Clear Sky Science · en
Adoptive γδ T cell therapy controls cytomegalovirus infection in preclinical transplantation models
New Hope Against a Hidden Virus
Cytomegalovirus, or CMV, is a common virus that most healthy people barely notice. For patients who have received a transplanted organ or bone marrow, however, CMV can be life-threatening and difficult to treat with drugs alone. This study explores a new kind of immune cell therapy, based on a rare T cell subtype called gamma-delta T cells, designed to help the body control CMV when standard treatments fail. The work suggests a path toward a universal, off‑the‑shelf cell therapy that could protect some of the most vulnerable transplant recipients.
Why CMV Is So Dangerous After Transplant
After a transplant, patients take strong medicines to prevent their immune systems from rejecting the new organ or donor cells. These same medicines weaken defenses against infections. CMV is particularly worrisome: it is the most frequent opportunistic infection after solid-organ transplantation and a major problem after stem cell transplants. High‑risk patients, such as those who have never encountered CMV but receive an organ from a CMV‑positive donor, often suffer recurrent or drug‑resistant infections. New antiviral drugs like maribavir help, but many patients still do not clear the virus completely or relapse soon afterward. Because long‑term control of CMV depends on a sustained immune response, researchers are turning to cell therapies to rebuild antiviral immunity rather than just suppress the virus temporarily.
A Different Kind of Immune Cell Therapy
Existing experimental cell therapies for CMV rely mainly on conventional alpha‑beta T cells, which recognize virus fragments only when displayed by specific tissue-matching molecules. This requirement limits who can receive a given cell product and makes manufacturing complex. Gamma‑delta T cells, by contrast, can sense stressed or infected cells without this strict matching. The team focused on a subset called Vδ2‑negative gamma‑delta T cells, which naturally expand during CMV infection and can attack infected cells. Using a clinical‑grade protocol known as the “Delta One T” (DOT) method, they showed that these cells can be grown to large numbers from both CMV‑positive and CMV‑negative donors, and from high‑risk kidney transplant recipients themselves, despite low lymphocyte counts and ongoing immunosuppression.
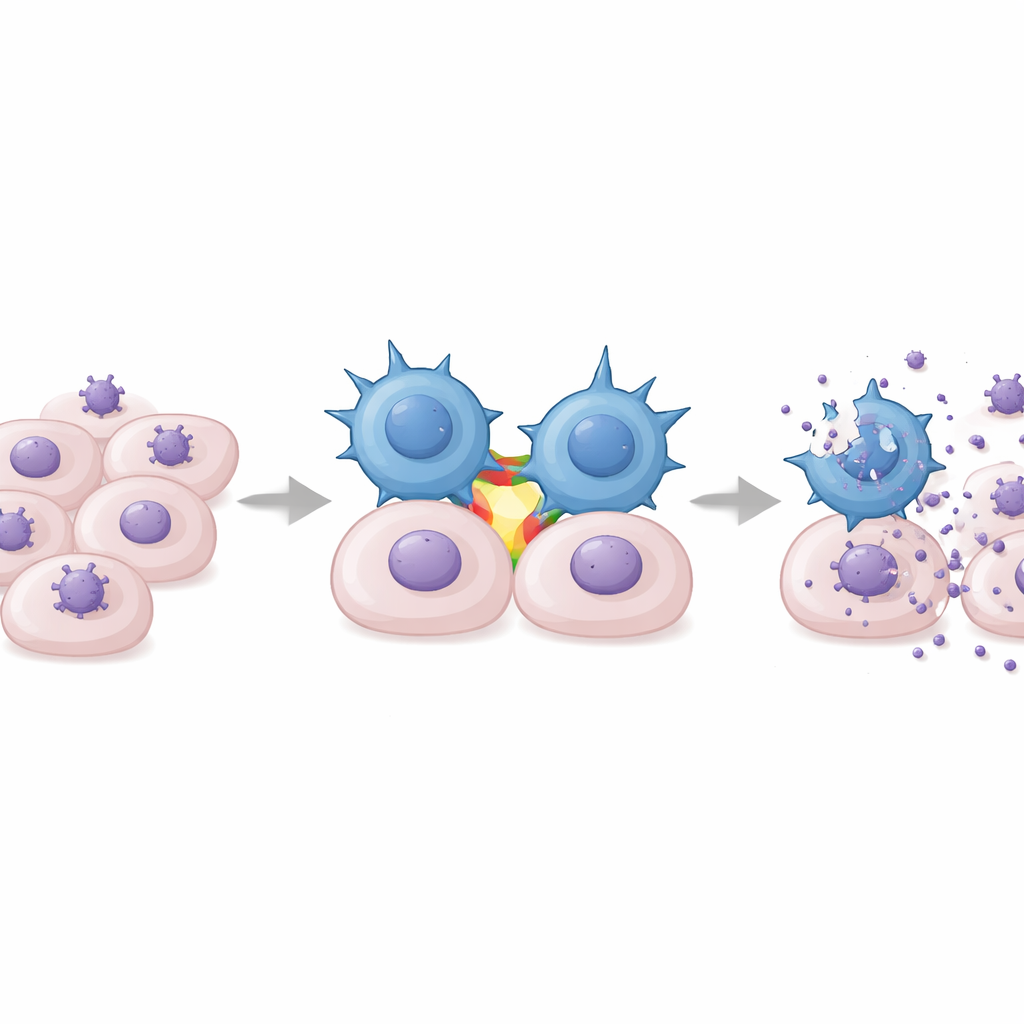
How DOT Cells Sense and Control CMV
In lab tests, DOT cells were mixed with human cells infected by clinical CMV strains. The expanded gamma‑delta T cells showed two key behaviors: they degranulated, a sign of direct killing, and they secreted large amounts of interferon‑gamma, a potent antiviral cytokine that helps halt viral replication and rally other immune cells. These responses were highly specific for CMV and did not occur with related viruses like herpes simplex or varicella‑zoster. Importantly, DOT cells slowed the spread of CMV through fibroblast cultures in a dose‑dependent way, acting more strongly as more effector cells were added. Unexpectedly, the researchers found that this recognition did not require the usual gamma‑delta T cell receptor. Instead, an adhesion molecule called LFA‑1, which binds to ICAM‑1 on infected cells, acted as a crucial co‑stimulator: blocking LFA‑1 or ICAM‑1 sharply reduced interferon‑gamma release, while artificially activating LFA‑1 triggered strong responses.
Proof of Protection in a Mouse Model
Because human CMV does not infect mice, the team built an animal model using murine CMV and mouse gamma‑delta T cells expanded with a DOT‑like protocol. These mouse cells acquired a similar activated, cytotoxic profile. When transferred into severely immunodeficient mice infected with murine CMV, the expanded gamma‑delta T cells migrated to key organs, reduced viral loads in spleen, liver, and lungs, and significantly improved survival compared with untreated controls. Protection was observed whether donor mice had previously seen CMV or not, mirroring the human finding that prior CMV exposure is not required to generate effective DOT cells.
Designed to Work Alongside Transplant Drugs
A major concern for any post‑transplant therapy is whether it can function in the presence of standard immunosuppressive drugs. The researchers grew human DOT cells with commonly used agents such as tacrolimus, cyclosporin, everolimus, and mycophenolate. While the antiproliferative drug mycophenolate essentially stopped DOT cell growth and function, tacrolimus, cyclosporin, everolimus, and typical doses of steroids allowed the cells to survive, divide, and still secrete interferon‑gamma in response to CMV‑infected targets. This suggests that, under regimens already used for patients with severe CMV disease—often based on calcineurin inhibitors with reduced or modified partners—DOT cell therapy could remain active.
What This Could Mean for Patients
To a non‑specialist, the key message is that scientists have engineered a type of immune cell that can be grown in large numbers, does not depend on strict donor–recipient matching, and can recognize and restrain CMV in ways that complement antiviral drugs. In cell cultures and mouse models, these DOT gamma‑delta T cells home to infected tissues, curb virus spread, and remain functional despite common transplant medications. The work lays the groundwork for early‑phase clinical trials in transplant recipients with recurrent, refractory, or drug‑resistant CMV, with the long‑term goal of turning a dangerous post‑transplant complication into a controllable condition using the patient’s own immune system, reinforced.
Citation: Marsères, G., Gentil, C., Tinevez, C. et al. Adoptive γδ T cell therapy controls cytomegalovirus infection in preclinical transplantation models. Nat Commun 17, 2847 (2026). https://doi.org/10.1038/s41467-026-69538-2
Keywords: cytomegalovirus, gamma-delta T cells, cell therapy, organ transplantation, immunotherapy