Clear Sky Science · en
Mitofusin-2 suppresses tumor immune escape through EGFR/STAT3-mediated PD-L1 transcription
Why our cells sometimes ignore cancer
Cancer treatments that harness the immune system have transformed care for many patients, yet tumors often learn how to hide in plain sight. This study explores a little-known gatekeeper protein inside cancer cells that helps decide whether immune cells can see and attack a tumor. Understanding how this internal switch works could point to new ways to make existing immunotherapies work better and for more people.
A hidden link between cell powerhouses and cancer defenses
Inside every cell, tiny structures called mitochondria act as power plants and communication hubs. The protein Mitofusin-2 (MFN2) helps mitochondria stay connected and healthy. The researchers noticed that MFN2 levels are often low in lung and kidney cancers, and patients with less MFN2 tend to have shorter survival. At the same time, these tumors show high amounts of PD-L1, a “do not attack” signal that shuts down immune cells. By examining patient samples and large cancer databases, the team found that when MFN2 is low, PD-L1 is usually high, suggesting that this mitochondrial protein somehow restrains the tumor’s ability to hide from the immune system.
How tumors turn down MFN2 to dodge immune attack
To test this idea, the scientists altered MFN2 levels in many types of cancer cells grown in the lab, including lung, kidney, and breast cancer. When they reduced MFN2, the cells made more PD-L1 and displayed more of it on their surface, even under stressful conditions like low oxygen or poor nutrients that mimic the tumor interior. When they restored MFN2, PD-L1 dropped back down. In mouse models, tumors lacking MFN2 grew faster and had fewer cancer-fighting CD8 T cells inside them. Clinical samples from patients showed a similar pattern: tumors richer in MFN2 contained more CD8 T cells, and those patients responded better to drugs that block PD-1 or PD-L1.
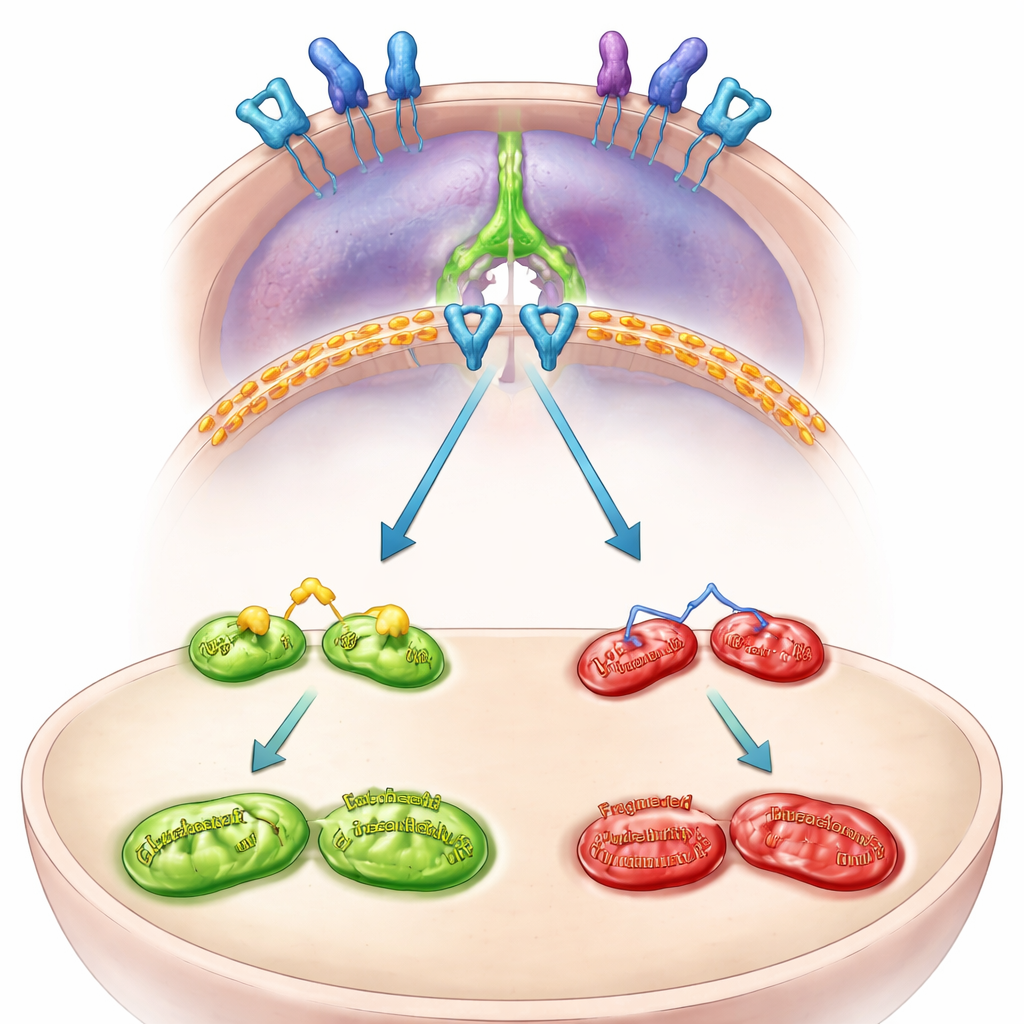
The internal wiring: from mitochondria to immune escape
The team then mapped the internal wiring that connects MFN2 to PD-L1. They found that MFN2 normally keeps a growth signal on the cell surface, called EGFR, in check. When MFN2 is lost, EGFR becomes more active and flips on another protein, STAT3. Activated STAT3 travels into the cell’s nucleus and boosts the instructions for making PD-L1. Blocking EGFR or STAT3 in MFN2-deficient cells prevented PD-L1 from rising and reduced its presence on the cell surface. In patient-derived mini-tumors grown as three-dimensional organoids, silencing MFN2 again switched on EGFR and STAT3 and drove up PD-L1, confirming that this pathway operates in human tumors, not just in cell lines.
Turning cold tumors hot again
Because PD-L1 helps tumors become “cold” to immune attack, the scientists asked whether shutting down STAT3 could reverse this effect in animals. In mice with MFN2-low tumors, treatment with STAT3-blocking drugs slowed tumor growth, restored the presence of CD8 T cells inside the tumors, and boosted immune messenger molecules that help T cells gather and stay active. These changes suggest that even when tumors have already dialed down MFN2, it may still be possible to reopen the door to immune attack by targeting the downstream EGFR–STAT3 pathway.
What this means for future cancer treatment
Overall, the study reveals that MFN2, a protein best known for maintaining mitochondrial health, also acts as a brake on a powerful immune-shielding pathway inside cancer cells. When MFN2 is lost, EGFR and STAT3 drive up PD-L1, helping tumors escape both recognition and destruction by immune cells. Measuring MFN2 in tumors could help predict who will benefit from current PD-1/PD-L1 drugs, and combining EGFR or STAT3 inhibitors with checkpoint therapies might turn resistant, “cold” tumors into ones the immune system can once again recognize and attack.
Citation: Liu, Y., Wang, N., Li, Z. et al. Mitofusin-2 suppresses tumor immune escape through EGFR/STAT3-mediated PD-L1 transcription. Cell Death Dis 17, 364 (2026). https://doi.org/10.1038/s41419-026-08668-3
Keywords: tumor immune escape, PD-L1 regulation, mitochondrial dynamics, EGFR STAT3 signaling, cancer immunotherapy resistance