Clear Sky Science · en
Genomic inference of sites of transmission during regional spread of blaNDM Klebsiella pneumoniae in Michigan
Why this hidden hospital threat matters
Antibiotic resistant germs can quietly move with patients as they transfer between hospitals, nursing homes, and rehab centers. When health officials spot a dangerous strain, they urgently need to know where it is spreading and which facilities are unknowingly passing it on. This study shows how reading the DNA of bacteria, together with routine patient movement records, can reveal those hidden transmission hot spots across an entire region.
Following a dangerous germ across Michigan
The researchers focused on a particularly worrisome bacterium called Klebsiella pneumoniae that had learned to resist powerful carbapenem antibiotics using a gene known as blaNDM-1. Between late 2019 and 2022, Michigan health authorities collected samples from 72 patients treated in 47 different healthcare facilities. What first looked like a problem in a single hospital grew into a regional issue as cases appeared in many more places. By comparing the full DNA sequences of the bacteria, the team found that almost all of these cases traced back to one recent introduction of a single strain that had expanded and spread, rather than many unrelated imports.
Using DNA like a contact tracing tool
To understand how the strain moved, the team did not just count how many DNA changes separated one sample from another. Instead, they used a "maximum shared variants" approach that simply asks: for each new case, which earlier samples are its closest genetic neighbors. Because this method does not depend on a hard cutoff in genetic distance, it can capture both very tight links and slightly more distant ones that still likely share a recent source. The scientists then laid patients' prior healthcare stays on top of the DNA family tree, asking where genetically close patients had overlapped in time and place.

Pinpointing likely sites of spread
For each patient, the team simulated a real-time investigation, pretending they only knew about cases reported up to that point. They looked for overlaps in facility exposure between a new case and its closest genetic matches. When more than half of those links pointed to the same facility, that site was tagged as the most likely place where the patient acquired the resistant strain. Using this rule, they could assign a single likely source facility for 66 of 70 analyzable patients. About half of cases were best explained by spread within the facility where they were diagnosed, while the rest appeared to reflect infections picked up in other institutions and carried in by patients.
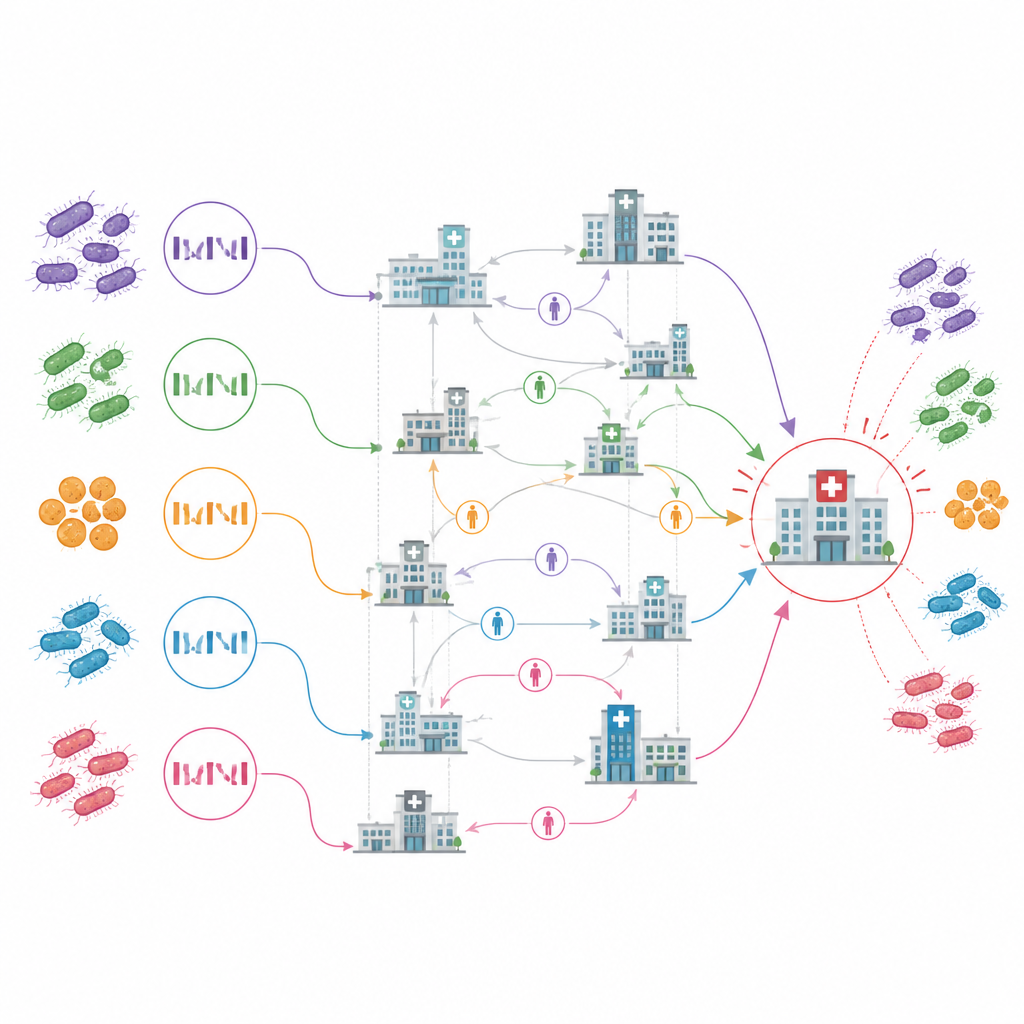
Revealing key hubs and hidden reservoirs
The resulting map of transmission between facilities highlighted one acute care hospital, labeled ACH10 in the study, as a central hub. It was the earliest site with cases, showed ongoing spread within its walls, and appeared as the source for more than a quarter of cross-facility transmission events. Most other links between facilities seemed to be one-time introductions with only limited onward spread. Strikingly, the analysis also implicated several facilities as likely sources of infection before they had ever reported a case themselves, and even flagged some facilities that never reported any cases at all but sat on common pathways of exposure for closely related infections.
What this means for infection control
By combining bacterial DNA with routine information on where patients have been, this study shows that health agencies can reconstruct how a resistant strain moves through a whole network of hospitals and care centers using samples they already collect. The approach helps identify which facilities are driving regional spread, which ones are mainly receiving imported cases, and where hidden reservoirs may be simmering before anyone notices. With this kind of real-time, genome-informed surveillance, public health teams could more quickly target cleaning, screening, and isolation efforts to the right places, slowing the march of dangerous antibiotic resistant bacteria across a region.
Citation: Wan, T., McNamara, S., Brennan, B. et al. Genomic inference of sites of transmission during regional spread of blaNDM Klebsiella pneumoniae in Michigan. Nat Commun 17, 4154 (2026). https://doi.org/10.1038/s41467-026-70839-9
Keywords: antibiotic resistance, hospital infections, genomic surveillance, Klebsiella pneumoniae, patient transfers