Clear Sky Science · en
14-3-3γ Knockdown promotes matrix mineralization in human mesenchymal stromal cells
Why stronger bones start in unexpected places
Our bones are constantly rebuilt throughout life, and tiny shifts in how bone-forming cells behave can mean the difference between healthy skeletons and fragile bones. This study looks at a little-known cellular helper protein, called 14-3-3γ, and shows that dialing it down in human fat-derived stem cells actually makes them build a richer, more mineralized bone-like matrix. Understanding this internal brake system could open new paths for treating bone loss and improving bone repair.
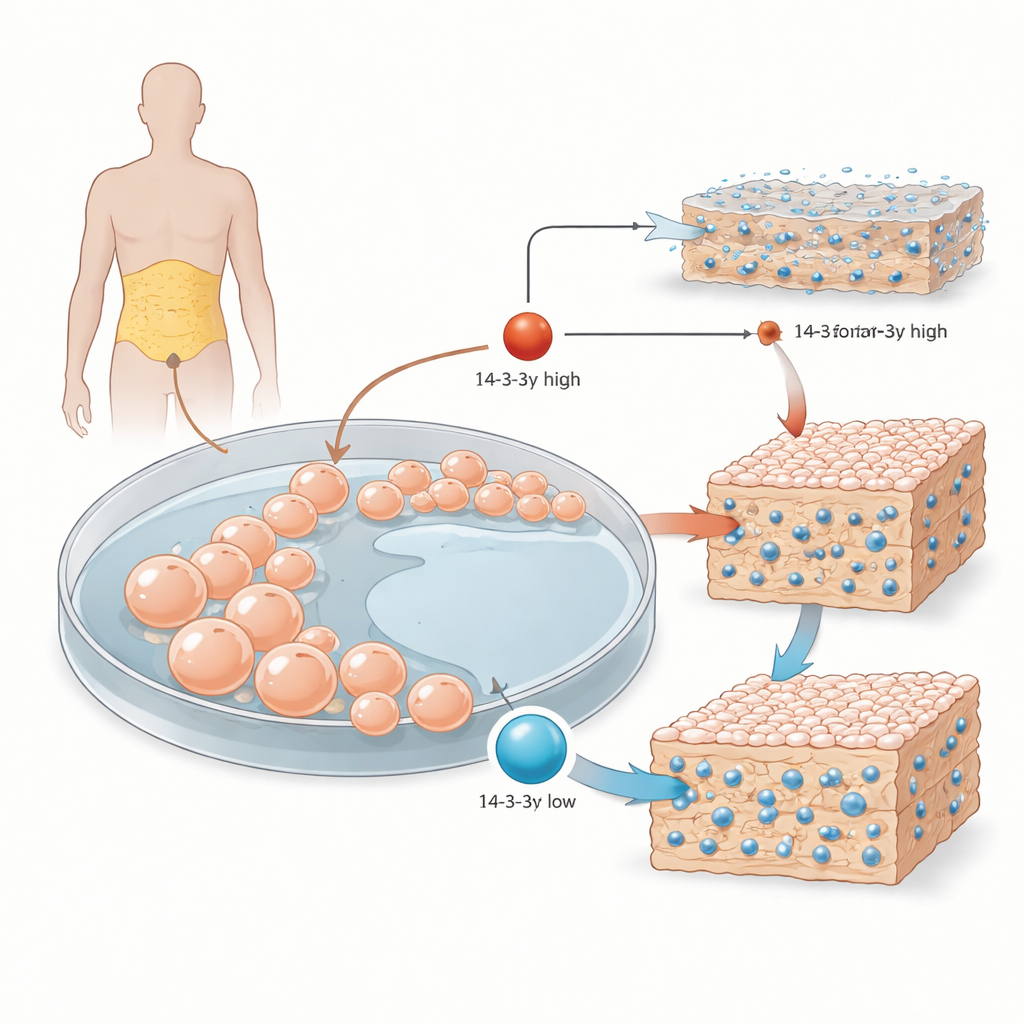
From body fat to bone-building cells
The researchers began with mesenchymal stromal cells taken from human fat tissue. These versatile cells can turn into bone, cartilage, or fat, depending on the signals they receive. In the lab, the team exposed them to a standard bone-inducing cocktail and followed three main stages: early growth, maturation of the supporting scaffold around the cells, and final hardening of that scaffold with mineral deposits. They focused on 14-3-3γ, one member of a family of proteins that act as molecular organizers, binding many different partners and steering their activity and location inside the cell.
A shifting brake during bone formation
As the stem cells were pushed toward becoming bone cells, 14-3-3γ levels did not stay constant. During the middle phase, when the cells ramp up production of collagen and other matrix components, 14-3-3γ levels rose. Later, when the matrix begins to harden by taking up calcium and phosphate, its levels fell. This wave-like pattern suggested that 14-3-3γ might fine‑tune different steps of bone formation rather than serving a single fixed role.
Turning the knob down boosts hardening
To test this idea, the team used engineered viruses to either lower or increase 14-3-3γ inside the cells. When 14-3-3γ was reduced, an early bone marker enzyme called alkaline phosphatase became more active, and by the time the cultures reached the hardening phase, the surrounding matrix contained more calcium crystals and more collagen fibers. In contrast, forcing cells to produce extra 14-3-3γ had the opposite effect: the matrix became poorer in minerals and collagen. Interestingly, some classic genetic switches of bone formation, such as the RUNX2 protein and the genes for osteocalcin and osteopontin, did not simply rise and fall in lockstep with 14-3-3γ, suggesting this protein acts through more subtle routes than just turning major genes on or off.
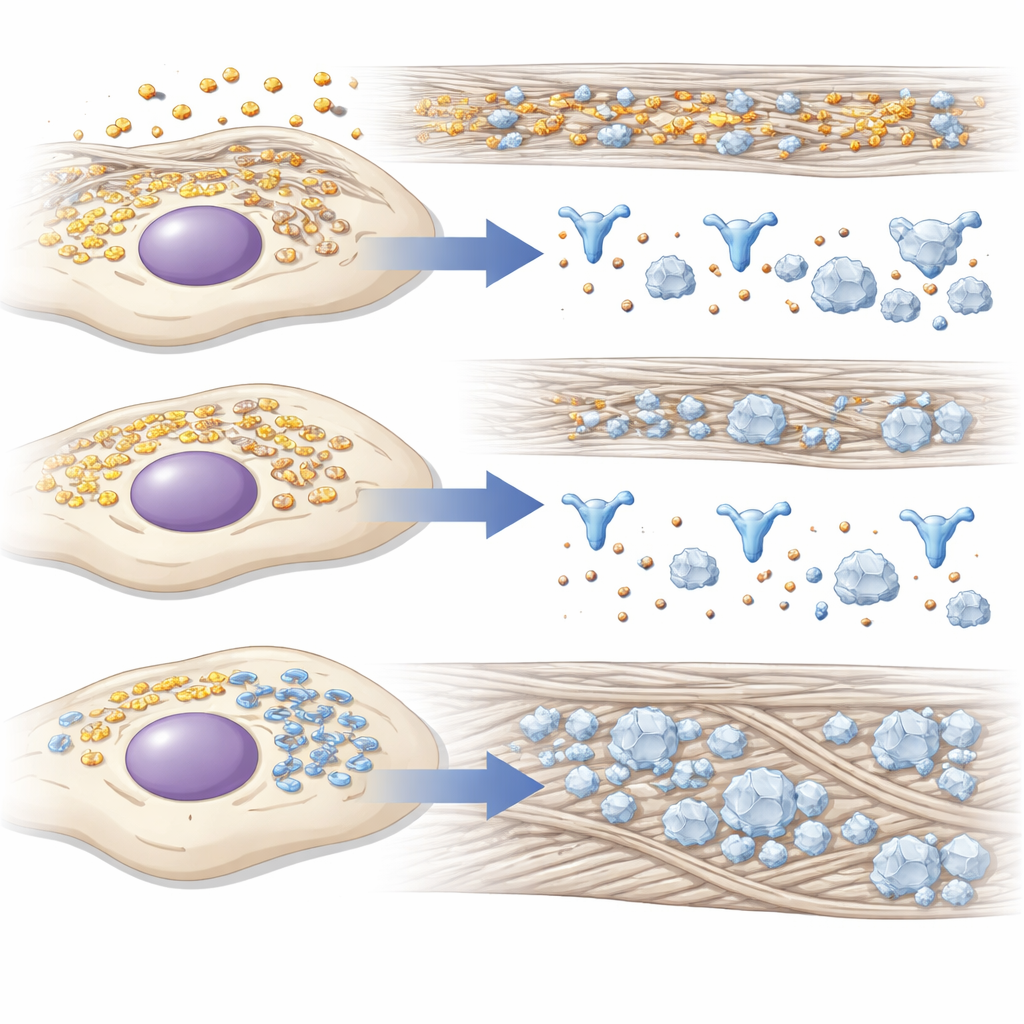
Clues from the cell’s factory floor
Because building bone requires massive protein production and careful handling of calcium and phosphate, the researchers looked at global protein changes when 14-3-3γ was knocked down. Using mass spectrometry, they found that proteins linked to energy use and responses to low oxygen tended to decrease, while those tied to protein folding, stress in the cell’s internal membrane network, and bone development tended to increase. Many of these upregulated proteins help fold and process type I collagen or organize it outside the cell, creating a better scaffold for mineral deposits. Microscopy added another layer: during bone induction, 14-3-3γ moved from a diffuse distribution to concentrate near the endoplasmic reticulum, the cell’s main protein factory, hinting that it helps manage traffic and quality control there.
What this means for future bone therapies
Taken together, the results paint 14-3-3γ as a built‑in brake on the final hardening of the bone matrix. When its levels are appropriately lowered at the right time, stem cells deposit more collagen and mineral, maturing more fully into bone‑forming cells. When its levels remain high, this maturation is held back. Because members of the 14-3-3 family can already be targeted by small molecules, learning how to selectively tune 14-3-3γ in human stem cells could eventually contribute to treatments for osteoporosis, slow-healing fractures, or other disorders where bone remodeling goes awry, while also reminding us that even tiny molecular organizers can have big effects on skeletal health.
Citation: Rivera, L., Müller, S., Uhart, M. et al. 14-3-3γ Knockdown promotes matrix mineralization in human mesenchymal stromal cells. Cell Death Dis 17, 415 (2026). https://doi.org/10.1038/s41419-026-08540-4
Keywords: bone mineralization, mesenchymal stem cells, osteogenesis, scaffold collagen, cell signaling proteins