Clear Sky Science · en
Transient remodeling of gut metabolism supports juvenile growth and adult fitness in Drosophila
How Growing Guts Shape a Whole Life
Childhood is a time of rapid growth, and that is as true for fruit flies as it is for humans. This study explores how a young animal’s gut briefly shifts into “high gear” to fuel a final growth spurt—and how that short-lived change leaves lasting marks on adult health. By tracking development in the fruit fly Drosophila, the authors reveal a temporary boost in intestinal metabolism that helps juveniles grow fast, store energy, withstand harsh environments, and reproduce successfully as adults.
A Sudden Sprint in a Short Life
Fruit flies pass through three larval stages before transforming into adults. The researchers followed body size and body chemistry across these stages and found that growth is not steady. Instead, it speeds up dramatically halfway through larval life, in the last stage known as L3. During just a couple of days, larval volume and key building blocks like protein, sugars, and fats multiply many times over, signaling an intense growth spurt that must be supplied with large amounts of fuel.
The Gut Turns Into a Fat-Processing Factory
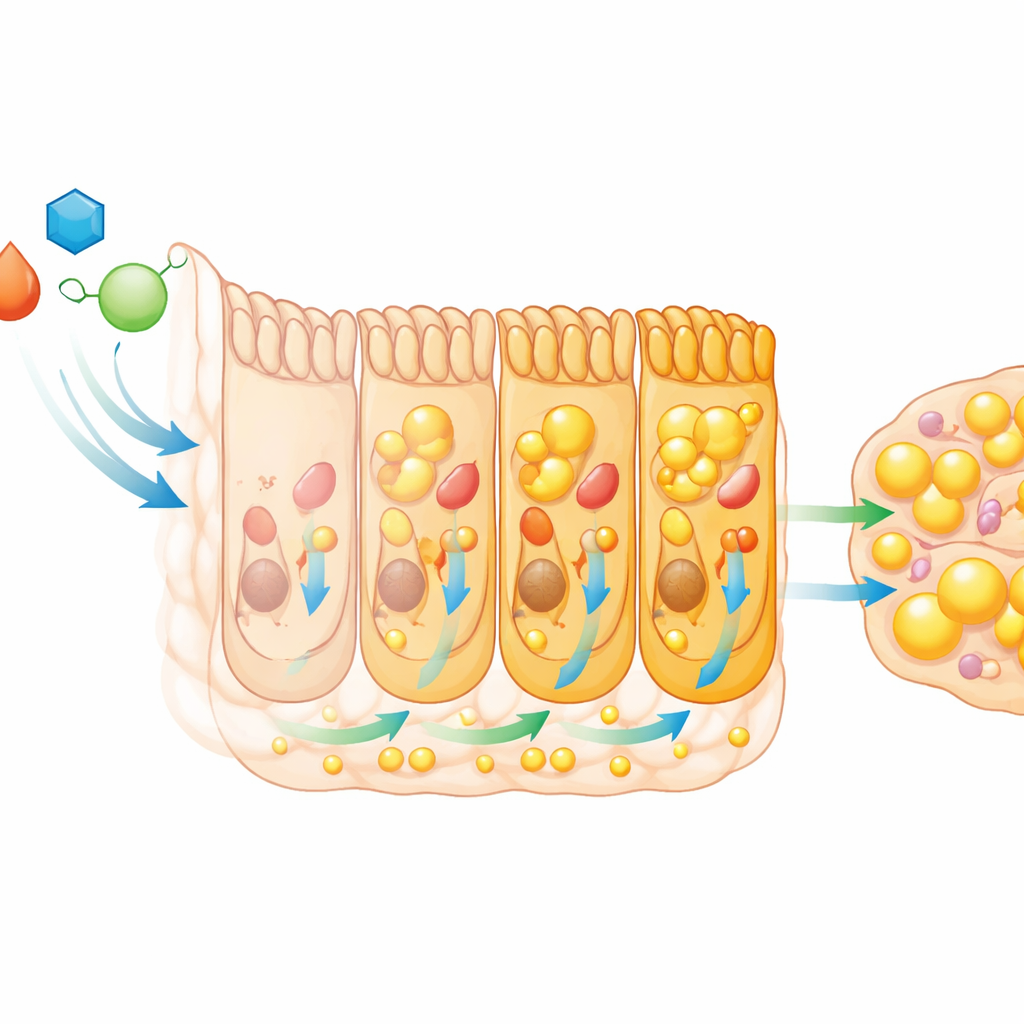
Because growth depends on food, the team examined whether the intestine changes its behavior at this critical time. By sequencing messenger RNA from the midgut—the fly equivalent of the small intestine—they discovered a sweeping reprogramming of gene activity when larvae enter the L3 stage. Hundreds of genes switch on or off, especially those involved in breaking down and handling dietary fats. Enzymes that digest fat become more abundant, and intestinal cells increase their ability to burn, store, and, crucially, export fat to the rest of the body. Measurements of gut enzymes and metabolites confirmed that fat digestion and mobilization intensify in L3 larvae, and circulating fat in the blood-like fluid rises accordingly. Carbohydrate digestion also ramps up, further feeding this energy surge.
Hormones That Tell the Gut When to Shift Gears
The timing of this intestinal makeover is not random. Two nuclear receptors—proteins that turn genes on or off in response to hormonal signals—play central roles. One is the receptor for the steroid hormone ecdysone, which coordinates developmental transitions such as molting and metamorphosis. The other is HNF4, a conserved regulator of lipid metabolism. In the L3 gut, ecdysone signaling turns on just as fat-handling genes are induced. When the researchers blocked ecdysone function specifically in intestinal cells, the normal rise in lipid-metabolism genes failed to occur. Fat piled up inside the gut while levels in the circulation fell, showing that the gut could no longer export lipids efficiently.
A Partnership for Growth and Energy Storage
HNF4 emerges as a key partner in this process. Its activity increases in regions of the larval gut rich in lipids, and nearly half of the fat-metabolism genes that rise in L3 depend on HNF4 for full induction. Silencing HNF4 in the gut led to the same pattern seen with disrupted ecdysone signaling: fat was trapped in intestinal cells, circulating lipids dropped, and overall body fat after the L3 stage was reduced. The data suggest that ecdysone boosts HNF4 levels, which in turn activates a battery of genes that transform the gut into a powerful conduit for dietary fat, prioritizing export to the rest of the body over local storage. Yet, HNF4 alone cannot fully compensate if ecdysone is blocked, indicating that multiple hormone-driven pathways jointly shape this metabolic switch.
From Juvenile Gut to Adult Success
The consequences of this fleeting intestinal program reach far beyond the larval stage. When ecdysone or HNF4 were impaired in the gut, larvae grew more slowly during L3, accumulated less protein, and reached the size needed for metamorphosis later, delaying their transformation. Adults that developed from larvae lacking proper gut remodeling emerged smaller, leaner, and with reduced fat reserves. These adults were more sensitive to drying out and, in females, had smaller ovaries and produced fewer eggs in early life. Thus, a brief window of boosted gut metabolism in youth stocks the body with the material needed to build a robust adult, resist environmental stress, and reproduce quickly.
Why This Matters Beyond Fruit Flies
This work shows that early-life changes in a single organ can shape growth, timing of puberty-like transitions, and fitness later on. In Drosophila, a hormone-guided burst of gut activity during a narrow developmental window powers a final growth spurt and fills energy stores that later support waterproofing of the body and egg production. Because many of the same molecular players and gut behaviors are conserved in mammals, including humans, these findings highlight how temporary shifts in nutrition and metabolism during childhood might have long-term consequences for health and disease risk across the life span.
Citation: Lefranc, C., Fichant, A. & Storelli, G. Transient remodeling of gut metabolism supports juvenile growth and adult fitness in Drosophila. Nat Commun 17, 3458 (2026). https://doi.org/10.1038/s41467-026-71776-3
Keywords: Drosophila development, gut metabolism, lipid digestion, steroid hormones, juvenile growth