Clear Sky Science · en
Investigating PKD2 deficiency-associated cardiomyopathies using hESC-cardiomyocytes and bioengineered 3D ventricular cardiac tissue strips
Why Kidney Disease Matters to the Heart
People with autosomal dominant polycystic kidney disease (ADPKD) are usually thought of as kidney patients, but many also develop serious heart problems. Doctors have long seen thickened heart walls, weak pumping, and abnormal rhythms in these patients, yet the direct link between the faulty genes that cause kidney cysts and damage to heart muscle has remained unclear. This study uses cutting-edge lab-grown human heart tissues to ask a simple but important question: how does a defect in one kidney-related protein, called polycystin-2, weaken the heart, and can that weakness be at least partly reversed?
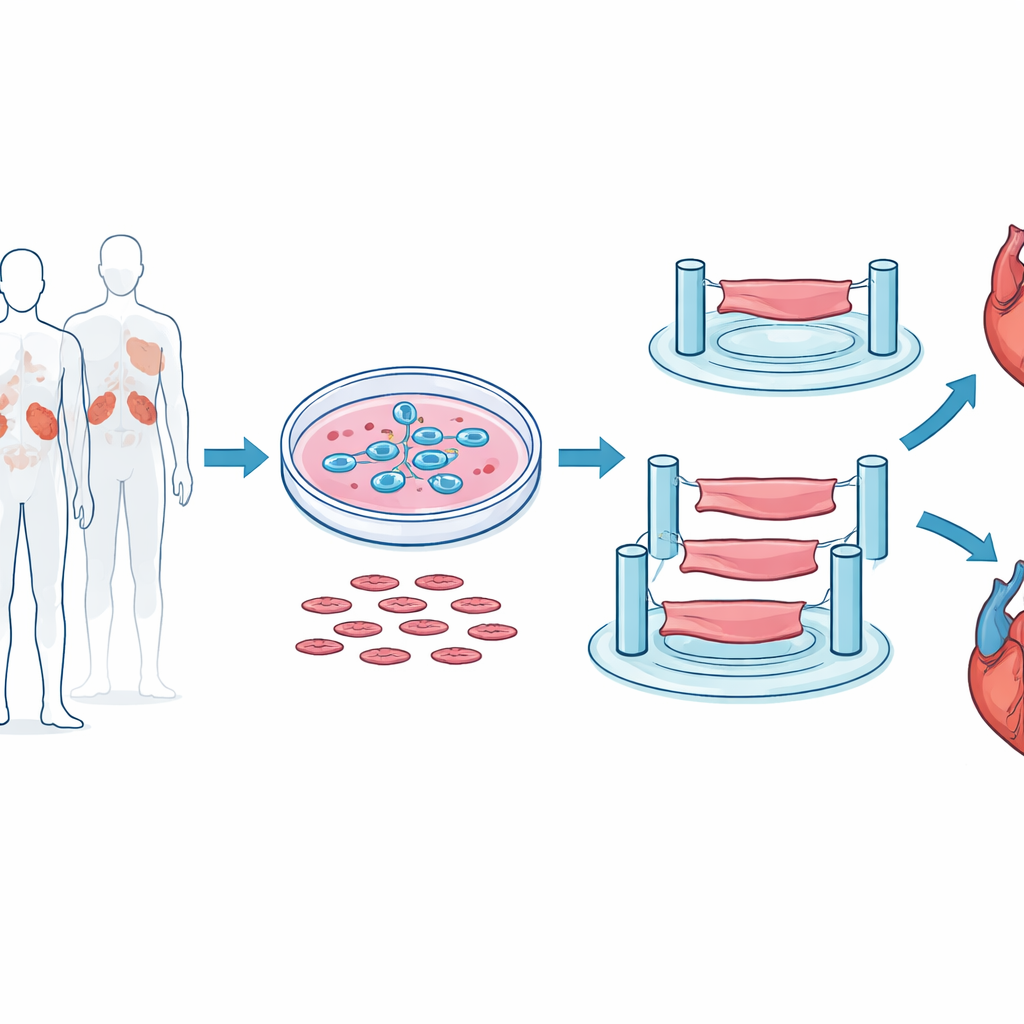
Building Mini Heart Strips in the Lab
To get as close as possible to real human heart behavior, the researchers did not rely on mouse hearts or flat cell layers. Instead, they started with human embryonic stem cells and carefully guided them to become beating heart muscle cells. These cells were then mixed with supporting cells and gel-like scaffolds and molded into tiny three-dimensional strips of ventricular tissue, each only a few millimeters long but able to contract rhythmically between flexible posts. This setup let the team measure how hard the strips pulled, how quickly they contracted and relaxed, and how they responded to electrical pacing, much like a miniature workout test for heart muscle.
What Happens When Polycystin-2 Is Lost
The team next reduced the amount of polycystin-2 in these heart cells using genetic tools that silence the PKD2 gene, mimicking the defect seen in many ADPKD patients. When polycystin-2 was knocked down, the tissue strips still beat, but they squeezed with much less force and their contractions and relaxations became sluggish. This weakness appeared in two independent stem cell lines and with different silencing methods, strengthening the case that polycystin-2 is truly important for human heart muscle performance. Importantly, the overall microstructure of the tissue—its basic organization and internal scaffolding—looked normal, pointing toward a problem with cell function rather than gross damage or disorganization.
Calcium Mismanagement and Cellular Stress
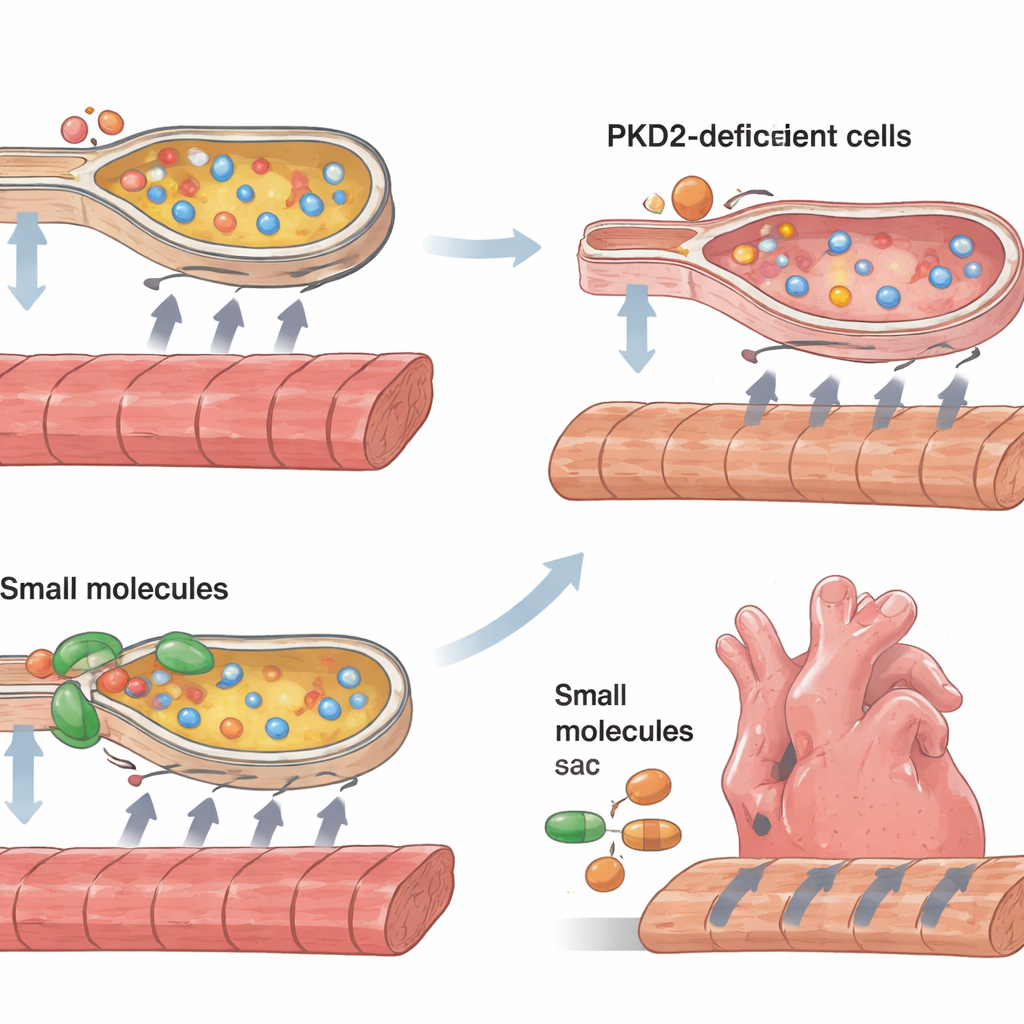
Heart cells rely on precise handling of calcium, which floods the cell to trigger contraction and is then pumped back into internal storage compartments so the cell can relax and get ready for the next beat. In cells lacking enough polycystin-2, the calcium surges during each beat were smaller and took longer to clear. The internal storage sacs, which overlap with a structure called the endoplasmic reticulum, appeared to hold less calcium and to refill more slowly. At the same time, molecular signs of endoplasmic reticulum stress rose sharply, and more cells showed markers of programmed cell death and features of enlargement associated with cardiomyopathy. A key calcium pump, SERCA2, and its regulatory partner phospholamban were both less active, offering a concrete mechanical link between the missing protein and impaired calcium cycling.
Testing Ways to Help the Failing Mini Hearts
Armed with this mechanistic picture, the researchers tried to rescue the weak tissue strips. They used two small molecules, 4-phenylbutyrate and tauroursodeoxycholic acid, that act as chemical helpers for protein folding and are known to ease endoplasmic reticulum stress. They also applied a compound called CDN1163 that boosts SERCA pump activity. All three treatments improved the strength of contraction and sped up both contraction and relaxation in the tissue strips, with particularly strong gains in strips lacking polycystin-2. The rescue was only partial—these interventions did not make the tissues completely normal—but they showed that easing cellular stress and restoring calcium pumping can meaningfully improve the performance of diseased human heart muscle in this model.
What This Means for Patients
This work shows that defects in polycystin-2 can directly weaken human heart muscle by disturbing calcium handling and overloading the cell’s internal quality-control system, even outside the context of failing kidneys or high blood pressure. By building and testing bioengineered human heart strips, the study connects a genetic glitch to a specific chain of events: reduced polycystin-2, weaker calcium pumps, rising internal stress, and ultimately sluggish, weaker contractions. Just as important, the same platform reveals that drugs already known to reduce cellular stress or enhance calcium pumping can partially restore function. While much work remains before these strategies could be tried in people, the findings point toward realistic, testable therapies for the hidden heart complications of polycystic kidney disease.
Citation: Li, J., Peng, W., Kwok, M. et al. Investigating PKD2 deficiency-associated cardiomyopathies using hESC-cardiomyocytes and bioengineered 3D ventricular cardiac tissue strips. Cell Death Dis 17, 368 (2026). https://doi.org/10.1038/s41419-026-08639-8
Keywords: polycystic kidney disease, cardiomyopathy, calcium signaling, stem cell heart models, endoplasmic reticulum stress