Clear Sky Science · en
Endothelial cell senescence shapes T cell activity in late-stage of chronic obstructive pulmonary disease
Why this matters for people with lung disease
Chronic obstructive pulmonary disease (COPD) is a major cause of breathlessness, hospital visits, and early death around the world, yet today’s medicines mainly ease symptoms rather than stop the disease from getting worse. This study asks a deeper question: what actually changes inside the lungs as COPD advances from mild to severe? By combining big biobank data, cutting-edge single-cell analyses, and machine learning, the authors uncover a hidden interaction between aging blood vessels in the lung and the immune cells that should protect us from infections.
How the researchers searched the blood for warning signals
To find molecules linked to COPD severity, the team started with blood samples from tens of thousands of people in the UK Biobank. Using machine learning, they compared protein patterns in people without COPD, those with the disease, and those with different levels of lung function. This approach highlighted a small group of proteins whose levels rose as COPD appeared and worsened. Two in particular, IL6 and MMP12, stood out: higher levels were tied not only to having COPD but also to more severe disease and poorer survival over time. Among these, IL6 emerged as the more stage-specific signal when the researchers later looked inside lung tissue itself.
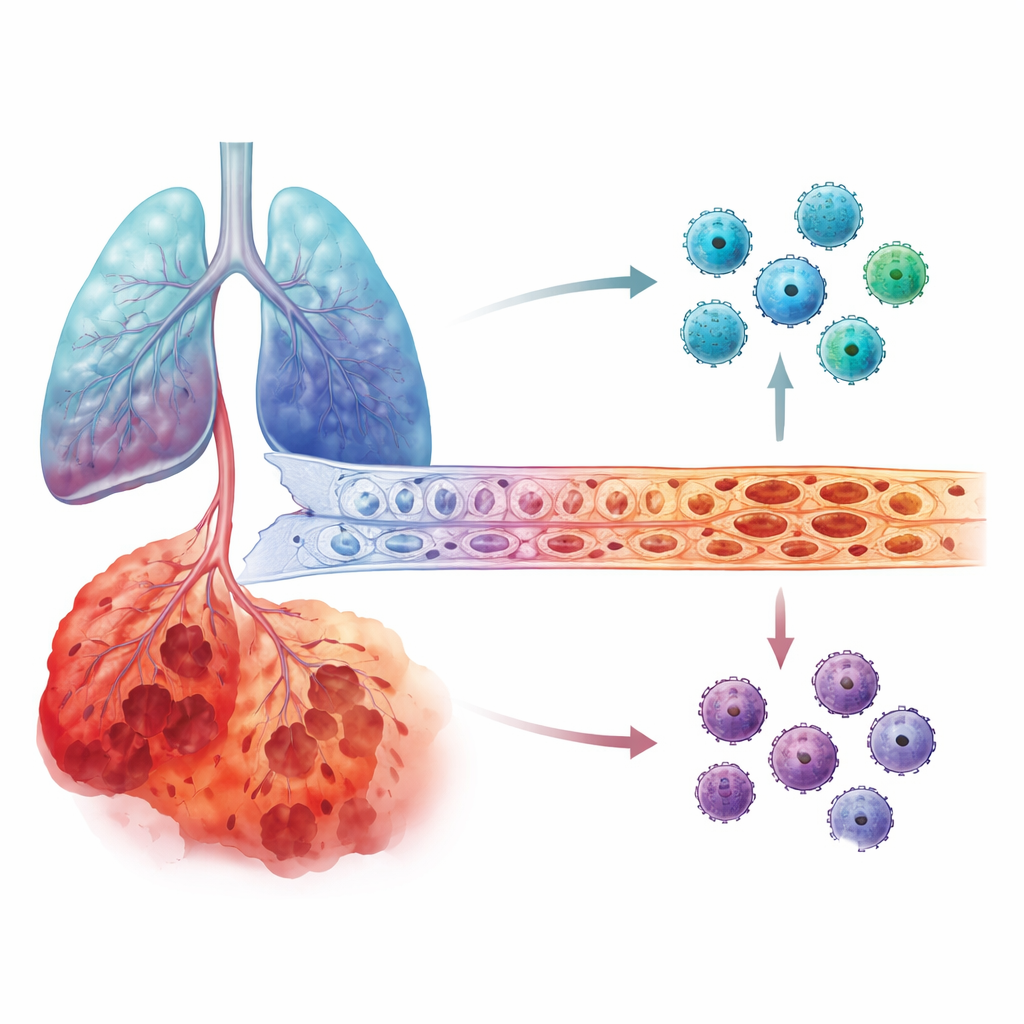
What they saw inside damaged lungs
The investigators next turned to lung tissue datasets, including both bulk measurements of gene activity and single-cell RNA sequencing, which can track what each individual cell type is doing. They found that IL6 gene activity was clearly higher in COPD lungs than in healthy lungs. Most strikingly, in the most severe cases, IL6 was concentrated in the cells lining lung blood vessels, known as endothelial cells, as well as in the delicate air sac cells. In contrast, MMP12 did not show such a sharp stage-specific jump. Across several independent patient cohorts, the same pattern emerged: as COPD advanced, endothelial cells in late-stage lungs became a dominant source of IL6.
When blood vessel cells grow old and restless
Digging deeper, the team asked why endothelial cells in severe COPD make so much IL6. Their gene patterns bore the hallmarks of cellular senescence—a form of permanent growth arrest often described as “aged” or “worn-out” cells. These cells expressed classic senescence genes, activated senescence-linked control switches, and produced a cocktail of inflammatory molecules known as the senescence-associated secretory phenotype. Spatial transcriptomics, which preserves where cells sit in real lung slices, showed that endothelial cells rich in senescence markers also pumped out more IL6. Blood from patients with severe COPD likewise carried stronger senescence-related protein signatures, suggesting this aging process is not just local but reflected throughout the body.
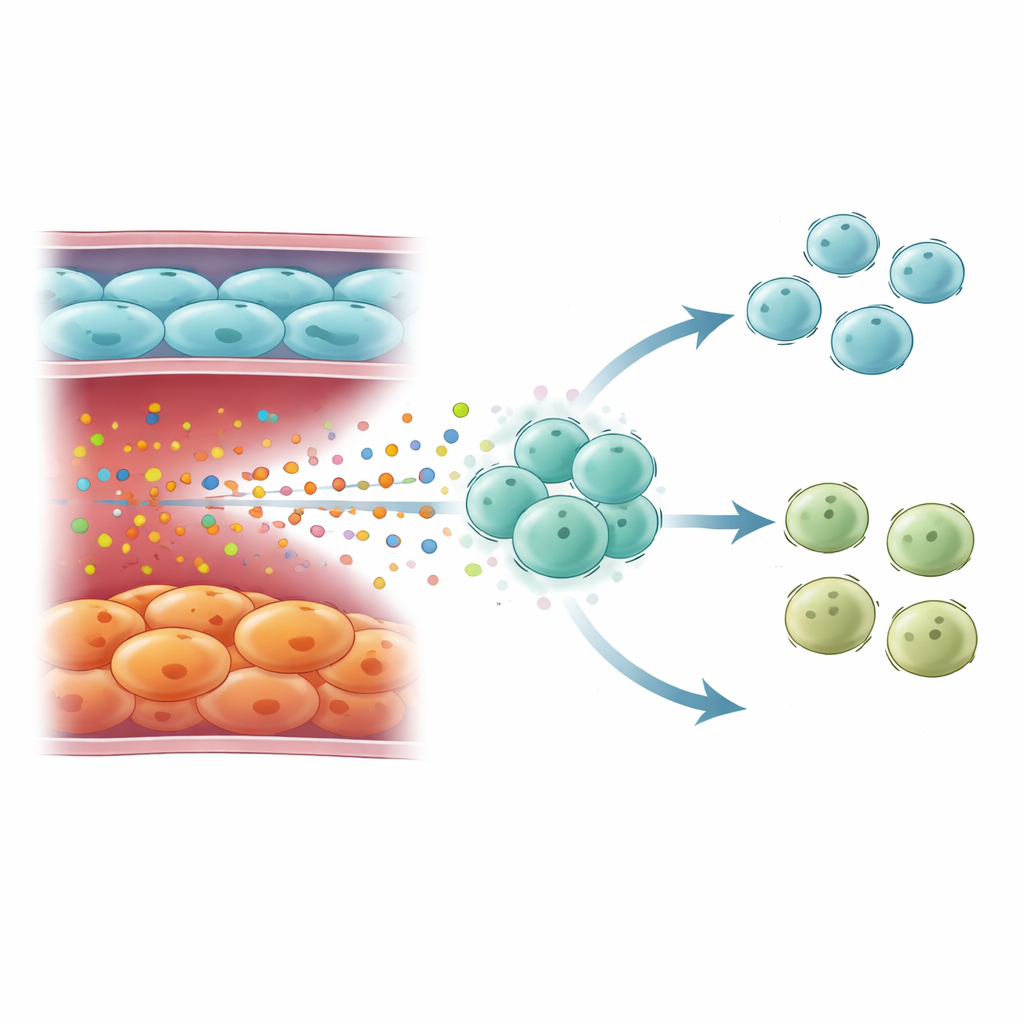
How aging vessels reshape immune defenses
High IL6 from senescent endothelial cells did not act in isolation. Single-cell maps of immune cells revealed that these signals were directed mainly toward naïve T cells, the versatile white blood cells that can become many different helper types. In severe COPD, T cell populations shifted: naïve CD4 T cells accumulated, while type 1 helper T cells (Th1)—important for fighting viruses and bacteria through the cytokine interferon-gamma—were depleted. Gene signatures of interferon-gamma responses dropped in severe disease, even though they could be higher in COPD overall when mild and moderate stages were included. T cells in late-stage COPD showed strong activation of IL6-responsive pathways and STAT3, consistent with long-term IL6 exposure steering them away from a robust Th1 fate and toward a less protective state.
What this means for future treatments
For people living with COPD, these findings offer a new way to think about why the disease becomes harder to control over time. The study suggests that in late-stage COPD, aging endothelial cells in lung blood vessels act like chronic signal beacons, releasing IL6 that dampens a key arm of immune defense. This could help explain why patients with severe disease are more prone to infections and frequent flare-ups. The work also implies that drugs targeting IL6 or senescent cells might only be effective if tailored to those with advanced COPD, rather than applied uniformly across all stages. In short, the paper reframes severe COPD not just as worsening inflammation, but as a stage marked by aging blood vessels that quietly disarm the immune system.
Citation: Lee, C.M., Kim, J., Song, J. et al. Endothelial cell senescence shapes T cell activity in late-stage of chronic obstructive pulmonary disease. Cell Death Discov. 12, 160 (2026). https://doi.org/10.1038/s41420-026-03020-2
Keywords: COPD, endothelial senescence, IL6 signaling, T cell immunity, chronic lung disease