Clear Sky Science · en
Alzheimer’s Aβ catalyzes Tau phase separation and aggregation via early nanocluster solubilization
Why this matters for understanding Alzheimer’s
Alzheimer’s disease is famous for two kinds of harmful protein build-ups in the brain: sticky amyloid-beta (Aβ) plaques outside nerve cells and tangled strands of a protein called Tau inside them. Doctors know both appear together and worsen memory loss, but how one drives the other has been a long‑standing mystery. This study uncovers a hidden, early stage of their partnership, showing that a common form of Aβ can act like a molecular catalyst that nudges Tau from a fluid, reversible state into rigid, toxic clumps. Understanding this shift could open new routes to stop damage long before full-blown plaques and tangles form.
Two troublemaking proteins, one connected story
Aβ and Tau have usually been studied as separate villains. Aβ forms short fragments that collect into plaques, while Tau, a helper of internal cell scaffolding, can twist into fibers that clog neurons. Plaque burden alone, however, does not closely track how badly a person’s thinking declines, whereas Tau tangles do. That pattern has fueled the idea that Aβ might set the stage and Tau delivers much of the damage. Yet what happens at the earliest steps—when both proteins are still swirling in solution or condensed into tiny droplets—has been unclear. The authors focus on Aβ40, the most abundant Aβ type in brain fluid, and ask how it alters Tau’s tendency to condense, solidify, and ultimately poison nerve cells.
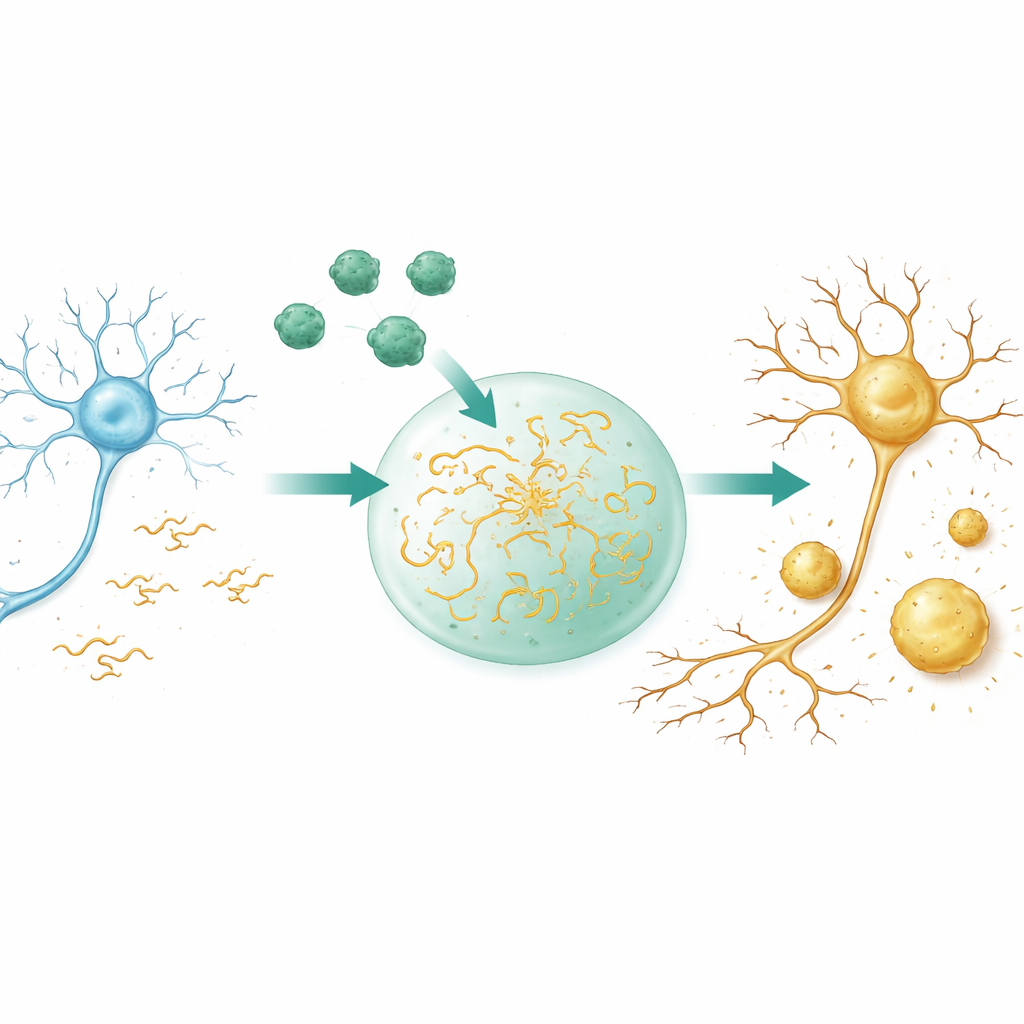
From fleeting droplets to hardened clumps
Many proteins in cells can separate into droplet-like condensates, somewhat like oil in water, without the help of membranes. These droplets concentrate molecules, speed reactions, and can later harden into more permanent deposits. Using microscopes and light-based measurements, the researchers show that Aβ40 alone does not form such liquid droplets under their conditions; instead, it aggregates directly into solid-like particles. Tau, in contrast, readily forms liquid droplets whose molecules move rapidly and mix. When the two proteins are combined in a crowded, cell‑like environment, Aβ40 is pulled into Tau droplets and dramatically boosts how easily Tau condenses. Droplets form at lower Tau levels, appear faster, grow larger, and, crucially, lose their liquid nature more quickly, becoming gel‑like and then rigid.
How Aβ reshapes Tau at the nanoscale
Before any visible droplets arise, Tau does not float only as single molecules; it also forms tiny, dynamic clusters that are hard to detect. Using sensitive mass measurements and nuclear magnetic resonance, the team finds that Aβ40 initially breaks up these Tau nanoclusters into smaller, more soluble species. At first glance that sounds protective, but computer simulations and further experiments reveal a different picture. Once Aβ40 and Tau encounter each other, they build mixed condensates where Tau molecules pack more tightly and move less, while Aβ40 remains relatively mobile inside. Detailed modeling shows that charged and water‑repelling regions on both proteins attract each other and simultaneously strengthen Tau–Tau contacts. The net effect is to lower the barrier for Tau to condense and for those condensates to harden.

From altered droplets to greater cell damage
The authors then ask what these changing states mean for living cells. They track amyloid‑like structures with a dye that lights up when proteins adopt the stacked shapes typical of fibrils. Tau droplets alone show little such signal, but when Aβ40 is present, bright, fibril‑like material quickly appears within the condensates, and the usual slow “lag” before fibrils grow is nearly erased. In nerve‑like cells grown in dishes, mixtures of Tau and Aβ40 that have undergone phase separation are markedly more toxic than Tau alone. Without crowding, Aβ40 can sometimes make Tau aggregates more soluble and slightly less harmful, suggesting that the balance between protective and destructive interactions depends strongly on the surrounding conditions—much as it would in different regions or stages of the brain.
What this means for future treatments
Taken together, the work supports a stepwise view of Alzheimer’s chemistry: Aβ40 first disperses early Tau clusters, then helps gather Tau and itself into dense droplets, and finally speeds the transformation of those droplets into rigid fibrils and toxic assemblies. In this sense, Aβ40 behaves less like a simple trigger and more like a catalyst that channels Tau through a particular path toward disease‑linked structures. For non‑specialists, the key message is that the damage in Alzheimer’s may be decided long before visible plaques and tangles appear, in a hidden world of nanoscale droplets and clusters. Therapies that interrupt Aβ–Tau contacts, keep Tau condensates in a fluid, reversible state, or prevent their hardening into solid aggregates could offer new ways to slow or halt neurodegeneration.
Citation: Sun, X., Tang, Y., Wang, X. et al. Alzheimer’s Aβ catalyzes Tau phase separation and aggregation via early nanocluster solubilization. Nat Commun 17, 3755 (2026). https://doi.org/10.1038/s41467-026-70083-1
Keywords: Alzheimer’s disease, amyloid-beta, Tau protein, phase separation, protein aggregation