Clear Sky Science · en
SIRT1 deficiency promotes age-related heart failure through enhancing ferroptosis via GATA4-HADHA-GPX4 axis
Why Aging Hearts Struggle
As people live longer, more of us develop heart failure, a condition where the heart can no longer pump blood effectively. Doctors have long known that aging and excess oxidative "wear and tear" damage the heart, but the exact kind of cell damage involved has remained murky. This study uncovers a specific iron-driven form of cell death in heart muscle, and maps out a chain of molecular events that may explain why older hearts fail—and how we might slow or prevent that decline.
Iron, Rust, and Failing Hearts
The researchers began by comparing young and old rats. Older animals showed weakened pumping function, thicker and stiffer heart walls, and higher blood levels of a stress hormone released by struggling hearts. Inside their heart tissue, the team found more reactive oxygen species—highly reactive molecules that can damage fats, proteins, and DNA—and strong signs of lipid peroxidation, essentially a “rusting” of the fatty components of cell membranes. They also observed iron buildup and decreased levels of a protective enzyme called GPX4, which normally neutralizes dangerous fat-derived oxidants. Together, these changes point to ferroptosis, a recently described type of cell death in which iron and oxidized fats combine to kill cells.
Proving That Iron-Driven Cell Death Matters
To test whether this iron-related cell death actually drives heart failure rather than simply accompanying it, the scientists manipulated iron levels and the key protective enzyme GPX4. Giving aged rats a high-iron diet worsened heart function and scarring and further suppressed GPX4. In contrast, treating old rats with ferrostatin-1, a drug that specifically blocks ferroptosis, improved heart pumping, reduced scarring, and restored more normal iron-handling proteins. In mice, deleting GPX4 specifically in heart muscle cells made age-related damage and dysfunction worse, while boosting GPX4 via a gene therapy vector preserved function in a separate aging model. These experiments strongly suggest that ferroptosis is not just present in aging hearts—it actively contributes to their decline.
A Metabolic Weak Link Inside Heart Cells
Looking for what makes aged heart cells more vulnerable to ferroptosis, the team scanned thousands of proteins in rat hearts and found a striking drop in a mitochondrial enzyme called HADHA, particularly when old animals were exposed to extra iron. HADHA helps heart cells burn fats efficiently and maintain healthy mitochondrial membranes. When the scientists reduced HADHA specifically in the heart cells of young mice, the animals developed more fibrosis and biochemical signs of ferroptosis, changes that could be reversed with a ferroptosis inhibitor. In isolated heart cells, lowering HADHA disrupted mitochondrial function, boosted reactive oxygen species, depleted the antioxidant glutathione, and further suppressed GPX4. Restoring HADHA had the opposite effect, rescuing energy production and antioxidant defenses. This positions HADHA as a crucial metabolic safeguard that keeps heart cells from tipping into iron-driven death.
An Aging Switch: SIRT1 and Its Downstream Partners
The next question was why HADHA drops with age. The authors focused on SIRT1, a protein long linked to lifespan and protection against age-related diseases. In aging hearts, SIRT1 levels fell along with HADHA. In cell experiments, shutting down SIRT1 reduced both HADHA and GPX4 and shifted iron-handling proteins toward a more dangerous profile. The team discovered that SIRT1 interacts with another protein, the transcription factor GATA4, which normally boosts HADHA production. When SIRT1 activity wanes, GATA4’s ability to turn on the HADHA gene is weakened, causing HADHA levels to sink and mitochondrial stress to rise. Importantly, activating SIRT1 with resveratrol, a small molecule often discussed in the context of “healthy aging,” or directly overexpressing SIRT1 in heart cells restored GATA4 and HADHA, strengthened antioxidant defenses, reduced ferroptosis, and improved heart function in aging animal models.
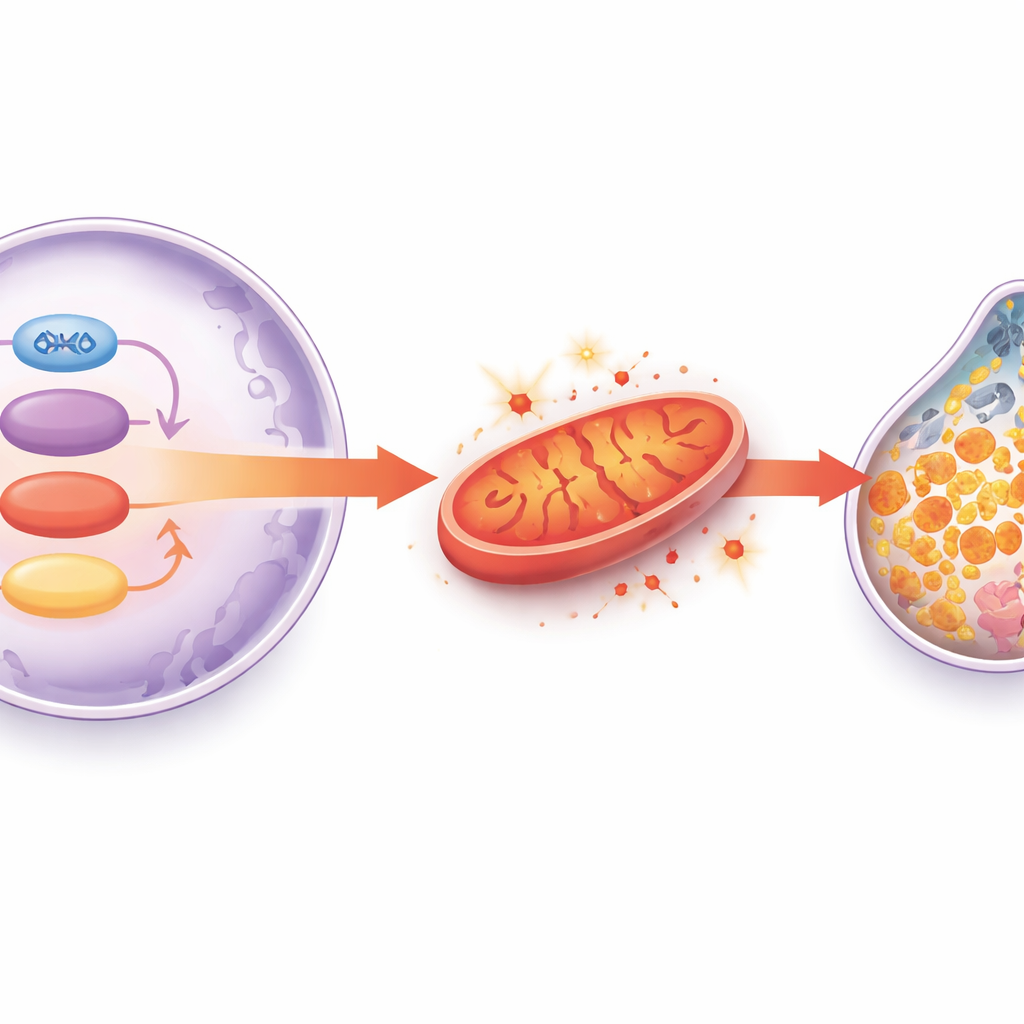
What This Means for Protecting the Aging Heart
Taken together, the study outlines a chain of events: as hearts age, SIRT1 activity drops, GATA4 can no longer keep HADHA levels up, mitochondria falter and spew reactive oxygen species, the antioxidant system centered on glutathione and GPX4 collapses, and iron-driven ferroptosis kills off heart muscle cells. This gradual loss of cells and rise in scarring ultimately weaken the heart’s pump. Although these results come from animals and cell models, they suggest that drugs or lifestyle strategies that preserve SIRT1 activity, support HADHA and mitochondrial metabolism, or directly block ferroptosis could one day help prevent or treat age-related heart failure in people.
Citation: Duan, Y., Luo, Y., Han, X. et al. SIRT1 deficiency promotes age-related heart failure through enhancing ferroptosis via GATA4-HADHA-GPX4 axis. Cell Death Dis 17, 343 (2026). https://doi.org/10.1038/s41419-026-08634-z
Keywords: aging heart failure, ferroptosis, SIRT1 pathway, mitochondrial dysfunction, cardiomyocyte death