Clear Sky Science · en
Linker histones consolidate heterogenous nucleosome fiber contacts by linking together multiple nucleosomes
How DNA is Packed Inside Our Cells
Every cell in your body squeezes nearly two meters of DNA into a nucleus far smaller than a speck of dust. This extreme packing is not just a storage trick; it helps decide which genes are active or silent. This study digs into one of the key but often overlooked players in this process, the linker histones, and shows that they act less like simple clips on individual DNA beads and more like versatile connectors that tie many beads and fibers together.
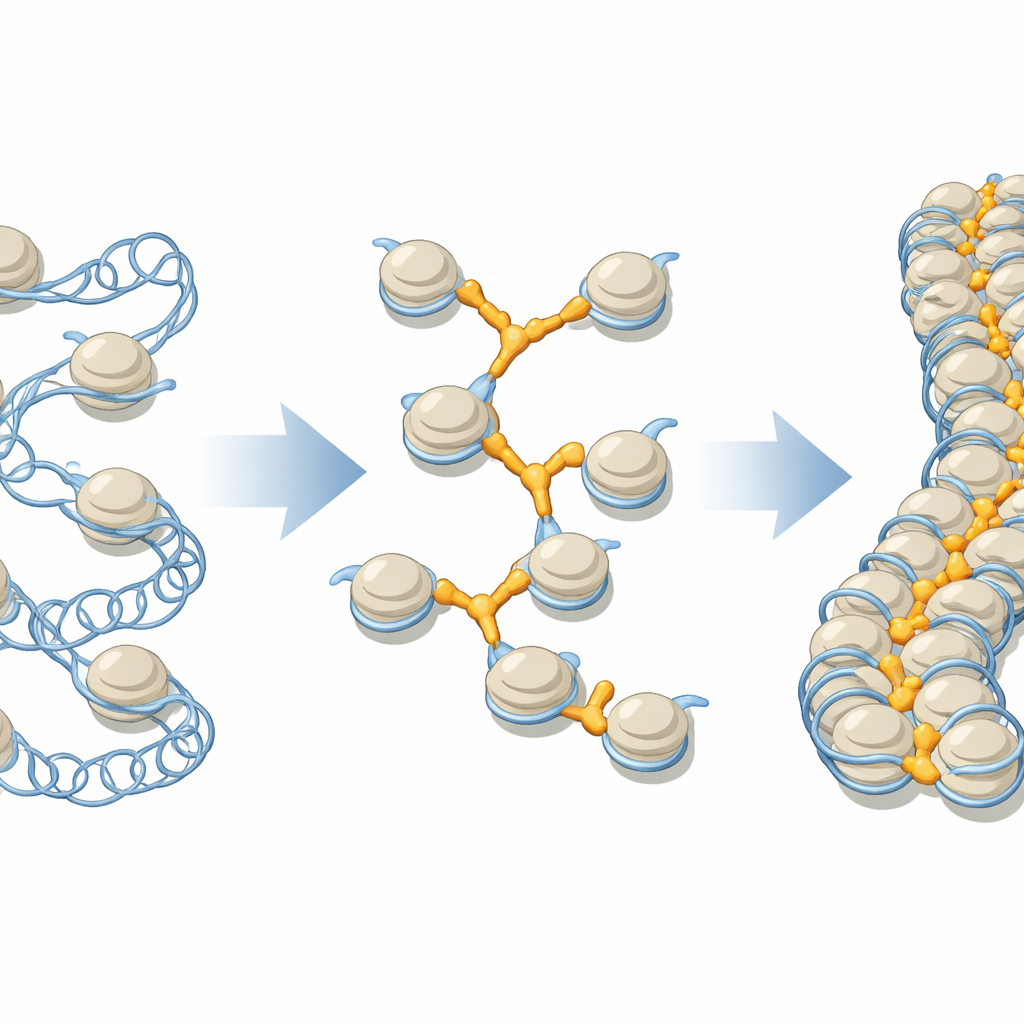
A Closer Look at the DNA Beads and Their Connectors
DNA in our cells is wrapped around protein cores to form units called nucleosomes, often pictured as beads on a string. These beads then fold into higher-order fibers that can run to more than a million units along a single chromosome. Linker histones, a family of proteins broadly labeled H1, have long been thought to sit on each bead in a standard way, helping tighten the local structure. But cells contain several different H1 variants, each with its own preferred locations and roles, and earlier structural work mostly focused on isolated beads, not the crowded, compact environment found in real chromosomes.
Engineering Model Fibers to Reveal Hidden Binding Modes
To explore how linker histones behave in a dense setting, the researchers engineered nucleosomes that naturally snap together into continuous fibers. These designer fibers could be crystallized and analyzed at near-atomic resolution using X-ray crystallography. By combining these fibers with different linker histone variants, including H1.0, H1x, H1.3, and the bird protein H5, the team observed not just the classic “on-center” binding to a single bead but multiple alternative arrangements in which one linker histone contacted several nucleosomes at once, or even multiple fibers. In effect, H1 proteins were found to occupy DNA-filled pockets formed only when beads pack together in three dimensions.
Versatile Connectors That Remodel DNA Shape
These structural snapshots revealed that the compact central portion of each linker histone recognizes recurring DNA shapes rather than just a single fixed site. Certain surface patches on the protein repeatedly engage widened grooves and bends in the double helix, whether those features reside at the center of one nucleosome or at special junctions between several nucleosomes. Some variants, such as H1x and H1.0, can even kink the DNA in specific spots, subtly shifting how it lies on the underlying protein core. Different H1 types favored different binding patterns: some primarily used a single strong interface, while others used two robust contact regions, creating a spectrum of “linking modes” that depend on both the protein variant and the local fiber geometry.
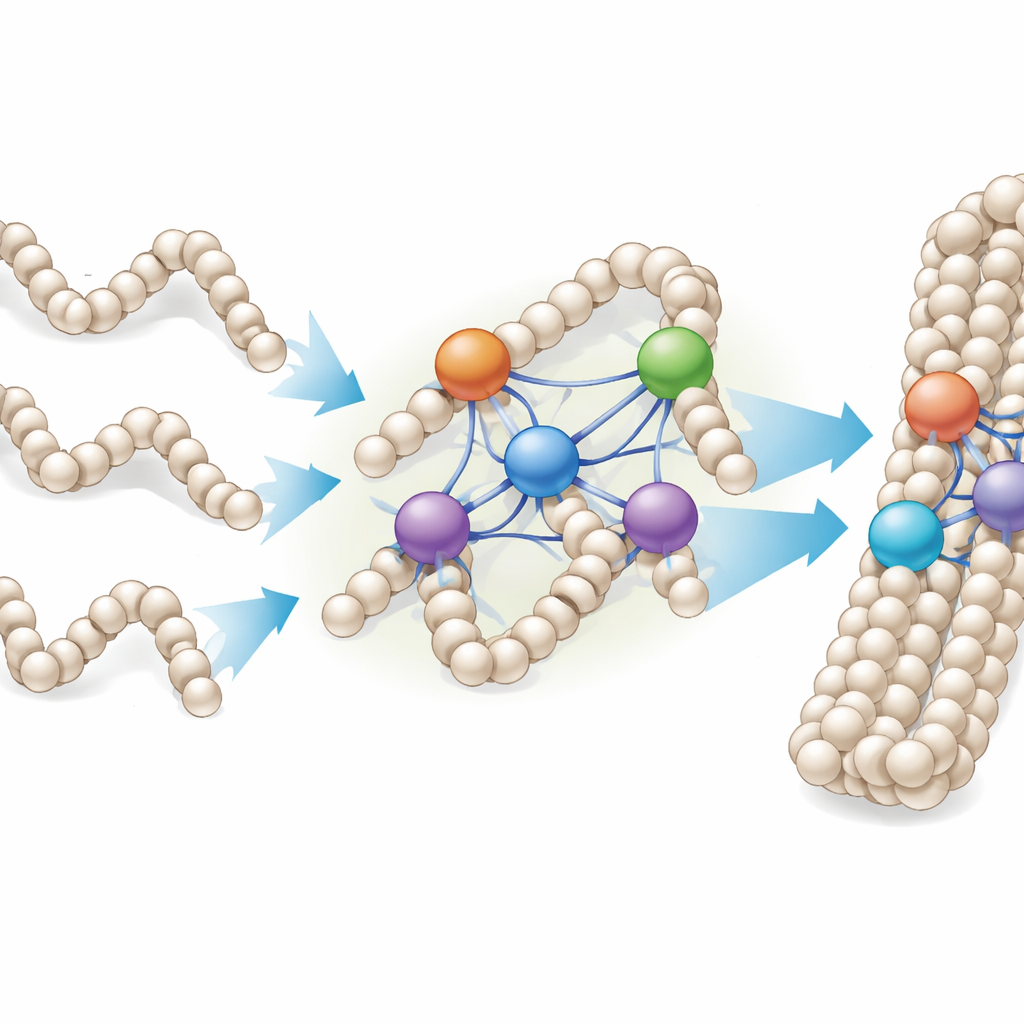
From Single Fibers to Dense Droplets of Chromatin
Because chromosomes in living cells resemble dense droplets of chromatin rather than neat, isolated fibers, the researchers also studied how linker histones bind to laboratory-made chromatin condensates—clumps of many repeated nucleosome arrays. They found that H1 proteins did not stop binding after reaching a simple one-to-one ratio with nucleosomes; instead, individual nucleosomes could be associated with two to four or more H1 molecules, depending on the variant. Under the microscope, adding more H1 caused small, spherical condensates to fuse into larger, bead-like chains, indicating that linker histones can stitch together multiple chromatin droplets and fibers into more compact assemblies.
Why This Matters for Gene Control and Disease
These results paint linker histones not as rigid clamps on single beads, but as adaptable, variant-specific connectors that exploit a range of binding niches within crowded chromatin. At low levels, H1 proteins may mostly occupy the classic single-bead positions, helping maintain basic chromatin organization. As their activity or concentration rises, additional linking modes become populated, enabling the same proteins to bridge neighboring beads, entire fibers, and even separate chromatin droplets, driving stronger compaction. This flexible toolkit helps explain how different H1 variants can tune gene activity, why some serve as tumor suppressors, and how modest changes in their abundance or modification might reorganize local chromatin architecture, tipping the balance between open, gene-rich regions and tightly packed, silent domains.
Citation: Adhireksan, Z., Sharma, D., Bao, Q. et al. Linker histones consolidate heterogenous nucleosome fiber contacts by linking together multiple nucleosomes. Nat Commun 17, 3807 (2026). https://doi.org/10.1038/s41467-026-69842-x
Keywords: chromatin compaction, linker histone H1, nucleosome fiber, genome organization, epigenetic regulation