different symptoms, and remote fine-tuning, could make this approach more widely usable and less burdensome.
What This Means for People with Parkinson’s
This early experience suggests that a brain implant that “listens and responds” may offer extra benefits beyond today’s constant-output stimulators, especially for walking and daily movement fluctuations. In practical terms, adaptive stimulation allowed many patients to spend more of their day moving well, without adding noticeable side effects, and it was consistently favored over the older approach. At the same time, the method currently demands careful patient selection, detailed setup, and ongoing adjustment by specialists. As the technology and algorithms mature, adaptive deep brain stimulation could become a more personalized, responsive therapy that follows the brain’s needs in real time rather than forcing patients to fit a fixed pattern of stimulation.
Citation: Cascino, S., Luiso, F., Caffi, L. et al. Chronic adaptive deep brain stimulation in Parkinson’s disease: ADAPT-START findings and programming principles. npj Parkinsons Dis. 12, 85 (2026). https://doi.org/10.1038/s41531-026-01269-z
Keywords: Parkinson’s disease, deep brain stimulation, adaptive neuromodulation, brain sensing, gait and movement