Clear Sky Science · en
Endometriosis immune microenvironment timing shifts: from immune escape to immune exhaustion
Why the Body’s Defenses Matter in Endometriosis
Endometriosis is a painful condition in which tissue similar to the uterine lining grows where it does not belong, often causing severe cramps, ongoing pelvic pain, and trouble getting pregnant. This article explores a less familiar side of the disease: how the body’s own defense system, the immune system, first fails to clear these misplaced cells and then gradually wears out. Understanding this shifting “immune landscape” could help explain why endometriosis persists, worsens over time, and often returns even after surgery.
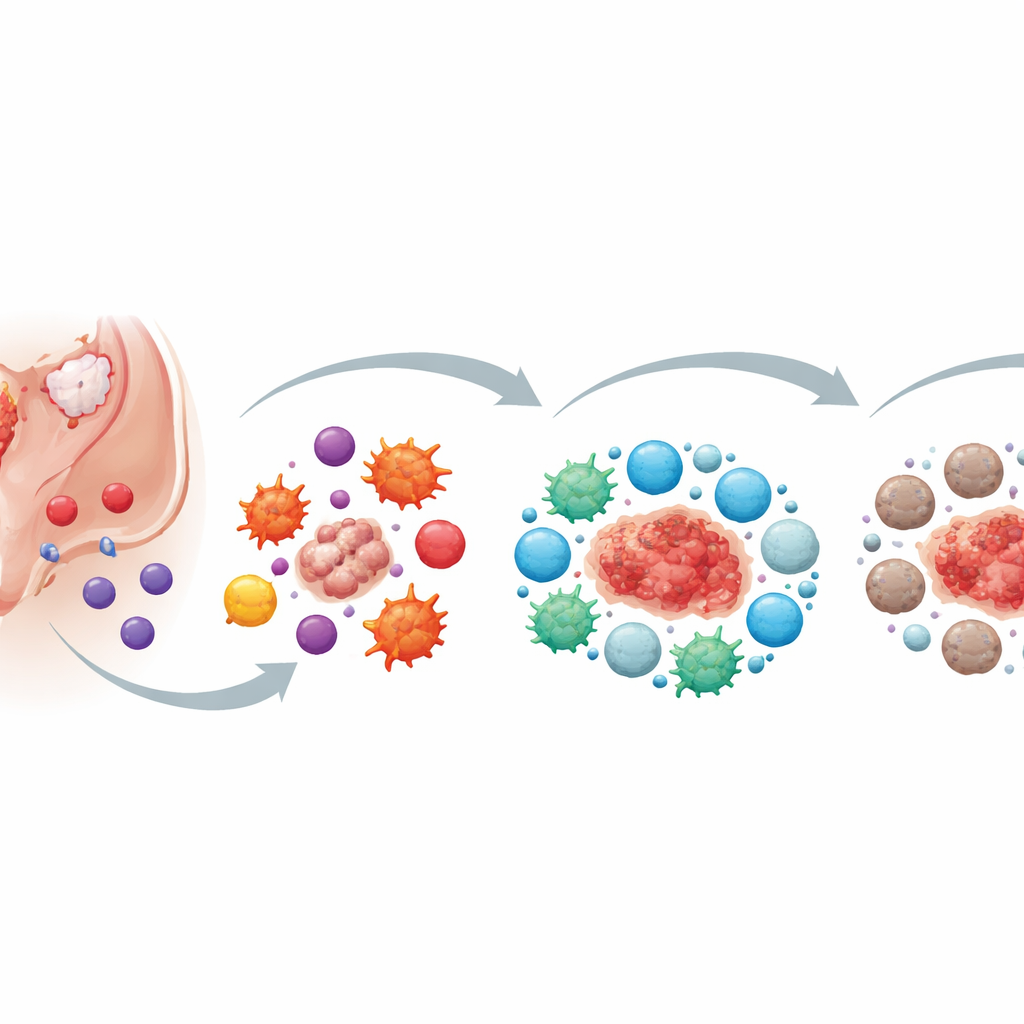
First Steps: How Problem Cells Slip Past Defense
The authors describe the earliest stage as “immune escape.” Normally, specialized white blood cells patrol the body and remove abnormal cells. In endometriosis, ectopic endometrial cells (EECs) end up in the abdominal cavity, but key “killer” cells—natural killer (NK) cells, certain T cells, and macrophages—do not remove them effectively. These killer cells are present, but their ability to attack is weakened. Their activating switches are turned down, inhibitory switches are turned up, and they are less able to move into the right place at the right time. EECs actively contribute to this failure by releasing chemical messengers and tiny membrane packages (exosomes) that dampen killer-cell activity and reduce the ability of macrophages to “eat” unwanted cells.
Monthly Cycles, Friendly Helpers, and a Storm of Inflammation
As menstrual blood and tissue flow backward into the abdominal cavity, other components of the period join the story. Platelets and the breakdown products of red blood cells further blunt the killing power of NK cells and macrophages. Instead of clearing the stray tissue, they help it stick, invade, and grow. At this point, the immune environment is strongly inflammatory: many cells release alarm signals that, in theory, should help fight the abnormal tissue. Yet these same signals also encourage blood vessel growth, cell division, and invasion by the endometriosis lesions. Inflammation becomes a double-edged sword—too weak to eliminate the lesions, but strong enough to fuel their expansion and the pain they cause.
From Hot Inflammation to a Soothing, Yet Harmful Calm
Over time, the balance of immune cells around the lesions shifts. Early on, “attack” macrophages and T helper cells dominate, releasing aggressive signals that promote inflammation. As lesions enlarge and bleed repeatedly, they begin to attract and reshape a different cast of immune cells—those that calm responses and limit damage. Anti-inflammatory macrophages, regulatory T cells, and other soothing cell types increase in number, drawn in by chemokines and “alarmin” molecules released during repeated bleeding. Platelets and local hormones, such as estrogen, also push the system toward a quieter, anti-inflammatory state. Although this calmer environment protects the surrounding tissues from chronic, uncontrolled inflammation, it simultaneously shields the lesions, promotes new blood vessel growth, and encourages the build-up of scar-like fibrous tissue.
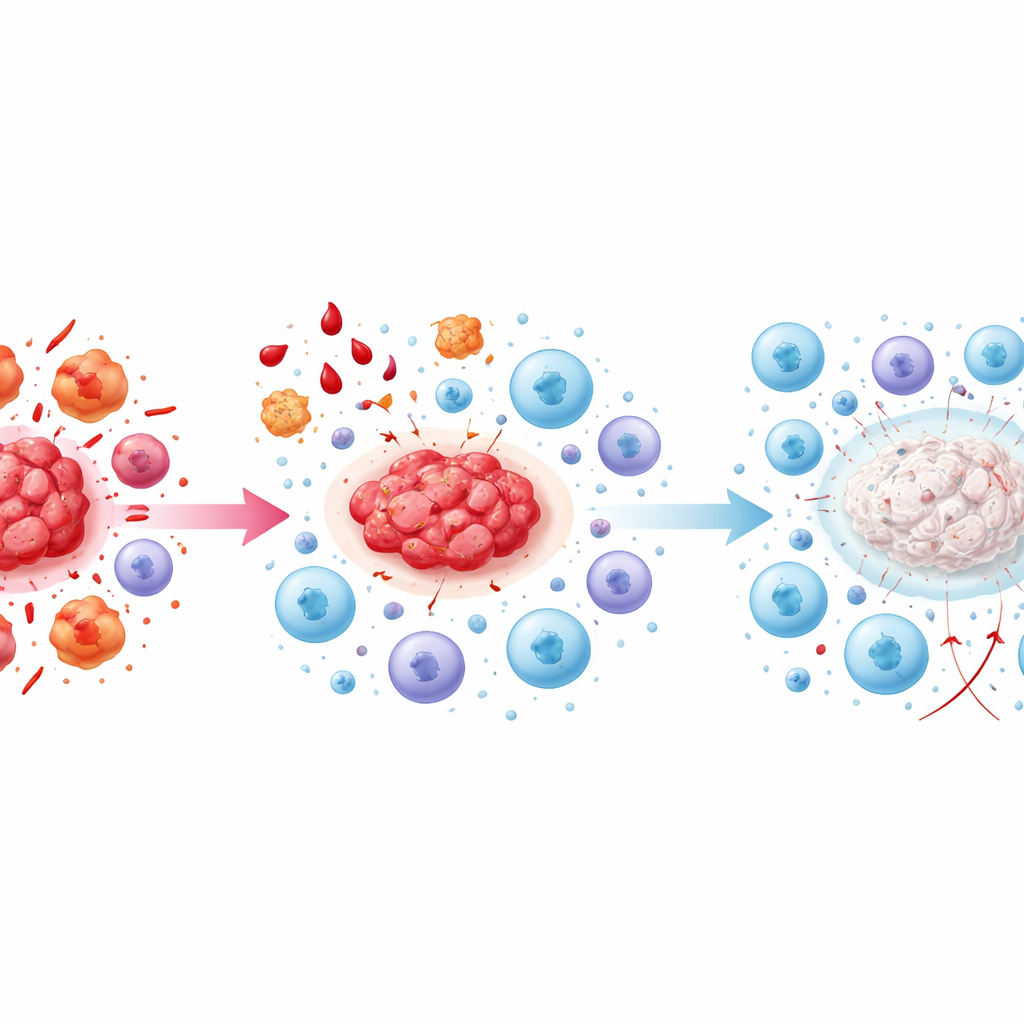
When the Killers Run Out of Energy
In the late stages, the article describes a state of “immune exhaustion.” Killer cells that once could, in principle, attack the lesions are now chronically overstimulated and continually suppressed by anti-inflammatory signals. They start to express surface markers typical of tired, ineffective cells, and their production of key attack molecules declines. Signals such as TGF-beta and IL-10, produced by anti-inflammatory cells and platelets, reinforce this exhaustion. Meanwhile, checkpoints on immune cells—molecular brakes that normally prevent excessive reactions—are increasingly engaged, further silencing the response. The result is a self-sustaining situation in which lesions persist, are richly supplied with blood, and become fibrotic, while the very cells that should control them are present but largely powerless.
What This Means for Future Treatment
To a non-specialist, the message is that endometriosis is not just a problem of wayward tissue, but of timing and balance in the body’s defenses. The review proposes that the disease moves through four immune phases: escape, early inflammation, later anti-inflammation, and finally immune exhaustion. Because each phase involves different mixes of cells and signals, treatments that work at one moment may fail at another. Strategies that carefully boost killer-cell activity, encourage the right kind of inflammation, or interrupt the harmful partnership between platelets, hormones, and calming immune cells might one day slow or even reverse lesion growth. By mapping this immune timeline, the authors aim to guide more precise, stage-matched therapies and to highlight why simple removal of lesions often isn’t enough on its own.
Citation: Fan, W., Fengting, Z. & Ruihua, Z. Endometriosis immune microenvironment timing shifts: from immune escape to immune exhaustion. Cell Death Discov. 12, 125 (2026). https://doi.org/10.1038/s41420-026-02965-8
Keywords: endometriosis, immune microenvironment, chronic inflammation, immune exhaustion, women’s health