Clear Sky Science · en
Loss of Pum2 exacerbates colitis by disrupting macrophage–epithelial crosstalk and promoting epithelial necroptosis
Why this matters for people with gut troubles
Ulcerative colitis is a chronic disease in which the lining of the large intestine becomes painfully inflamed and prone to flare-ups. Many patients cycle between periods of calm and sudden bouts of bleeding, cramps, and urgent bowel movements. This study uncovers a previously overlooked "brake" inside gut immune and lining cells—a protein called Pum2—that helps keep inflammation in check. Understanding how this internal brake works could point to new ways to calm the gut, protect the intestinal wall, and perhaps improve treatments for ulcerative colitis.
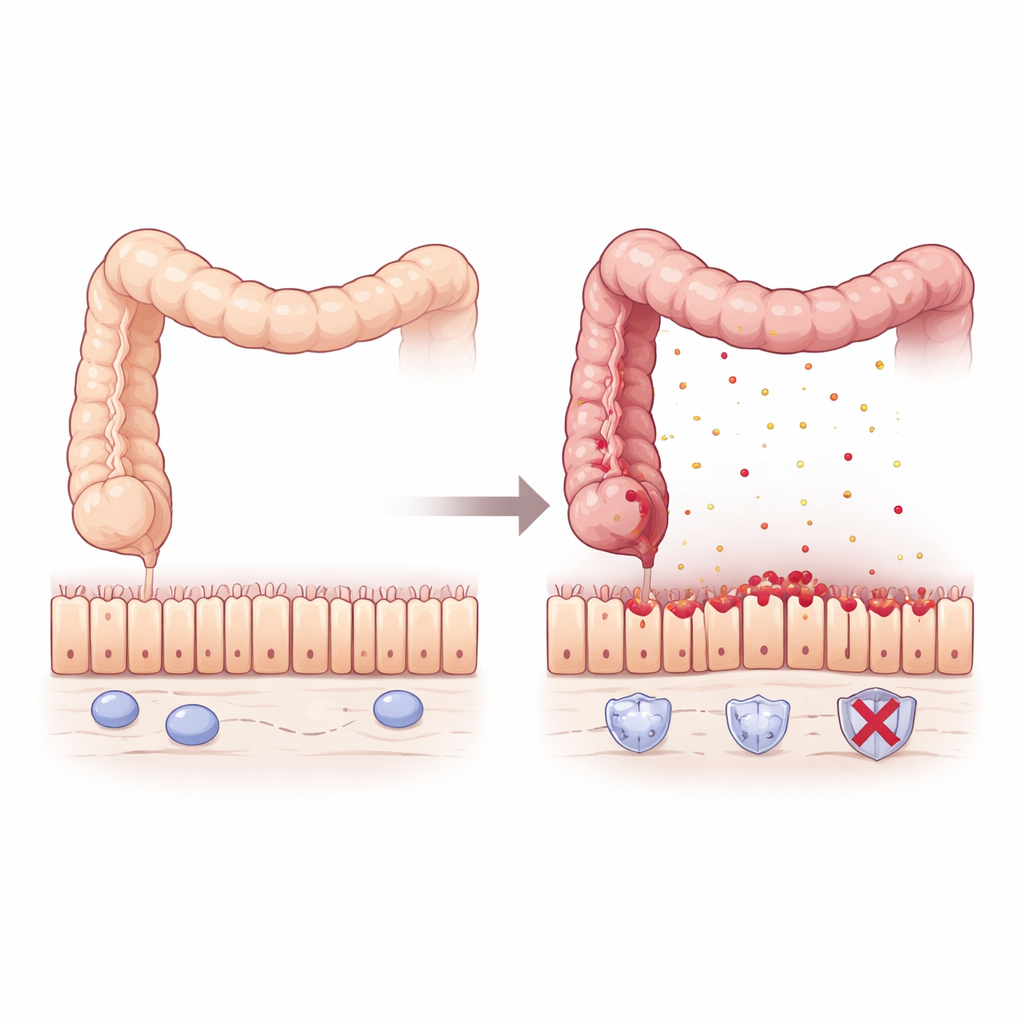
A quiet guardian in the inflamed gut
The researchers began by mining large collections of patient tissue data from people with inflammatory bowel disease and colorectal cancer. They found that levels of Pum2, a protein that binds to RNA and fine-tunes how other proteins are made, were consistently lower in actively inflamed areas of the colon and rectum. When the gut lining healed after treatment, Pum2 levels rose again, especially in patients whose disease went into sustained remission. Intriguingly, in intestinal tumors that arise after long-term colitis, Pum2 was turned back on to levels similar to healthy tissue, hinting that this protein may play different roles during inflammation and cancer.
When the brake is missing, colitis gets worse
To see what happens when Pum2 is lost, the team used mice engineered to lack the Pum2 gene and exposed them to a chemical that triggers colitis by damaging the intestinal lining. Compared with normal mice, animals without Pum2 lost more weight, had shorter, more inflamed colons, larger spleens, and higher death rates. Microscopic examination showed severe disruption of the gut lining, with distorted glands and heavy immune-cell infiltration. Even before obvious disease appeared, genetic profiling revealed that Pum2-deficient colons were "primed" for trouble: they quietly produced more chemokines—signal molecules that attract inflammatory cells—especially those that draw in macrophages, a key type of immune cell.
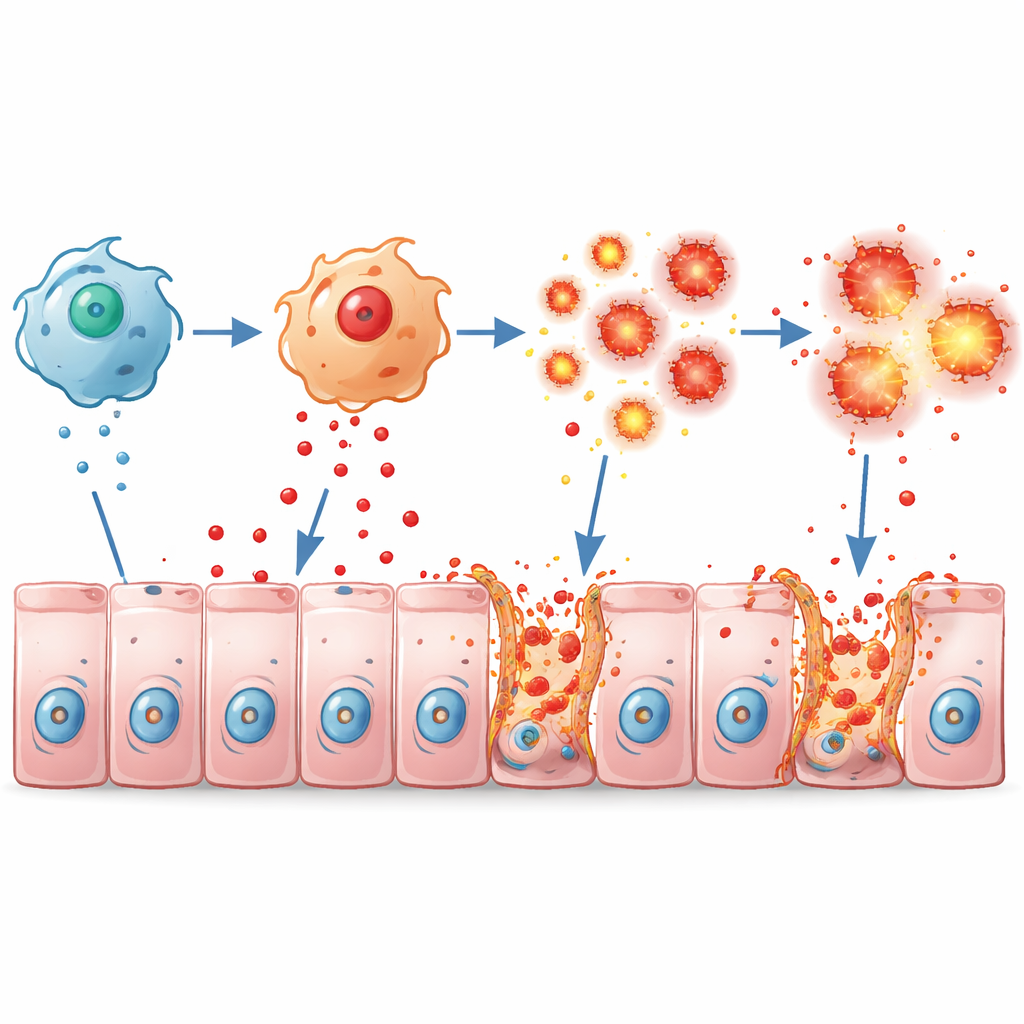
Overheated immune cells and a fragile barrier
Macrophages normally help defend the gut but can drive damage when overactivated. In lab dishes, macrophages from Pum2-lacking mice responded to bacterial components by pumping out far more inflammatory messengers such as TNFα, IL-1β, and IL-6, and adopted a strongly aggressive state. In living mice with colitis, these hyperactive macrophages crowded into the colon and flooded the tissue with TNFα. The gut lining responded with widespread cell death and breakdown of the tight junctions—protein "zippers" like ZO-1, Occludin, and Claudin1—that usually seal neighboring cells together. The result was a leaky, fragile barrier that allowed inflammation to feed on itself.
How stressed cells die and open the floodgates
The study also zoomed in on the intestinal lining cells themselves. Pum2 normally helps these cells keep reactive oxygen species—highly reactive forms of oxygen—under control. When Pum2 was boosted in human intestinal cell lines, the cells produced less oxidative stress in response to hydrogen peroxide or TNFα, and they were less likely to die. Without adequate Pum2, oxidative stress rose, and a specific form of explosive cell death called necroptosis switched on, marked by activation of molecules like RIPK1 and MLKL that punch holes in cell membranes. In a co-culture system that mimicked the inflamed gut, macrophages bathed epithelial cells in TNFα, which triggered necroptosis, leakage of cell contents, and loss of barrier proteins—effects that could be largely reversed either by restoring Pum2 in macrophages or by blocking TNFα.
What this means for future care
Taken together, the findings paint Pum2 as a crucial peacekeeper at the border between gut immune cells and the intestinal wall. When Pum2 levels fall during a flare, macrophages become overzealous and produce excess TNFα, while epithelial cells become more vulnerable to oxidative stress and necroptotic death. This toxic conversation erodes the barrier and worsens colitis. Because Pum2 levels track with disease activity and recovery, and because boosting Pum2 dampens damaging signals in both immune and lining cells, targeting this pathway—or preserving Pum2 activity—could offer a new strategy to strengthen the gut barrier and reduce flares in ulcerative colitis.
Citation: Wang, X., Han, X., Qiu, W. et al. Loss of Pum2 exacerbates colitis by disrupting macrophage–epithelial crosstalk and promoting epithelial necroptosis. Cell Death Discov. 12, 137 (2026). https://doi.org/10.1038/s41420-026-03041-x
Keywords: ulcerative colitis, intestinal barrier, macrophages, cell death, RNA-binding proteins