Clear Sky Science · en
Dynamics of phage-host interactions in Bacteroides fragilis resolved by single-cell transcriptomics
Why tiny gut battles matter
Our intestines are home to countless bacteria and the viruses that hunt them. This study zooms in on one such duel between a common gut bacterium, Bacteroides fragilis, and a virus that infects it. By watching tens of thousands of single bacterial cells at once, the researchers reveal why some bacteria fall victim while others stay safe and allow the population to bounce back. These hidden survival tricks shape how our gut ecosystem responds to viral attacks and may help guide future phage therapies.
Looking at single cells instead of cloudy broth
Most past work on bacteria and their viruses has been done in bulk test tubes, where the behavior of millions of cells is averaged together. That approach hides the fact that not every cell behaves the same way. Here, the team used a method called microbial single cell RNA sequencing to read which genes were active in about 50,000 individual B. fragilis cells after exposure to a newly isolated virus from wastewater. This allowed them to separate infected from uninfected cells in the very same culture and to see how both host and virus activity changed over the course of infection.
Reconstructing an invisible infection timeline
Even though the researchers took only one snapshot in time, infected cells were caught at many different stages of disease. By comparing their gene activity patterns, the team ordered these cells along a virtual timeline that tracks the progress of infection. Early on, the virus drove the bacterium to boost its basic gene reading and protein building machinery. In the middle phase, the host switched on genes that supply DNA building blocks, which the virus likely hijacks to copy its genome. Late in the process, the host's own messages mostly vanished while viral genes for building new virus particles and breaking open the cell surged, signaling an imminent burst of virus offspring.
Hidden subtypes that shrug off attack
Not all cells in the virus-exposed culture were infected. A sizable fraction remained untouched, and single cell data showed that this was not just luck. The bacterium naturally shuffles certain DNA switches, creating subgroups that display different sugar coats, called capsules, and different surface fibers, or fimbriae. The team found that cells wearing specific capsule types, especially ones named PSB and PSG, were far less likely to be infected than cells with other coats. Another surface system, controlled by a DNA switch called Tsr16, became especially protective when highly active, further lowering infection odds.
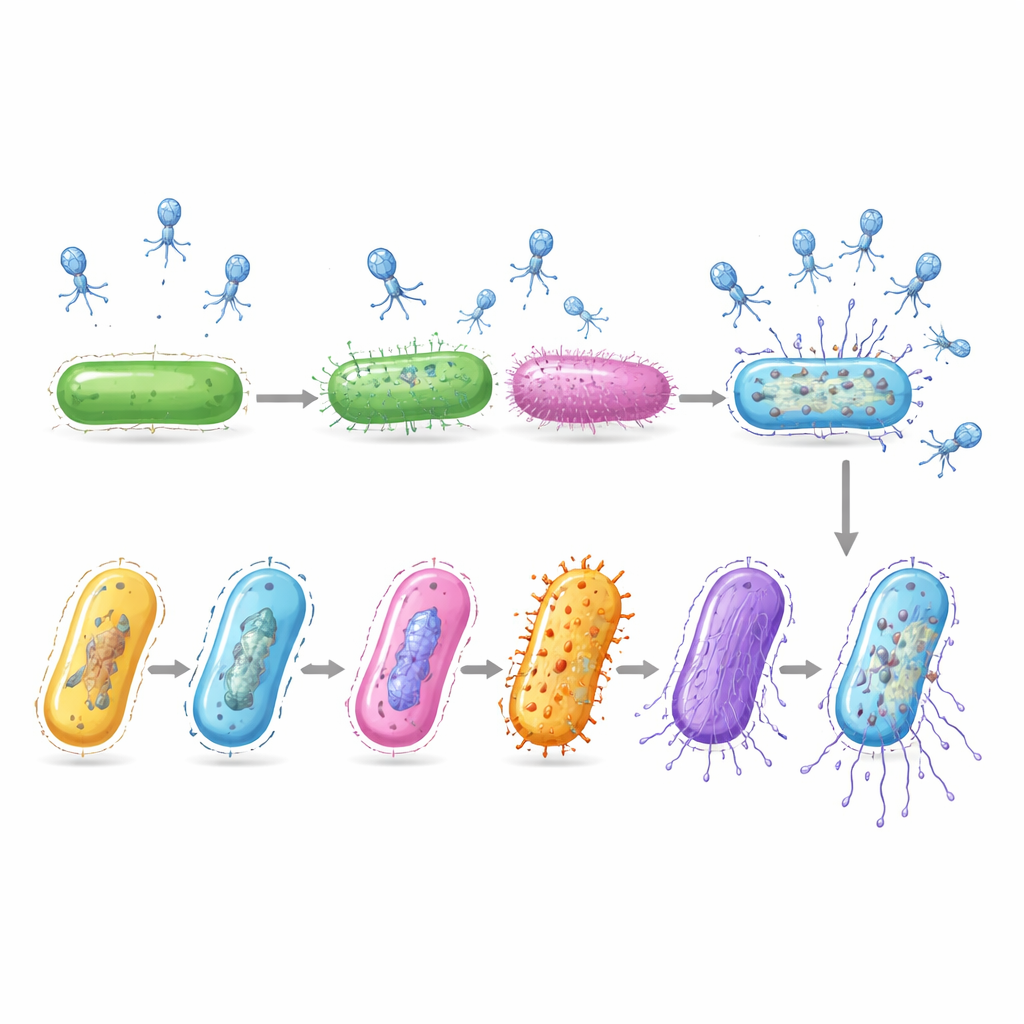
Layers of defense working together
The study also looked at known antiviral tools inside the bacterium, such as systems that cut foreign DNA or sacrifice an infected cell. On their own, these defenses did not fully explain which cells survived. But when certain internal defenses were active in the same cells that carried protective capsules, the chance of infection dropped to nearly zero. In separate growth experiments using strains locked into producing only one capsule type, bacteria with the PSG coat resisted the virus while strains with more vulnerable coats were wiped out. Follow up DNA sequencing of surviving bacteria showed that resistance mainly came from selecting these preexisting protective types, rather than from new mutations.
What this means for our gut and for phage use
To a lay reader, the main message is that gut bacteria do not all look or behave the same, even when they share the same genes. By constantly mixing their surface features and other defenses, they create small subgroups that can survive sudden viral attacks and later repopulate the community. This built in bet hedging helps Bacteroides fragilis persist in the turbulent environment of the human intestine and suggests that successful medical use of phages will need to account for these hidden, hard to kill subpopulations.
Citation: Gupta, A., Morella, N., Sutormin, D. et al. Dynamics of phage-host interactions in Bacteroides fragilis resolved by single-cell transcriptomics. Nat Commun 17, 4035 (2026). https://doi.org/10.1038/s41467-026-70381-8
Keywords: bacteriophages, gut microbiome, Bacteroides fragilis, single cell sequencing, bacterial defense