Clear Sky Science · en
Genome-wide analysis of cardiac ventricular phenotypes reveals novel loci and therapeutic targets for heart failure
Why the Heart’s Hidden Code Matters
Heart failure is one of the leading causes of death and disability, yet by the time symptoms appear, much of the damage to the heart has already been done. Doctors can now take detailed scans of the heart’s main pumping chambers, the left and right ventricles, to see how well they are working. But until recently, we knew far less about the genetic code that shapes these structures and how it might point to new treatments. This study dives into the DNA of more than fifty‑six thousand people to uncover how thousands of small genetic differences influence heart size, strength, and ultimately the risk of heart failure.
Looking Inside Thousands of Hearts
The researchers used cardiac magnetic resonance imaging, a highly precise scanning method, to measure twenty aspects of how the ventricles are built and how they pump blood. These included classic measures such as how much blood the left and right sides of the heart hold and eject with each beat, as well as newer, more holistic measures of pump performance that combine heart size and muscle mass. All scans came from participants in the UK Biobank, a large health study, and advanced computer algorithms were used to trace the heart chambers automatically and consistently across more than 56,000 people.
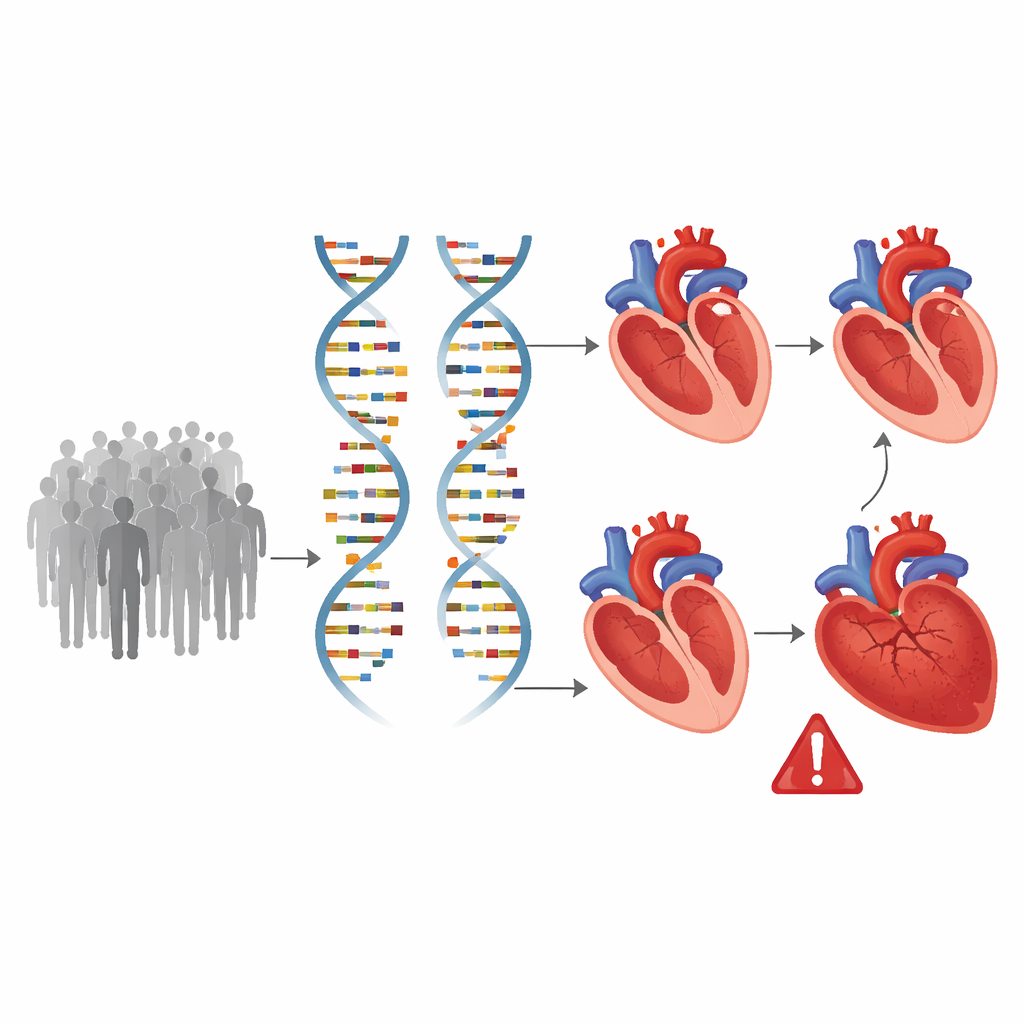
Finding Heart‑Shaping Genes Across the Genome
Armed with these detailed heart measurements, the team scanned the entire genome, testing about 8.9 million common genetic variants to see which ones tracked with differences in ventricular structure and function. They discovered 200 locations in the genome linked to at least one of the heart traits, 58 of which had not previously been connected to cardiac imaging. Many of these genetic sites influenced more than one measurement, suggesting shared biological pathways that affect both heart size and pumping power. Calculations showed that inherited factors explain roughly one‑fifth to one‑third of the variation in these traits, confirming that our DNA plays a substantial role in how our ventricles develop and remodel over time.
From DNA Signals to Disease Risk
The scientists next asked how these heart‑shaping variants relate to real‑world disease. They compared their findings with previous genetic studies of heart failure and other cardiovascular conditions and found 23 genomic regions that seem to influence both ventricular traits and heart failure itself. Some genes in these regions are already known players in inherited heart muscle disease, while others, such as those involved in responding to mechanical stress or low oxygen, highlight additional biological routes to damage. By building polygenic risk scores—combined measures of many small DNA effects—the team showed that people whose genetics predict more enlarged or thickened ventricles also have higher rates of heart failure, high blood pressure, and rhythm problems. In contrast, genetic profiles favoring more efficient pumping, captured by newer measures like the left ventricular global function index and myocardial contraction fraction, were linked to lower risk of these diseases.
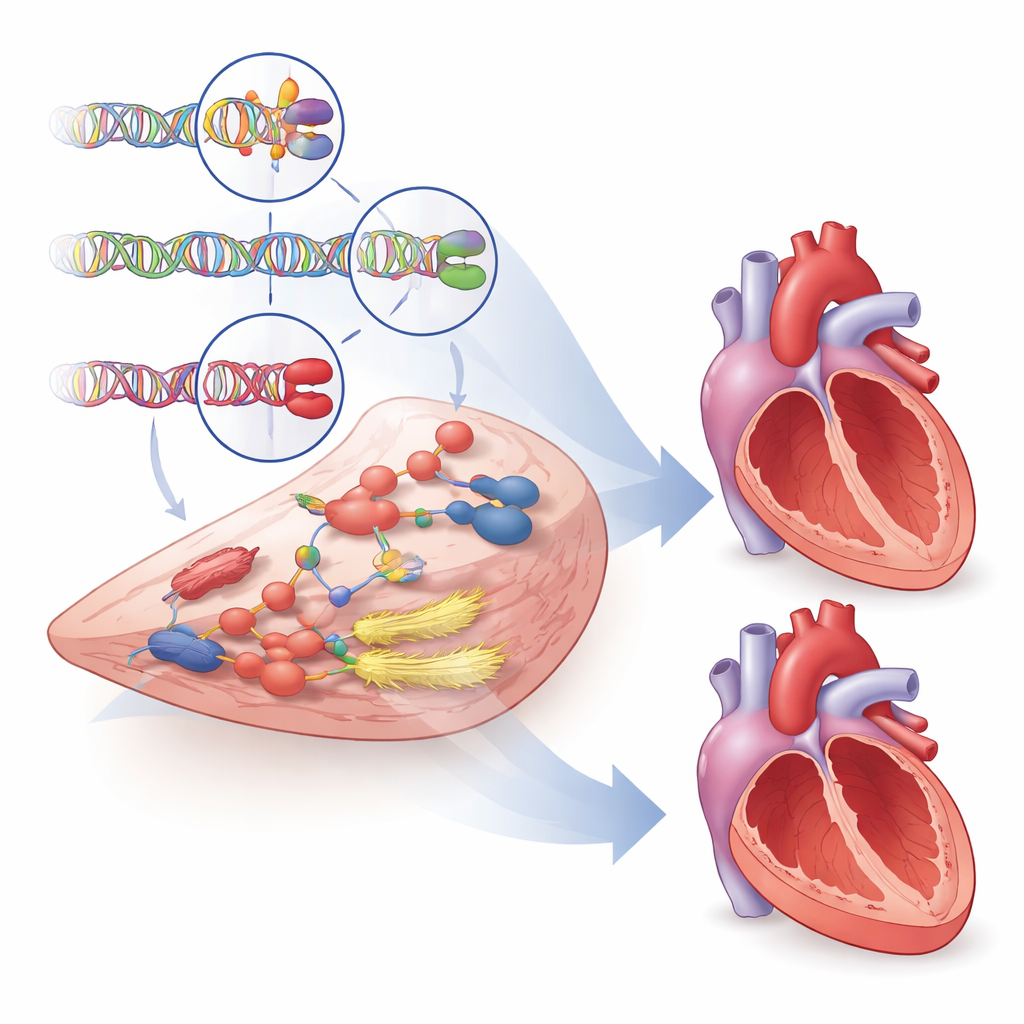
Hints for New and Better Treatments
Beyond explaining why some hearts are more vulnerable than others, the study searched for genes that might be turned into drug targets. Using multiple databases, animal studies, and patterns of gene activity, the authors systematically scored more than a thousand nearby genes and highlighted nearly 500 with evidence of involvement in ventricular structure and function. Dozens of these genes interact with existing cardiovascular medicines, including drugs that affect heart muscle contraction and blood vessel tone, pointing to opportunities for repurposing or refining therapies. One striking example is a gene called PDE3A, whose variants were linked to signs of harmful remodeling. Existing drugs block both PDE3A and its close relative PDE3B, but the genetic data suggest that designing treatments that more selectively target PDE3A could improve outcomes while avoiding side effects seen with current non‑selective drugs.
What This Means for Patients
In everyday terms, this research connects tiny differences in our DNA to the way the heart’s main pumping chambers are built and how well they work, and then links those patterns to who is more likely to develop heart failure. By combining cutting‑edge imaging, large‑scale genetics, and drug‑target analysis, the study not only deepens our understanding of why hearts fail but also points to concrete molecular handles that future medicines could grab onto. While more work is needed—especially in diverse populations and in laboratory models—these findings move us closer to a future where a scan and a genetic test together help predict heart failure early and guide more precise, genetically informed treatments.
Citation: Nicholls, H.L., Vargas, J.D., Sanghvi, M.M. et al. Genome-wide analysis of cardiac ventricular phenotypes reveals novel loci and therapeutic targets for heart failure. Nat Commun 17, 3293 (2026). https://doi.org/10.1038/s41467-026-69982-0
Keywords: heart failure, cardiac MRI, genetics, ventricular remodeling, drug targets