Clear Sky Science · en
USP7 deubiquitinase stabilizes FAN1 to support DNA crosslink repair and suppress CAG repeat expansion
Protecting Our DNA’s Instruction Manual
Every cell in our body relies on DNA as its master instruction manual. But this delicate code is constantly under attack from chemicals, radiation, and even routine cell processes. When damage goes unrepaired, it can lead to cancer or brain disorders such as Huntington’s disease. This study uncovers how two cellular “handymen,” proteins called USP7 and FAN1, team up to guard DNA against a particularly dangerous kind of damage and to slow the genetic changes that drive Huntington’s disease.
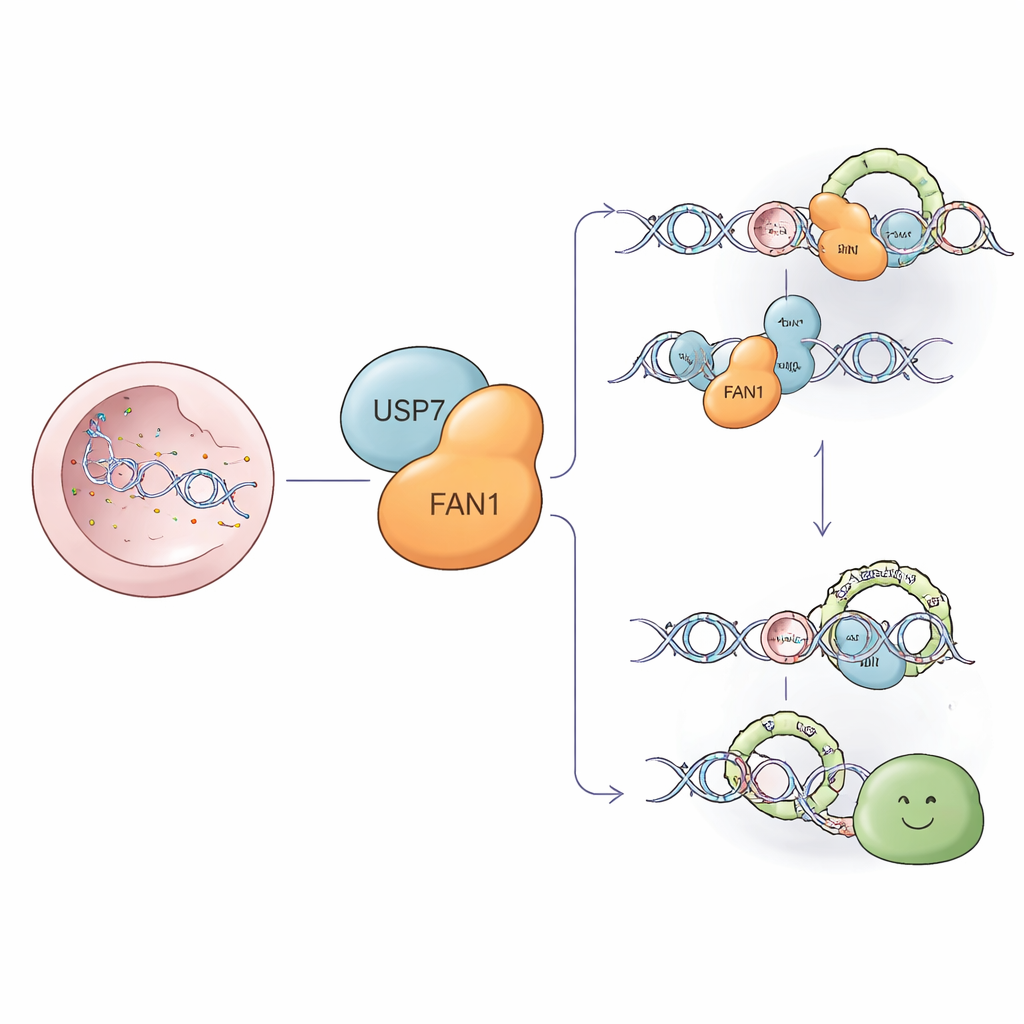
A Molecular Cleanup Crew for Stubborn DNA Damage
Some of the most harmful injuries to DNA are interstrand crosslinks—molecular “staples” that glue the two strands of the DNA double helix together. These staples block the machinery that copies DNA, threatening cell survival. FAN1 is a specialized cutting enzyme that can slice around these staples and help remove them so that repair can proceed. FAN1 also trims troublesome extra loops that form in stretches of repeated DNA letters, including CAG repeats in the gene linked to Huntington’s disease. Keeping FAN1 at the right level and in the right place is therefore crucial for both cancer prevention and healthy brain function.
USP7: The Protein Bodyguard
Proteins in cells are constantly being tagged for destruction by small molecules called ubiquitin. This tagging is like putting a “trash” sticker on a protein so the cellular recycling machinery will break it down. USP7 is a deubiquitinase—an enzyme that removes these tags and rescues selected proteins from the shredder. The researchers used mass spectrometry, a technique to catalog protein partners, and discovered that USP7 physically binds FAN1, particularly at FAN1’s floppy N‑terminal region. By latching on through two distinct surfaces, USP7 can repeatedly recognize FAN1 and pluck off the destruction tags.
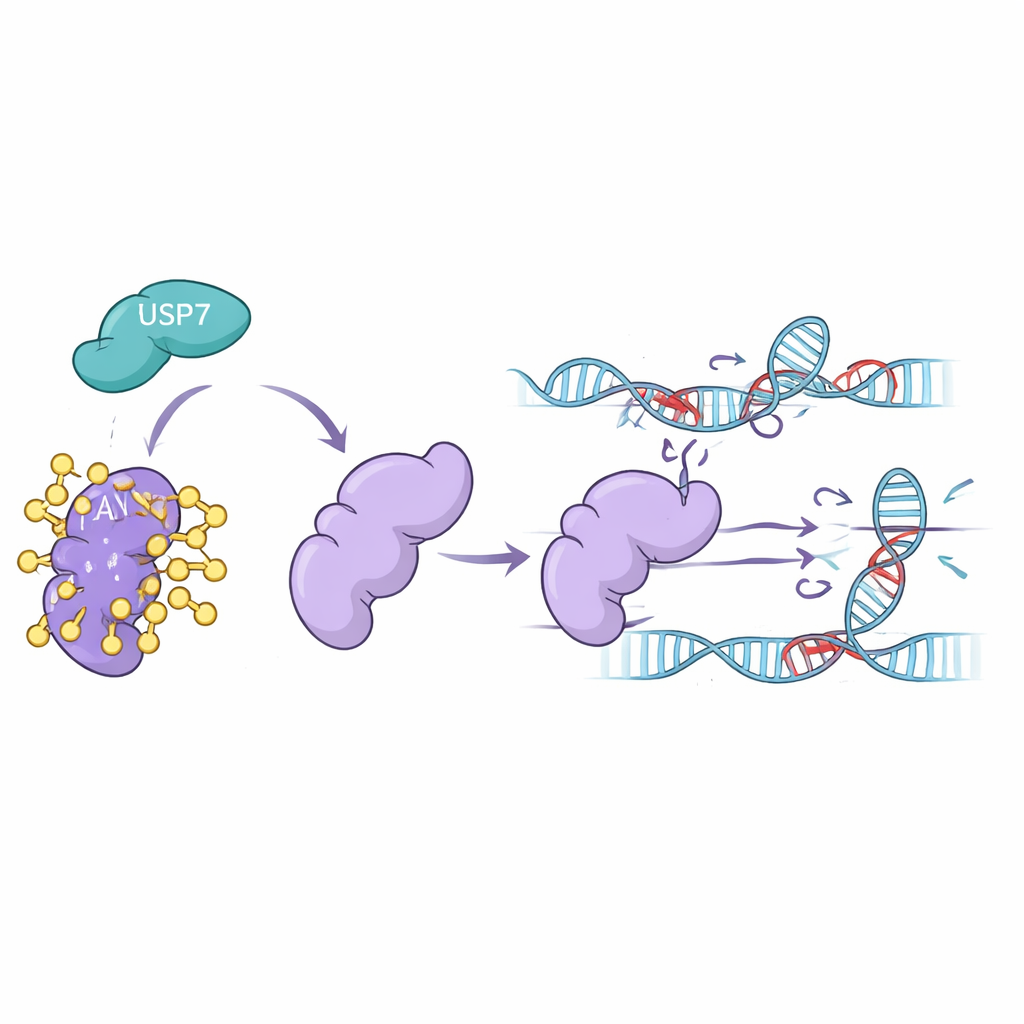
How USP7 Keeps FAN1 on the Job
Laboratory experiments in human cell lines showed that when USP7 levels were reduced—either by silencing its gene or by treating cells with USP7-blocking drugs—FAN1 levels dropped sharply. FAN1 became heavily decorated with ubiquitin and was sent to the proteasome, the cell’s protein recycling center. Blocking the proteasome rescued FAN1, confirming that USP7 normally protects FAN1 from this fate. The team further showed that only the active form of USP7 could strip off the specific ubiquitin chain type that flags FAN1 for destruction. Without USP7, FAN1 disappeared more quickly and could no longer accumulate effectively at damaged DNA where crosslinks needed to be removed.
Consequences for DNA Repair and Huntington’s Disease
When cells were exposed to a crosslinking chemotherapy drug, those lacking USP7 showed fewer FAN1 repair foci on DNA, more cell death, and greater sensitivity to damage. Under moderate stress, removing USP7 or removing FAN1 caused similar fragility, suggesting FAN1 is a main target of USP7 in this repair pathway. The authors then turned to Huntington’s disease, where expansion of CAG repeats in the huntingtin gene worsens the disorder over time. In a human retinal cell model engineered to carry a long repeat, loss of FAN1 sped up repeat growth, whereas disabling a mismatch repair factor slowed it. Strikingly, knocking down USP7 accelerated CAG repeat expansion almost as much as losing FAN1, indicating that USP7 indirectly restrains repeat growth by keeping FAN1 stable.
Why This Discovery Matters
This work reveals USP7 as a key guardian of FAN1, tying together two major threats to genome health: hard-to-fix DNA crosslinks and runaway expansion of CAG repeats in Huntington’s disease. To a lay reader, the take-home message is that USP7 acts like a bodyguard, shielding FAN1 from being prematurely destroyed so it can keep DNA repair on track and slow the genetic drift that drives neurodegeneration. The findings suggest a delicate balance for future therapies: blocking USP7 might make cancer cells more vulnerable to certain chemotherapies, while boosting the USP7–FAN1 partnership could help stabilize harmful DNA repeats in the brain.
Citation: Collotta, G., Gatti, M., Ungureanu, IM. et al. USP7 deubiquitinase stabilizes FAN1 to support DNA crosslink repair and suppress CAG repeat expansion. Nat Commun 17, 3551 (2026). https://doi.org/10.1038/s41467-026-70051-9
Keywords: DNA repair, genome stability, Huntington’s disease, protein degradation, trinucleotide repeats