Clear Sky Science · en
Sn catalyst reconstruction and microenvironment modulation for efficient amino acid electrosynthesis via C–N coupling
Turning Waste into Useful Building Blocks
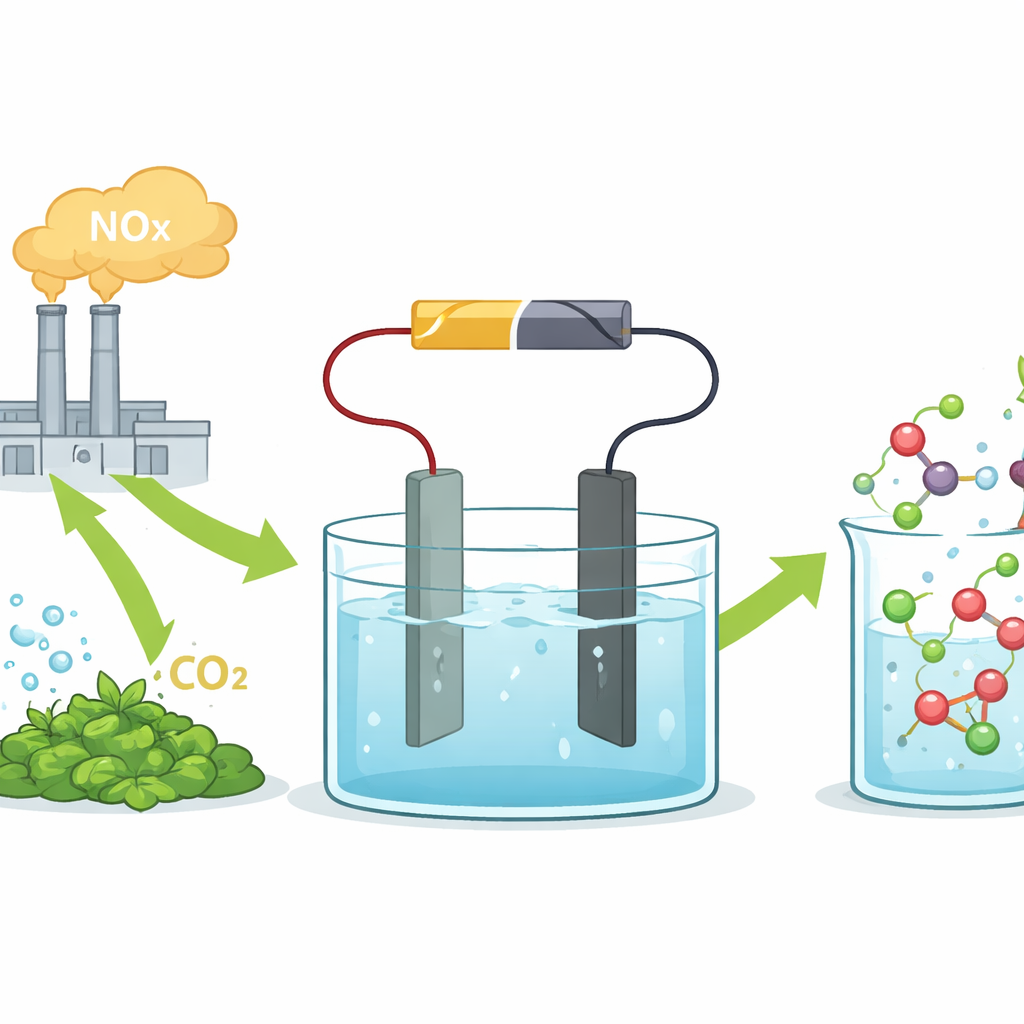
Amino acids, the tiny molecules that make up proteins, are essential ingredients for medicines, animal feed, and food supplements. Today, many amino acids are made using routes that rely on toxic chemicals and energy-hungry processes. This study explores a different path: using electricity and common industrial waste streams to make glycine, the simplest amino acid, in a cleaner, more efficient way.
Why Rethinking Amino Acid Production Matters
Conventional methods for making amino acids, such as the Strecker process, depend on cyanide and ammonia, both of which come with safety and climate concerns. Biological routes using enzymes or microbes can be gentler, but they are often slow, costly to run, and tailored to only a narrow range of products. With the world seeking greener chemistry powered by renewable electricity, there is intense interest in finding new ways to join carbon- and nitrogen-containing building blocks into amino acids without hazardous reagents or high temperatures and pressures.
Using Electricity to Build Glycine from Simple Acids
The researchers focus on electrosynthesis, in which an applied voltage drives chemical reactions in a liquid solution. They feed the system with nitric acid, which can be derived from industrial NOx exhaust, and oxalic acid, which can be made from carbon dioxide or biomass. At the heart of their setup is a tin-based electrode that selectively steers the reaction toward glycine instead of unwanted byproducts or hydrogen gas. In a simple lab cell, this tin catalyst converts the two acids to glycine with a high selectivity and at current densities approaching those used in industry, demonstrating that the approach is not just a laboratory curiosity.
How the Catalyst Surface and Local Conditions Do the Work
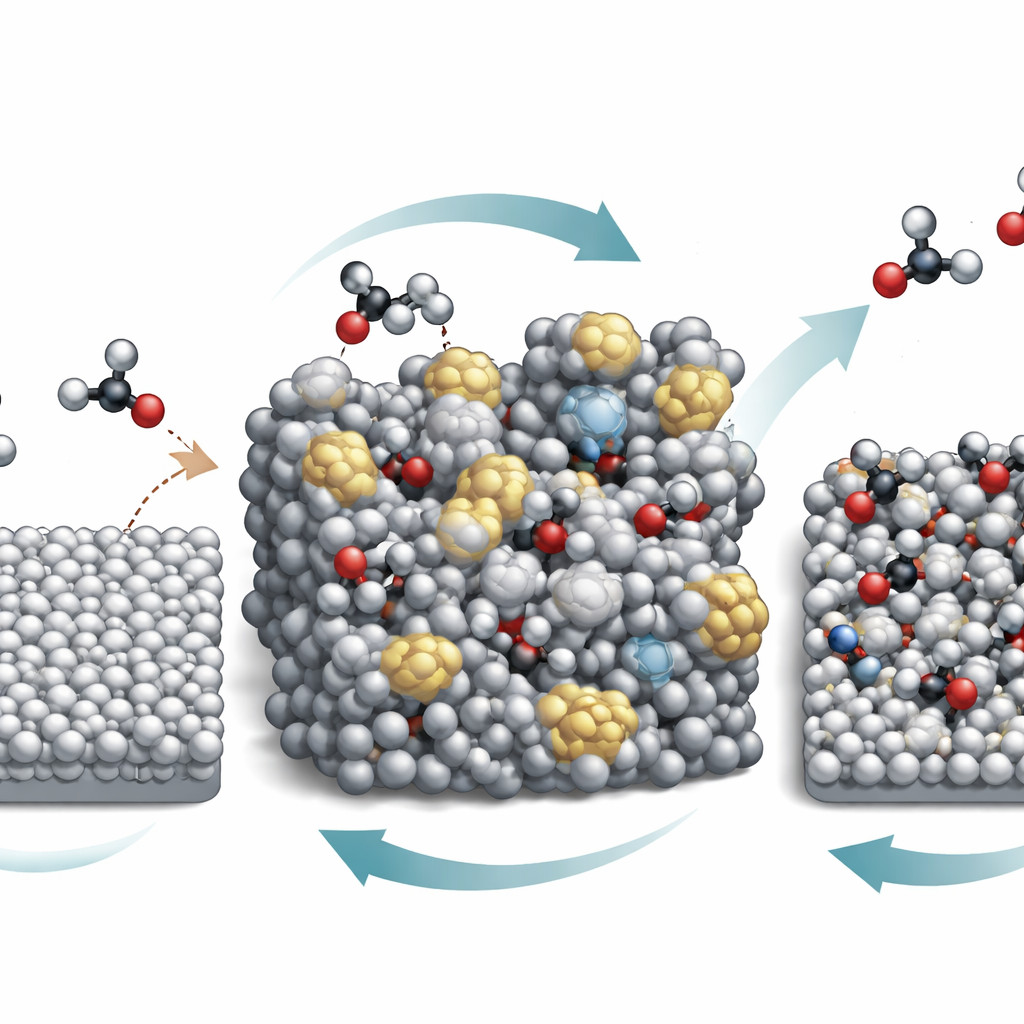
A key finding is that the tin surface does not stay static while it works. Under strongly acidic conditions, tin and nitric acid undergo a sort of valence "breathing" cycle: tin partially oxidizes and then is reduced back by the applied voltage. This continuous back-and-forth gradually breaks down the ordered crystal structure and produces an amorphous, disordered tin surface. Advanced probes, including X-ray and vibrational techniques used while the reaction is running, show that this amorphous form binds crucial carbon–nitrogen intermediates more strongly than the original smooth surface. At the same time, the vigorous reaction raises the pH right at the electrode, even though the surrounding solution remains strongly acidic, so that weak acids like oxalic acid and its derivatives exist mostly in a negatively charged form near the surface.
From Slow Chain Reactions to Fast Surface Chemistry
These changes in both the catalyst and its microenvironment flip the reaction pathway. At first, intermediates such as glyoxylic acid and its oxime are formed in the liquid and then slowly converted to glycine through a chain of steps, leading to modest yields. As the tin becomes amorphous and the local pH increases, the negatively charged intermediates stick more tightly to the surface and are hydrogenated directly at the interface instead of drifting into the solution. The authors show that this surface-driven route is faster and more selective, pushing the fraction of electrical current that ends up in glycine above 90% when operated in a specially designed flow reactor. They also demonstrate that related molecules, such as other small keto acids, can be converted to their corresponding amino acids, hinting that the strategy is broadly applicable.
Scaling Up to Practical Production
Guided by their mechanistic insights, the team builds a flow cell that continuously circulates the acid mixture past a pre-activated amorphous tin electrode. By tuning the flow rate and feed composition to preserve the beneficial local environment, they reach industrially relevant current densities while keeping glycine selectivity high. Because the process uses only the reactant acids and no extra supporting salts, the final product can be isolated simply by removing water, yielding nearly pure glycine. A basic economic analysis suggests that, if powered by inexpensive electricity, this method could produce glycine at roughly half the current market cost, especially if coupled to low-temperature plasma systems that turn air and water into nitric acid on-site.
A Cleaner Road to Everyday Molecules
Overall, the study shows that carefully managing both the dynamic state of a catalyst and the tiny layer of solution around it can transform an inefficient reaction into a highly selective, scalable one. By harnessing waste nitrogen and carbon sources and driving the chemistry with electricity, the authors outline a path toward cleaner amino acid production. Their work suggests that similar control of catalyst reconstruction and local pH could unlock greener syntheses for many other nitrogen-containing molecules that underpin modern life.
Citation: Han, S., Liu, H., Timoshenko, J. et al. Sn catalyst reconstruction and microenvironment modulation for efficient amino acid electrosynthesis via C–N coupling. Nat Commun 17, 3614 (2026). https://doi.org/10.1038/s41467-026-71694-4
Keywords: electrosynthesis, green chemistry, glycine, tin catalyst, amino acids