Clear Sky Science · en
MAGI3 deficiency unleashes β-catenin conformational change to drive metastatic progression and mTOR inhibitor resistance in ccRCC
Why this kidney cancer study matters
Clear cell kidney cancer is one of the most deadly urological cancers because it often spreads and stops responding to treatment. This study uncovers a previously hidden "safety switch" inside kidney tumor cells, a protein called MAGI3, that keeps a powerful growth driver in check. When this safety switch is lost, tumors become more invasive and resistant to a commonly used drug class, mTOR inhibitors such as Everolimus. The work not only explains why some patients fare worse than others, but also points to a practical way to improve therapy by combining existing drugs.
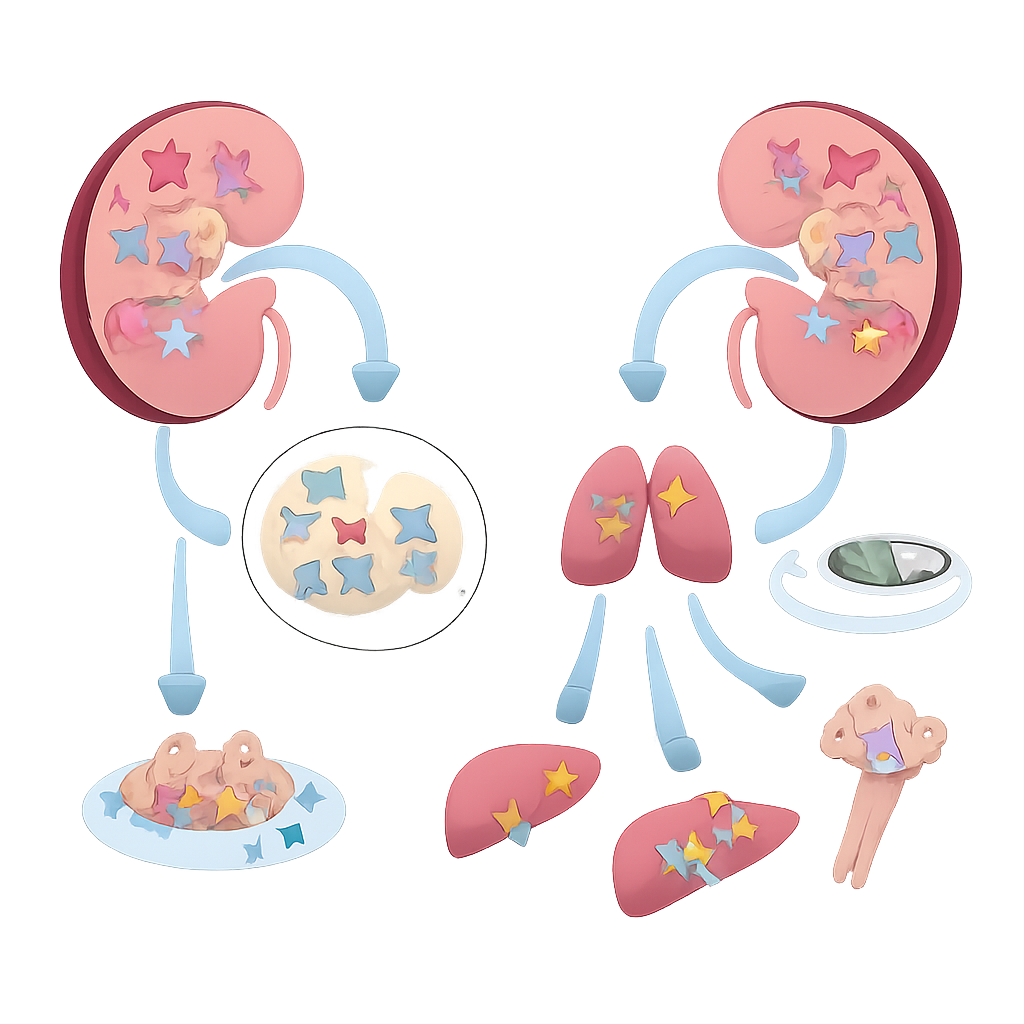
A missing guardian in kidney tumors
The researchers began by combing through large public cancer datasets to search for genes that are specifically altered when clear cell kidney cancer becomes metastatic. Among thousands of candidates, MAGI3 stood out: its activity was lower in tumor tissue than in normal kidney, and even lower in tumors that had already spread. These patterns were confirmed at the protein level in patient samples, where MAGI3 staining was strongest in normal kidney tissue, weaker in primary tumors, and weakest in metastatic deposits. Patients whose tumors had low MAGI3 tended to have more advanced stage, higher grade disease, and significantly shorter survival, regardless of classic genetic changes such as VHL mutations.
How MAGI3 reins in a powerful growth switch
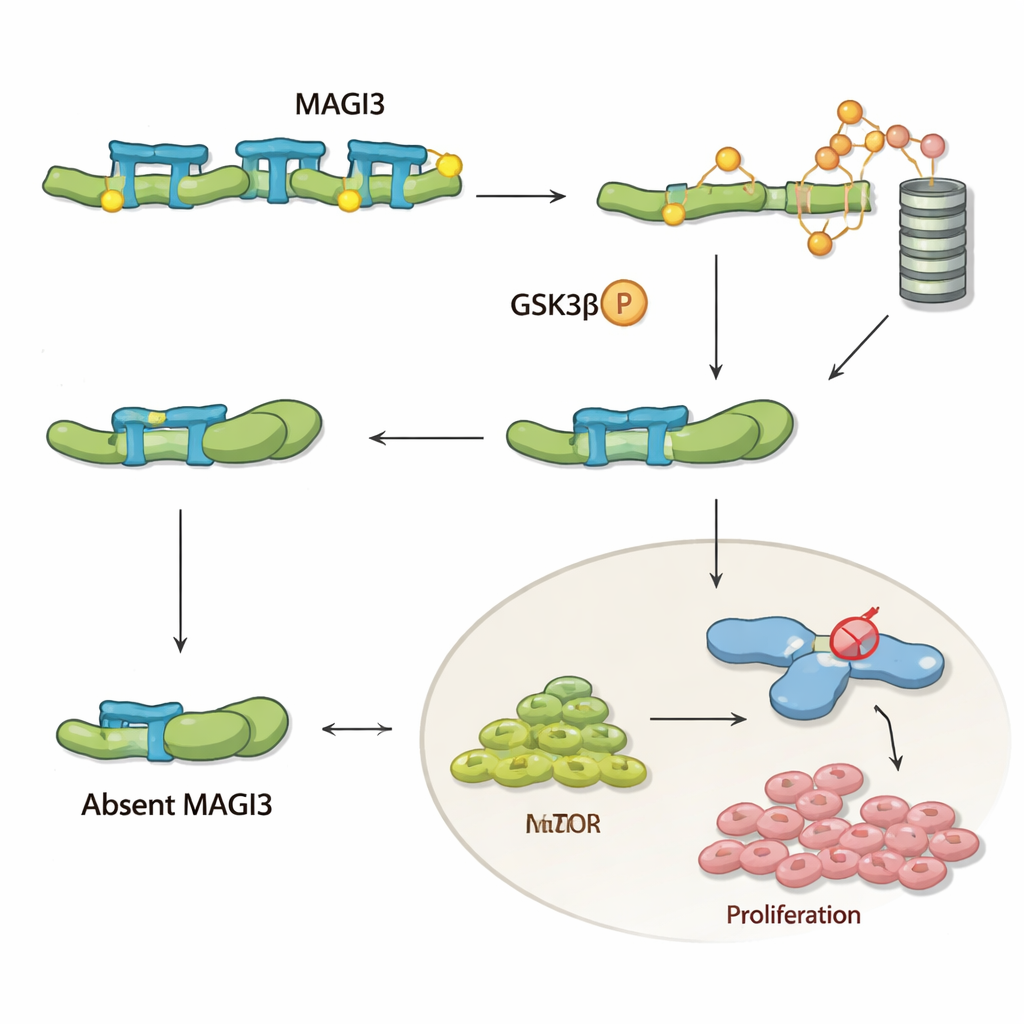
To understand what MAGI3 actually does inside cells, the team focused on β‑catenin, a key player in the Wnt signaling pathway that promotes cell growth, survival, and movement. In many cancers, β‑catenin accumulates and drives disease, but in clear cell kidney cancer it is rarely mutated, suggesting that its control systems are altered instead. The authors show that MAGI3 physically binds to the tail end of β‑catenin and forces the protein into a more “open” shape. This exposes sensitive sites at the opposite end of β‑catenin, allowing another enzyme, GSK‑3β, to tag it for destruction by the cell’s waste disposal machinery. When MAGI3 is present, β‑catenin is continually marked, dismantled, and kept at safe levels; when MAGI3 is missing, β‑catenin stays folded, escapes destruction, builds up in the cell, and switches on genes that help tumors grow and spread.
From cell movement to drug resistance
In kidney cancer cell lines, boosting MAGI3 reduced migration and invasion in lab assays, while lowering MAGI3 had the opposite effect. When MAGI3‑deficient tumor cells were injected into mice, they formed many more lung metastases, directly linking loss of this protein to aggressive behavior. The team then asked how this pathway intersects with current treatments. Many patients with advanced clear cell kidney cancer receive mTOR inhibitors (often called rapalogs), but responses are limited and resistance is common. Here, MAGI3 loss made cancer cells much less sensitive to the mTOR inhibitor Everolimus, increasing the drug concentration needed to slow growth several‑fold. Crucially, blocking Wnt/β‑catenin signaling in these MAGI3‑deficient cells restored their sensitivity to Everolimus and sharply curtailed their ability to invade.
Evidence from patients and animal models
To test whether these laboratory findings translate to real patients, the researchers examined tumors from 53 people with metastatic clear cell kidney cancer treated with Everolimus. Those whose disease was controlled had high MAGI3 and low β‑catenin protein in their tumors, while patients whose cancer progressed quickly showed the reverse pattern. Low MAGI3 predicted shorter overall survival in this group. In mice bearing MAGI3‑low tumors, Everolimus alone or a Wnt‑pathway drug alone only modestly slowed growth. However, combining the two treatments dramatically shrank tumors without causing noticeable side effects, suggesting that dual targeting of these pathways can overcome the resistance driven by MAGI3 loss.
What this means for future care
This study reveals MAGI3 as a crucial internal brake that keeps the Wnt/β‑catenin growth switch under control in clear cell kidney cancer. When MAGI3 is lost, β‑catenin escapes destruction, fueling metastasis and making tumors less responsive to mTOR‑targeted drugs. Measuring MAGI3 and β‑catenin in tumor samples could therefore help doctors identify patients more or less likely to benefit from Everolimus. Even more importantly, the work suggests that combining mTOR inhibitors with drugs that dampen Wnt/β‑catenin signaling may offer a powerful new strategy for patients with aggressive, treatment‑resistant disease.
Citation: Gu, S., Wang, H., Liu, H. et al. MAGI3 deficiency unleashes β-catenin conformational change to drive metastatic progression and mTOR inhibitor resistance in ccRCC. Cell Death Dis 17, 372 (2026). https://doi.org/10.1038/s41419-026-08563-x
Keywords: clear cell renal cell carcinoma, MAGI3, beta-catenin, mTOR inhibitor resistance, Wnt signaling