Clear Sky Science · en
Dual-stiffness nanoparticles for compartment-specific drug delivery in stroke
Targeting Stroke on Two Fronts
Stroke is often described as a brain attack, but this research shows that the damage does not stop at the brain. When a major artery in the brain is blocked, a storm of inflammation erupts both in the blood and inside the brain itself. This study introduces a clever nanomedicine strategy that sends one type of tiny particle to calm the immune system in the bloodstream while another type slips into the brain to nurse damaged nerve cells back toward health—all from a single injection.
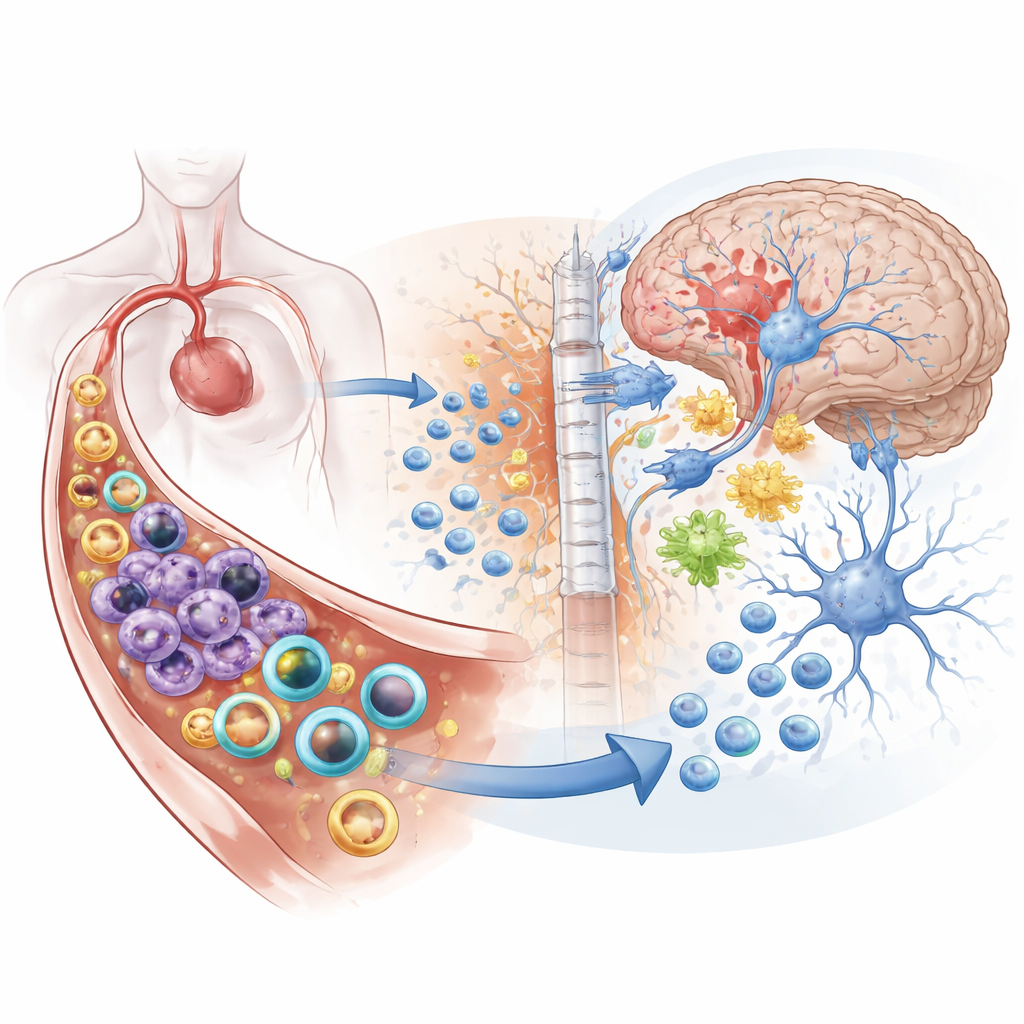
Why Stroke Is More Than a Local Injury
Ischemic stroke, caused by a clot blocking blood flow to the brain, is the most common form of stroke and a leading cause of death and disability worldwide. When brain tissue loses oxygen, nerve cells die, toxic molecules are released, and the blood–brain barrier—the protective wall between blood and brain—starts to leak. Distress signals spill into the bloodstream, summoning armies of immune cells such as neutrophils and monocytes. These cells rush toward the brain, squeeze through blood vessels, and worsen swelling and tissue damage. Many current treatments focus either on quieting the immune system everywhere or trying to protect neurons directly, but rarely both in a coordinated way.
Designing Two Kinds of Healing Particles
The research team reasoned that the physical “feel” of a nanoparticle—how stiff or soft it is—might decide which cells it ends up in. They built two nearly identical particles from approved materials: soft, squishy liposomes and stiffer particles made by placing a solid polymer core inside a similar lipid shell. Both types had similar size, charge, and outer chemistry, but drastically different stiffness. In cell experiments, stiff particles were greedily swallowed by immune cells, while neurons showed a clear preference for the softer ones. When both were given together, immune cells focused even more on the stiff particles and largely ignored the soft ones, allowing the latter to pass through in greater numbers.
How Mechanics and Blood Proteins Steer the Particles
Computer simulations and biochemical tests helped explain this split behavior. Stiff particles required less energy for cells to wrap their membranes around them, making them easier to engulf. Soft particles stuck well to the membrane but were harder to fully pull inside, especially for stiff, highly active immune cells. At the same time, the blood coated the particles with a layer of proteins known as a “corona.” Stiff particles attracted more complement proteins—molecular tags that flag objects for immune attack—while soft particles picked up fewer of these tags. When both types were present, stiff particles hogged the complement proteins, leaving soft ones relatively untagged. This combination of easier engulfment and heavier tagging pulled stiff particles into immune cells and shielded soft particles from clearance, helping them stay in circulation and reach the brain.
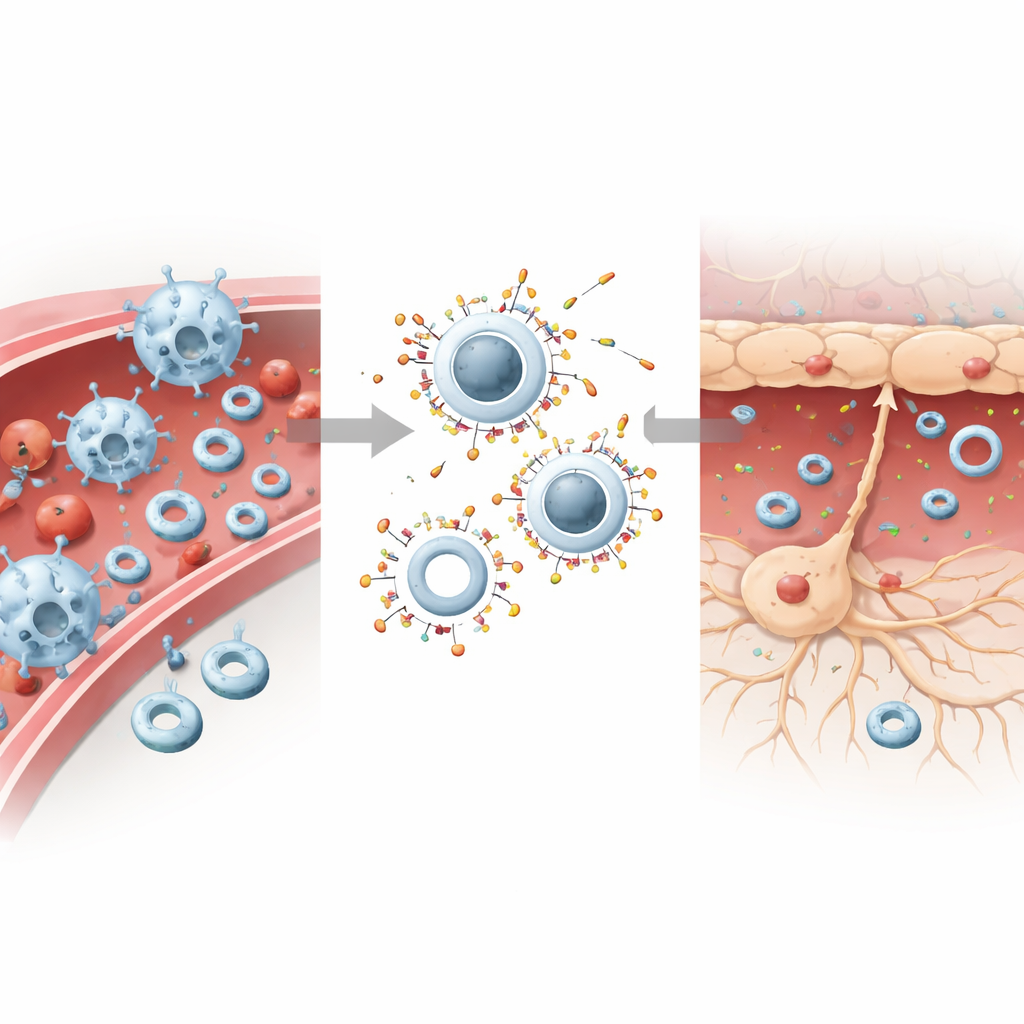
Splitting the Job: Calming Blood, Healing Brain
To turn this behavior into therapy, the scientists loaded the stiff particles with piceatannol, a drug that dampens signals driving immune cell adhesion and invasion, and loaded the soft particles with DL-3-n-butylphthalide, a neuroprotective compound used to counter oxidative stress and cell death. In mouse models of stroke, the stiff drug-loaded particles accumulated in the liver and immune cell–rich areas, cutting levels of key adhesion molecules and sharply reducing the number of immune cells entering the injured brain. The soft, drug-loaded particles, now less likely to be swallowed by immune cells, crossed the damaged blood–brain barrier more efficiently and released their cargo to stressed neurons, improving cell survival and easing local inflammation.
From Mice to a New Kind of Precision Medicine
When tested over 28 days in mice with severe stroke, this dual-stiffness combination greatly reduced brain swelling and infarct size, preserved the integrity of the blood–brain barrier, and lowered markers of inflammation and cell death. Most strikingly, survival rose from about one in nine mice to nearly eight in nine, and neurological function recovered much better than with any single-particle or free-drug treatment. For non-specialists, the key message is that matching drug carriers to the right body compartment by tuning their softness or stiffness can orchestrate a two-pronged attack: one in the blood to stop harmful immune overreaction, and one in the brain to foster repair. This “mechanically guided” delivery concept could extend beyond stroke to other diseases where the immune system and the brain—or other organs—are locked in a damaging dialogue.
Citation: Liu, H., Zheng, J., Li, Y. et al. Dual-stiffness nanoparticles for compartment-specific drug delivery in stroke. Nat Commun 17, 3837 (2026). https://doi.org/10.1038/s41467-026-70340-3
Keywords: ischemic stroke, nanoparticles, drug delivery, neuroinflammation, precision medicine