Clear Sky Science · en
Doxorubicin promotes the production of inflammatory cytokines in tumor-associated macrophages through activating lactate dehydrogenase A
Turning a Cancer Drug into an Immune Booster
Glioblastoma is one of the deadliest brain cancers, in part because it hides behind a protective shield of “friendly” support cells that help the tumor evade the immune system. This study shows that a long‑used chemotherapy drug, doxorubicin, can be repurposed to wake up those support cells and push them to attack the tumor instead of protecting it. By rewiring how these cells use sugar, the drug helps convert a silent, suppressive tumor niche into a hotter, more hostile environment for cancer.
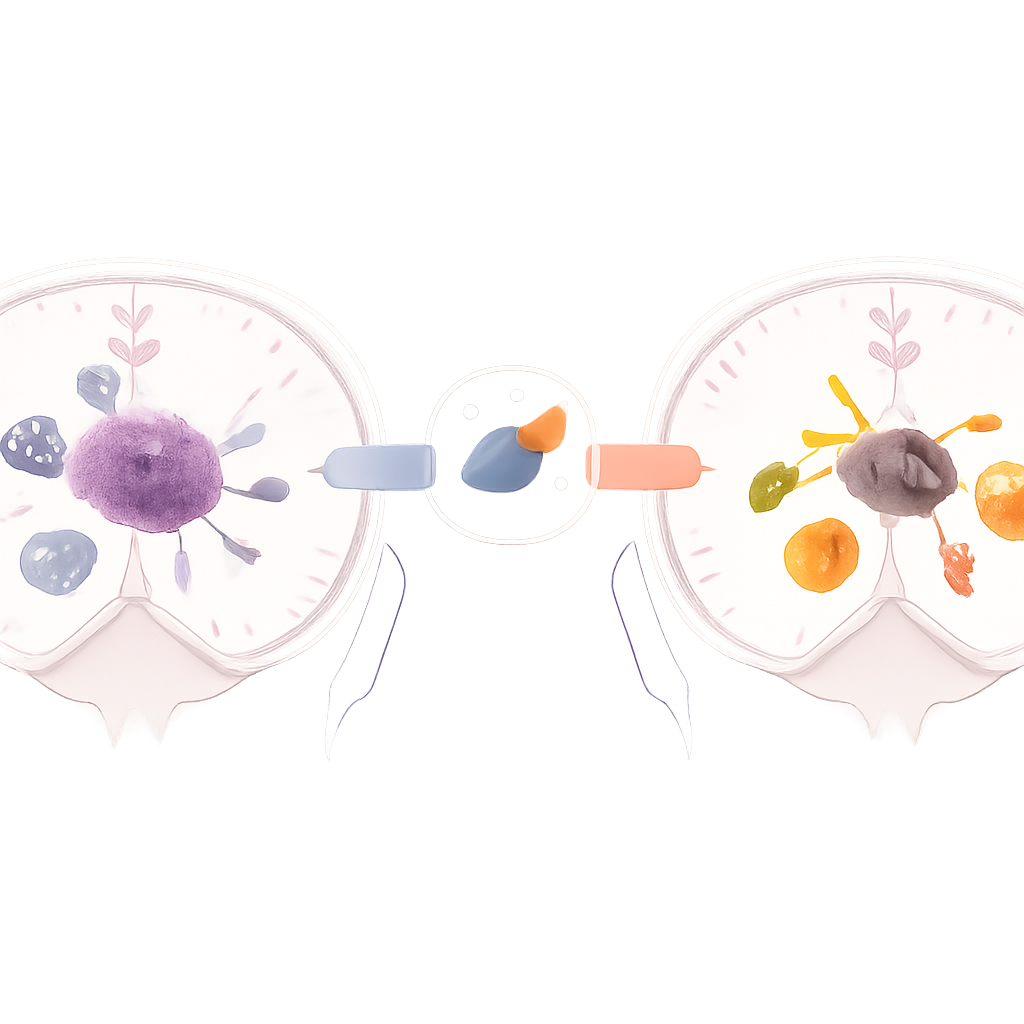
The Challenge of a Quiet Tumor Neighborhood
Standard glioblastoma treatment—surgery, radiation, and the drug temozolomide—has done little to improve survival over decades. One major reason is the tumor’s local neighborhood, or microenvironment, which is packed with immune‑related cells called tumor‑associated macrophages and microglia. These cells can either act as fierce defenders (inflammatory “fighters”) or as calm, wound‑healing helpers (suppressive “healers”). In glioblastoma, they skew heavily toward the healer state, dampening immunity and helping the tumor resist therapy. The authors set out to learn whether doxorubicin, a classic chemotherapy drug already known to stir up immune responses, could reprogram these cells into fighters and thereby make existing treatments work better.
More Time for Mice and a Less Friendly Tumor
Using a mouse model of glioblastoma, the team combined radiation and temozolomide with doxorubicin. Mice that received the triple therapy lived longer than those given radiation and temozolomide alone, even though the overall number of immune‑type cells in the tumors did not change. Instead, what changed was their behavior. Markers associated with the suppressive, tumor‑aiding state fell, while markers linked to an activated, anti‑tumor state rose. At the same time, levels of inflammatory messenger proteins such as IL‑6, TNF‑α, and IL‑1β were higher inside tumors, in bone‑marrow‑derived macrophages from treated mice, and in the animals’ blood. Together, these shifts indicate that doxorubicin does not simply add more immune cells—it flips existing ones into a more aggressive, tumor‑opposing mode.
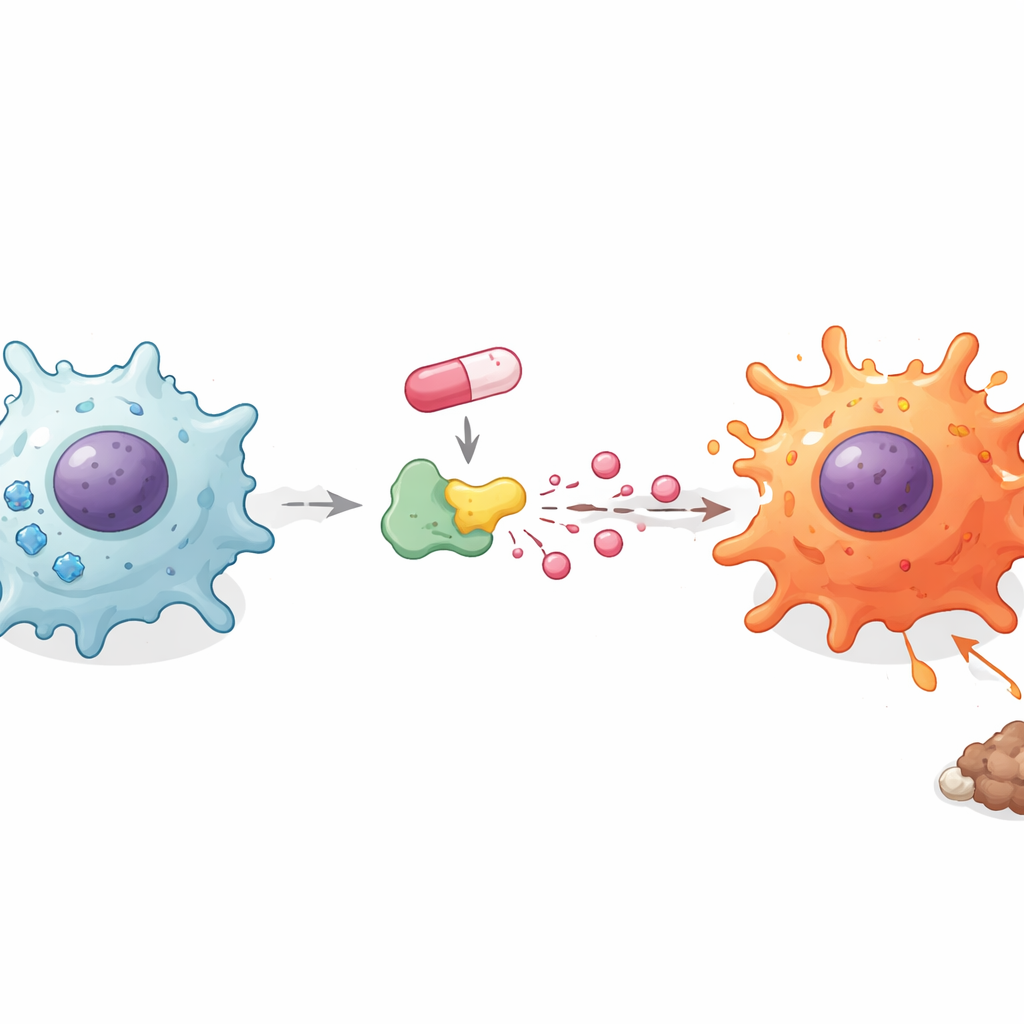
How Sugar Use and Acids Drive Immune Awakening
Macrophages change their fuel use when they switch between quiet and inflammatory states. The researchers probed how doxorubicin affected this hidden layer of metabolism. Surprisingly, the drug did not make cells take up more sugar overall, but blocking sugar breakdown with a standard inhibitor erased the boost in inflammatory messengers. That led the team to focus on what happens to sugar after it enters the cell. They found that doxorubicin lowered the cell’s energy stores yet increased the amount of lactic acid released into the surroundings. Adding extra lactate alone pushed macrophages and microglia to secrete more inflammatory messengers, while blocking lactate transport had the opposite effect. These findings suggest that it is not just sugar consumption but the way sugar is processed into lactate that helps flip macrophages into fighters.
A Key Enzyme as the Drug’s Metabolic Lever
To connect doxorubicin more directly to lactate production, the team examined lactate dehydrogenase A (LDHA), a central enzyme that turns a sugar breakdown product into lactate. Computer simulations indicated that doxorubicin can bind LDHA at specific amino acids, and experiments showed that LDH activity rose in macrophages and microglia after treatment. When LDHA was blocked with a small‑molecule inhibitor or reduced using genetic tools, doxorubicin could no longer drive high levels of inflammatory messengers. In contrast, boosting LDHA made these signals stronger. Importantly, blocking LDHA also partly reversed doxorubicin’s ability to strip away suppressive markers and enhance activating markers on macrophages, tying the change in cell identity to this enzyme‑controlled lactate surge.
What This Could Mean for Patients
In simple terms, this work reveals that doxorubicin does more than damage tumor DNA: it also presses a metabolic switch in nearby support cells, encouraging them to burn sugar in a way that generates lactic acid and, in turn, powerful inflammatory signals. This helps shift macrophages and microglia from guarding the tumor to attacking it, making radiation and temozolomide more effective in mice. While challenges such as safely delivering doxorubicin to the brain remain, the study points to a new strategy for glioblastoma therapy: use metabolism‑targeting combinations that turn the tumor’s own neighborhood from an immune refuge into a battleground.
Citation: Liu, B., Yang, W., Feng, S. et al. Doxorubicin promotes the production of inflammatory cytokines in tumor-associated macrophages through activating lactate dehydrogenase A. Cell Death Discov. 12, 208 (2026). https://doi.org/10.1038/s41420-026-03014-0
Keywords: glioblastoma, tumor microenvironment, tumor-associated macrophages, doxorubicin, lactate metabolism