Clear Sky Science · en
Hepatocyte-derived LRG1 primes the liver for metastasis and impairs immunotherapy
Why this research matters for cancer patients
Cancer often returns not in the original organ but in distant sites such as the liver, and once that happens, treatments become far less effective. This study uncovers how the liver is quietly “prepared” in advance to welcome wandering cancer cells, and why tumors that spread to the liver often resist modern immunotherapy drugs. By pinpointing a single liver-made protein, LRG1, as a key switch in this process, the work suggests new ways to predict, prevent, and better treat liver metastases.
A warning signal in the blood
Doctors have long noticed that cancers such as colorectal, pancreatic, and stomach cancer frequently spread to the liver, but they lacked good tools to foresee who was at highest risk. The researchers analyzed blood samples from patients with these cancers and found that those who already had liver metastases carried much higher levels of a protein called LRG1 in their blood. Even more striking, among people with early-stage disease and no detectable spread, those who later developed liver metastases already had elevated LRG1 at diagnosis and tended to relapse sooner. In several mouse models that mimic human cancer, LRG1 in the bloodstream rose weeks before visible liver tumors appeared, marking the so‑called “pre‑metastatic” phase when the organ is being reshaped to favor future tumor growth. 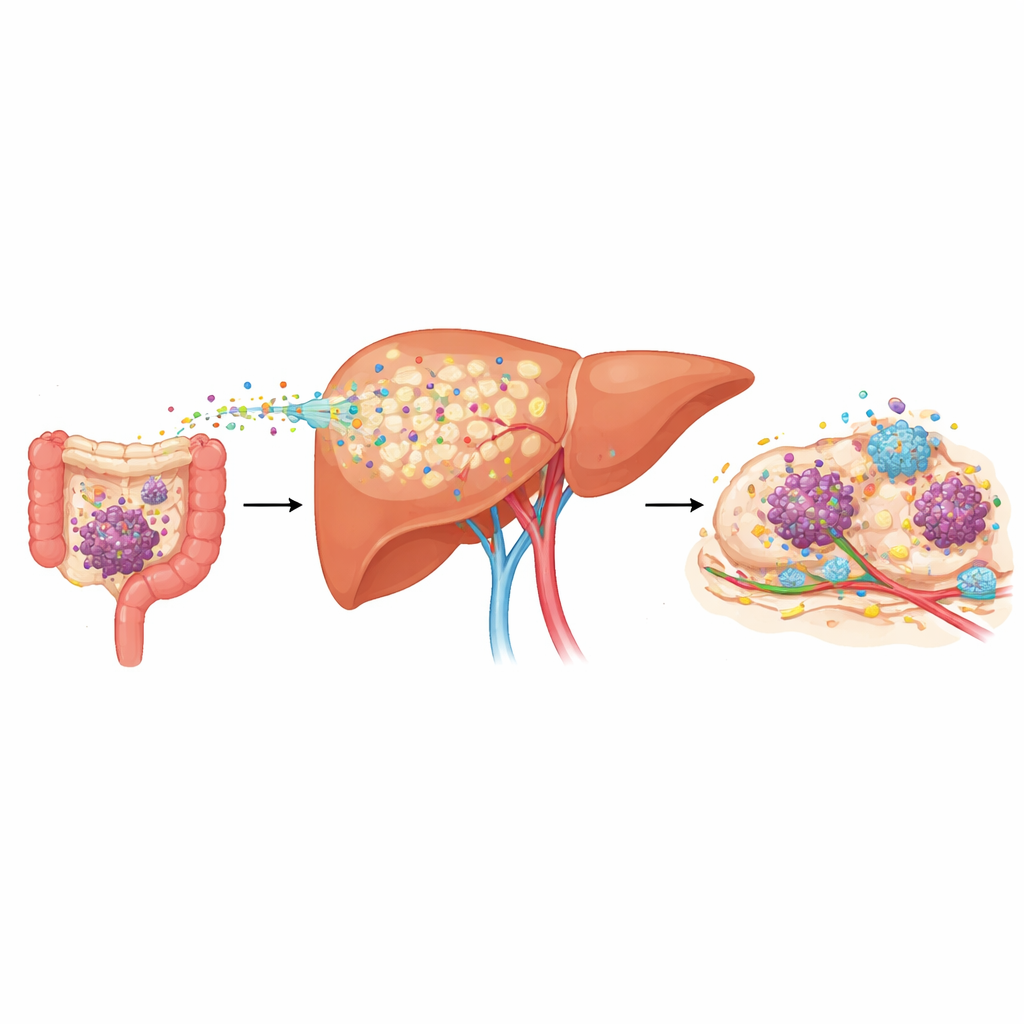
The liver is not just a passive victim
To track where this surge of LRG1 was coming from, the team examined many organs and cell types in tumor-bearing mice. The liver stood out as the dominant source, and within the liver, ordinary liver cells (hepatocytes) produced far more LRG1 than immune or blood-vessel cells. When the scientists used genetic engineering to delete the Lrg1 gene only in hepatocytes, blood levels of LRG1 barely rose during cancer progression, and the animals developed far fewer and smaller liver metastases. In fact, when liver tumors were seeded in a way that normally yields a high burden of disease, removing hepatocyte-derived LRG1 dramatically cut both the number of metastatic spots and their size, showing that this liver-made protein is a powerful enabler of spread.
Rewiring the liver’s immune landscape
The next question was how LRG1 turns a healthy liver into fertile ground for cancer. Using single-cell sequencing, the researchers mapped tens of thousands of individual liver cells from healthy and tumor-bearing mice. In livers exposed to a distant tumor, they saw an influx of certain white blood cells that dampen immunity, especially neutrophils and related myeloid cells, while helpful T cells and dendritic cells dwindled or became exhausted. When LRG1 was absent from hepatocytes, this skewing toward an immunosuppressive environment was largely reversed. The team found that LRG1 directly activates neutrophils to release sticky, web-like structures known as neutrophil extracellular traps, or NETs. These DNA webs form inside liver blood vessels, snaring circulating cancer cells and helping them anchor, survive, and grow. Breaking down NETs with an enzyme, or depleting neutrophils, sharply reduced liver metastases and largely erased the advantage conferred by LRG1, tying this mechanism directly to tumor spread.
How inflammation in tumors talks to the liver
What makes hepatocytes ramp up LRG1 in the first place? The investigators showed that it is not direct contact with tumor cells, but inflammatory signals circulating in the blood. Among many candidate molecules, the inflammatory messenger IL‑6 emerged as the key driver: adding IL‑6 to liver cells in a dish strongly induced LRG1, while blocking IL‑6 or its receptor prevented this. In mice, IL‑6 levels rose alongside LRG1 during the pre‑metastatic phase, and artificially boosting IL‑6 in the liver increased LRG1 production and accelerated metastasis—unless hepatocytes lacked LRG1. Further analysis pointed to macrophages—immune cells educated by the growing tumor—as a major source of IL‑6 in the liver, revealing a chain of communication: tumor inflammation activates macrophages, macrophages release IL‑6, IL‑6 instructs hepatocytes to secrete LRG1, and LRG1 then remodels the liver’s immune and vascular environment in favor of future cancer cells. 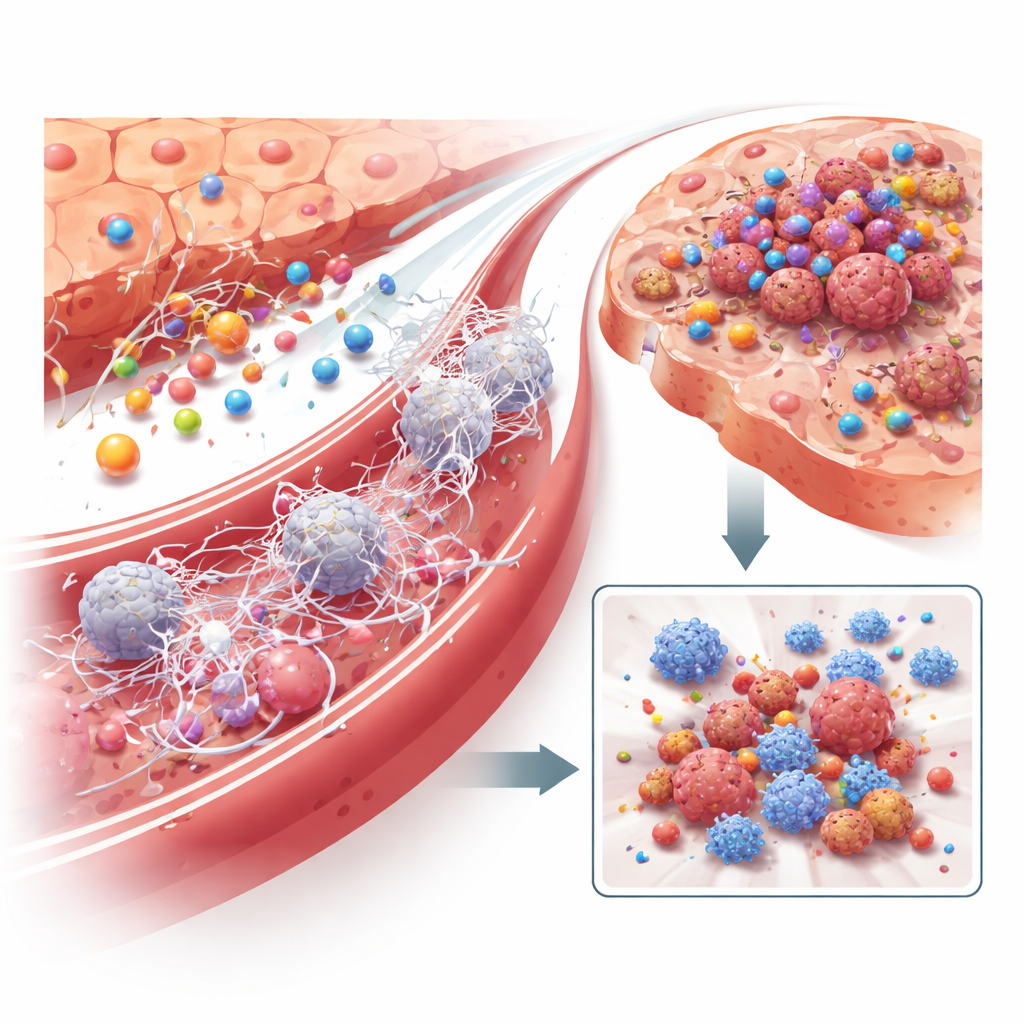
Turning a hostile niche back into friendly ground
Because LRG1 sits at this critical crossroads, the researchers tested whether blocking it could be therapeutic. In mice with experimentally induced liver metastases, treatment with an antibody against LRG1 reduced NET formation and substantially lowered the number and size of liver tumors. A gene therapy approach that selectively silenced Lrg1 in hepatocytes produced similar benefits. Importantly, liver metastases are known to blunt the effect of immune checkpoint drugs such as anti‑PD‑1 antibodies. When the scientists combined LRG1 blockade with anti‑PD‑1 therapy in a liver tumor model, tumors shrank much more than with either treatment alone, and aggressive immune cells (CD8 T cells) became more numerous and more actively toxic to cancer. This suggests that targeting LRG1 can convert an immunologically “cold” liver metastasis into a “hotter” one that responds better to immunotherapy.
What this means for future care
Overall, this work reveals that the liver is actively coached by distant tumors to become a safe harbor for cancer cells, and that hepatocyte-derived LRG1 is a key messenger in that coaching. Measuring LRG1 in blood could help identify patients at high risk of liver spread before metastases appear, while drugs that block LRG1 or its upstream IL‑6 signal could both prevent liver metastasis and make existing lesions more vulnerable to immune attack. Although these findings are so far limited to mice and patient samples rather than clinical trials, they highlight a promising new strategy: instead of waiting to treat metastases after they form, we might preemptively disarm the “welcome mat” the liver lays out for cancer.
Citation: Long, G., Cheng, B., Jiang, Y. et al. Hepatocyte-derived LRG1 primes the liver for metastasis and impairs immunotherapy. Cell Mol Immunol 23, 560–574 (2026). https://doi.org/10.1038/s41423-026-01408-9
Keywords: liver metastasis, LRG1, immune microenvironment, neutrophil extracellular traps, cancer immunotherapy