Clear Sky Science · en
CircIQGAP1-CARM1 axis promotes renal cell carcinoma progression through glycolytic reprogramming
Why Kidney Cancer’s Sugar Habit Matters
Kidney cancer, especially the common clear cell type, is on the rise worldwide and often goes undetected until it has already spread. Like many tumors, these cancers rewire how they use sugar, burning glucose at high speed even when oxygen is plentiful. This work uncovers a hidden molecular circuit that lets kidney cancer cells lean into that sugar addiction, grow faster, and become more aggressive—and suggests new ways doctors might cut off the tumor’s energy supply or better predict which patients face poorer outcomes.
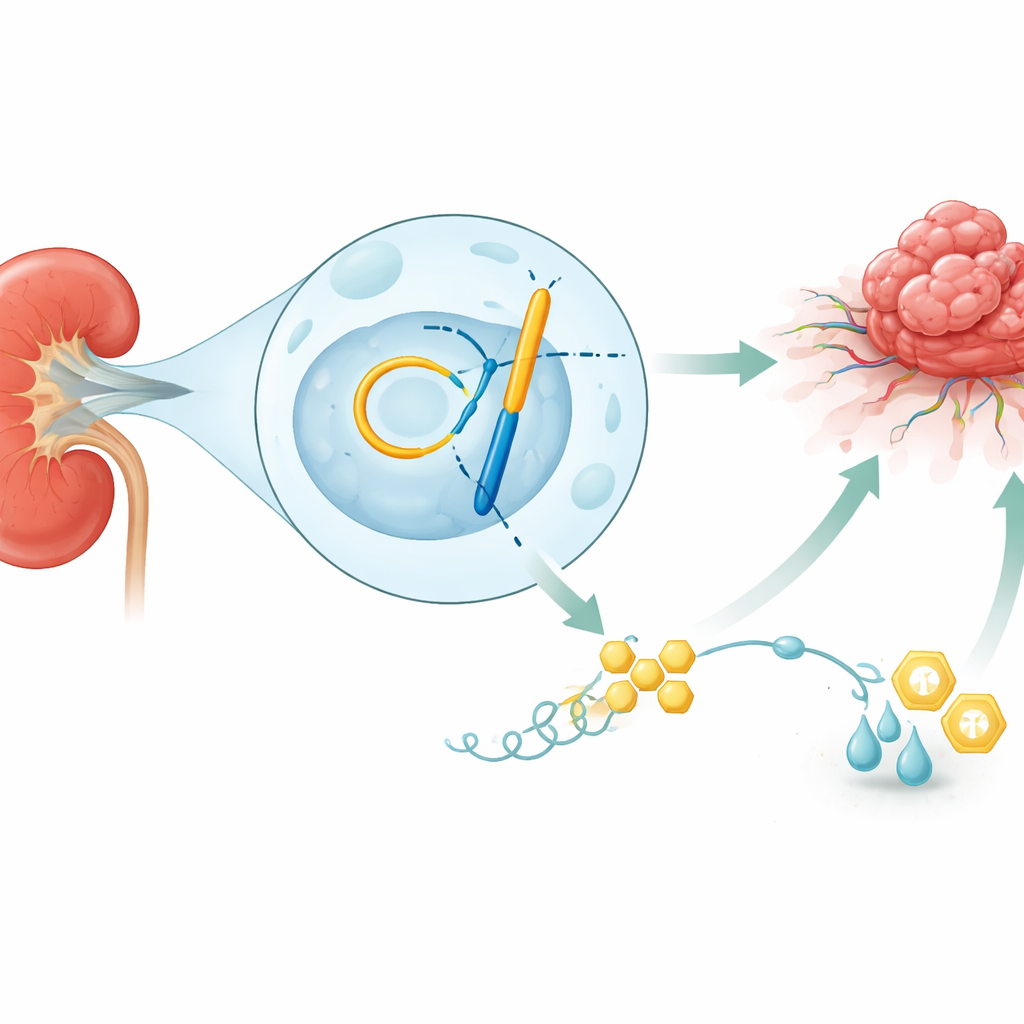
A Cancer That Loves Sugar
Our kidneys quietly filter blood around the clock, but when renal cell carcinoma develops, some of those cells turn into sugar-hungry factories. Instead of using the efficient, oxygen-dependent route to make energy, they favor a quick-and-dirty pathway that converts glucose to lactate. This “turbo mode” not only churns out enough fuel and building blocks to sustain rapid growth, it also acidifies the surroundings and weakens nearby immune cells. The study shows that kidney tumors with this revved-up metabolism tend to be more invasive, more resistant to treatment, and linked to worse patient survival, underscoring why it is vital to understand what flips this metabolic switch.
A Circular Molecule Steps Into the Spotlight
Researchers looked for unusual RNA molecules that change when kidney cancer cells are starved of glucose. They discovered one standout: a circular RNA called circIQGAP1. Unlike typical linear RNA, this molecule forms a closed loop, making it highly stable in cells. Under low-sugar stress, cancer cells ramp up circIQGAP1, with help from a splicing protein called U2AF2 that boosts its production. Laboratory tests showed that extra circIQGAP1 makes kidney cancer cells divide faster, move more easily, invade surrounding tissue, and, crucially, increase their glycolytic—or sugar-burning—activity. When circIQGAP1 was silenced, the cells produced less lactate and ATP, slowed their growth, and were more likely to die, and blocking glycolysis erased much of circIQGAP1’s tumor-promoting power.
A Protein Guardian That Stabilizes a Key Switch
Digging deeper, the team asked how this circular RNA reshapes cancer metabolism. They found that circIQGAP1 physically binds to a protein enzyme called CARM1, which is known for modifying other proteins and altering which genes are turned on. Normally, CARM1 is tagged with molecular “flags” that signal it for destruction. CircIQGAP1 acts like a shield, preventing one specific type of degradation tag from attaching to CARM1. As a result, CARM1 lingers longer and accumulates inside the cell. This stabilized CARM1 is strongly linked to poorer survival in kidney cancer patients, and when researchers reduced CARM1 levels, they could blunt the growth and glycolytic surge driven by circIQGAP1, showing that the circular RNA and the enzyme act together as a functional unit.
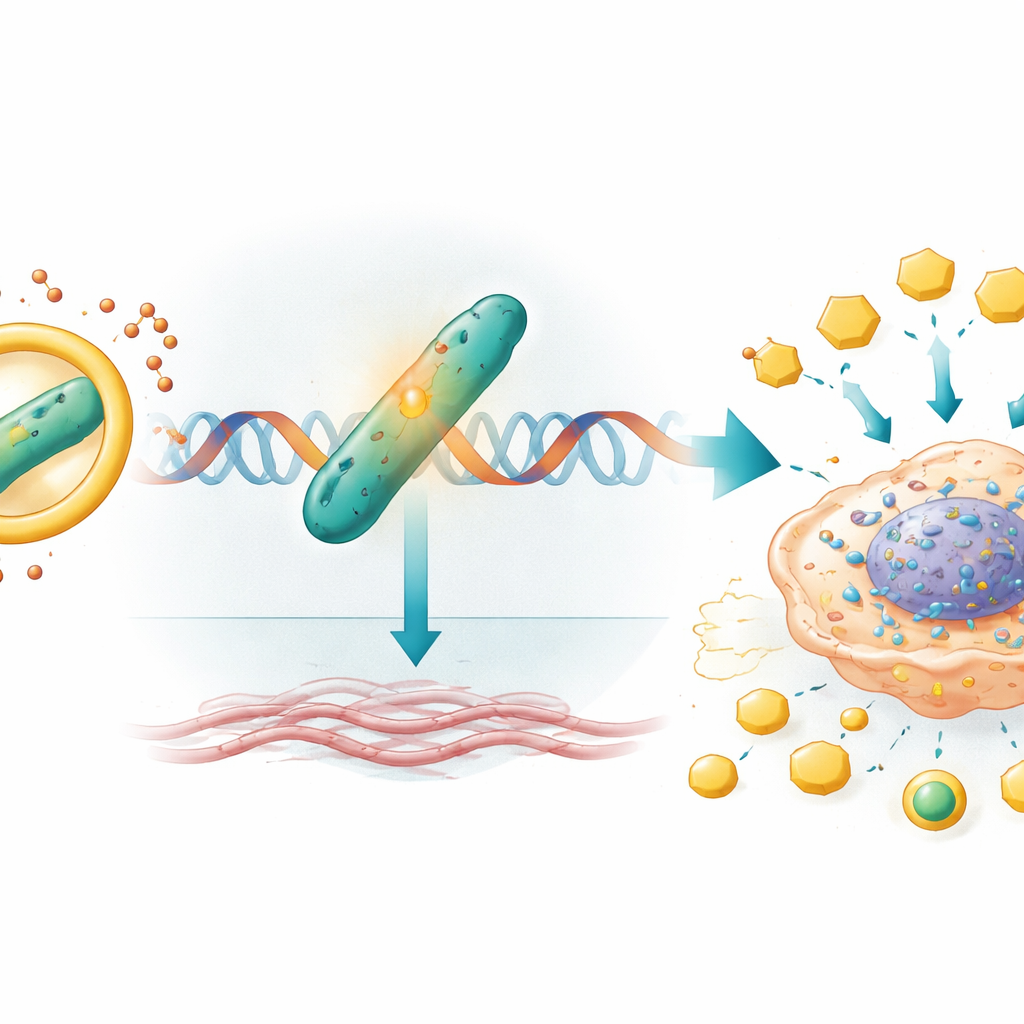
From Gene Switches to a Hardened Tumor Environment
Once protected by circIQGAP1, CARM1 travels to the cell’s genetic material and boosts the activity of a gene called COL5A1. This gene encodes a component of collagen, a structural protein that helps form the tumor’s surrounding scaffold. CARM1 chemically modifies histones—proteins that package DNA—near the COL5A1 switch, making that gene easier to read and more actively transcribed. Higher COL5A1 levels are tied to stiffer, more invasive tumors and have been associated with worse outcomes in kidney cancer and other malignancies. In this study, turning off COL5A1 reduced lactate and ATP production and completely erased the boost in glycolysis, growth, migration, and invasion that circIQGAP1 and CARM1 usually provide, marking COL5A1 as a crucial downstream driver of this metabolic reprogramming.
What This Means for Patients
When the researchers implanted kidney cancer cells with high circIQGAP1 into mice, the animals developed larger, faster-growing tumors rich in CARM1, COL5A1, and a proliferation marker. Human patients with renal cell carcinoma also showed higher circIQGAP1 levels in their blood, and those with more of this circular RNA tended to have poorer survival. Together, the findings outline a clear chain of events: U2AF2 boosts circIQGAP1, circIQGAP1 stabilizes CARM1, CARM1 switches on COL5A1, and this trio pushes kidney cancer cells into a sugar-fueled, aggressive state. Because each step in this axis can, in principle, be targeted—with RNA-based approaches, enzyme inhibitors, or drugs that disrupt collagen remodeling—this work points toward new strategies to both monitor and treat kidney cancers that rely heavily on altered glucose metabolism.
Citation: Jia, R., Zou, B., Liang, Y. et al. CircIQGAP1-CARM1 axis promotes renal cell carcinoma progression through glycolytic reprogramming. Cell Death Dis 17, 414 (2026). https://doi.org/10.1038/s41419-026-08661-w
Keywords: renal cell carcinoma, cancer metabolism, circular RNA, CARM1, glycolysis