Clear Sky Science · en
Dual inhibition of mTOR and HSP90 enhances cisplatin efficacy and overcomes resistance in ovarian cancer
Why this research matters
Epithelial ovarian cancer is one of the deadliest gynecologic cancers, largely because many tumors eventually stop responding to standard chemotherapy based on the platinum drug cisplatin. Once this resistance appears, treatment options are limited and survival drops sharply. This study explores a new way to make stubborn tumors sensitive to cisplatin again by hitting two key internal stress and growth systems in cancer cells at the same time.
When standard drugs stop working
Many common cancers, including ovarian, lung, and head and neck cancers, are treated with cisplatin and related platinum-based drugs. These drugs work mainly by damaging the DNA of rapidly dividing cells, pushing them toward self-destruction. Over time, however, cancer cells can adapt. They pump the drug out more efficiently, repair DNA damage more effectively, or switch off death signals, leading to so-called platinum resistance. For women with ovarian cancer, more than half of those who initially respond well to treatment eventually relapse with resistant disease, for which effective therapies are scarce.
Finding hidden switches inside cancer cells
To understand what changes inside resistant ovarian cancer cells, the researchers used a technique called phosphoproteomics, which maps thousands of chemical “on/off” switches on proteins. Comparing platinum-sensitive and platinum-resistant ovarian cancer cell lines, they found 542 proteins that differed between the two. Two major control hubs stood out: the mTOR signaling pathway, which drives cell growth and survival, and a stress-response pathway centered on the factor HSF1 and its helper proteins HSP90, HSP70, and HSP40, which act like chaperones that stabilize many other proteins. In resistant cells, these systems were more active and more heavily switched on than in their sensitive counterparts, and similar patterns appeared in platinum-resistant lung cancer cells and in primary cells from patients.
Turning stress helpers into an Achilles’ heel
The team then tested whether shutting down these overactive hubs could expose a weakness in resistant tumors. They focused on HSP90, a key chaperone, and mTOR, a central growth regulator. Using genetic engineering, they showed that removing HSP90 made resistant lung cancer cells more vulnerable to cisplatin, while forcing extra HSP90 into previously sensitive ovarian cancer cells made them harder to kill and less prone to DNA damage. These experiments supported the idea that platinum resistance depends, at least in part, on boosted HSP90 and mTOR activity and that resistant cells may be unusually dependent on these pathways to survive ongoing drug stress.
A triple-drug strategy to re-sensitize tumors
Building on these insights, the researchers tried a three-pronged treatment: cisplatin combined with ganetespib, an HSP90 inhibitor, and temsirolimus, an mTOR inhibitor already approved for some cancers. In multiple ovarian cancer models grown in dishes, in three-dimensional microtissues that better mimic real tumors, and in mice bearing resistant tumors, this triple combination showed strong synergy. It sharply reduced cancer cell growth, almost eliminated colony formation, and caused marked shrinkage of tumor-like microtissues. In mice with cisplatin-resistant ovarian or lung cancer grafts, the three-drug regimen nearly halted tumor growth, cut overall tumor burden by about 85% in one model, and significantly prolonged survival, all with acceptable weight loss and no major signs of toxicity.
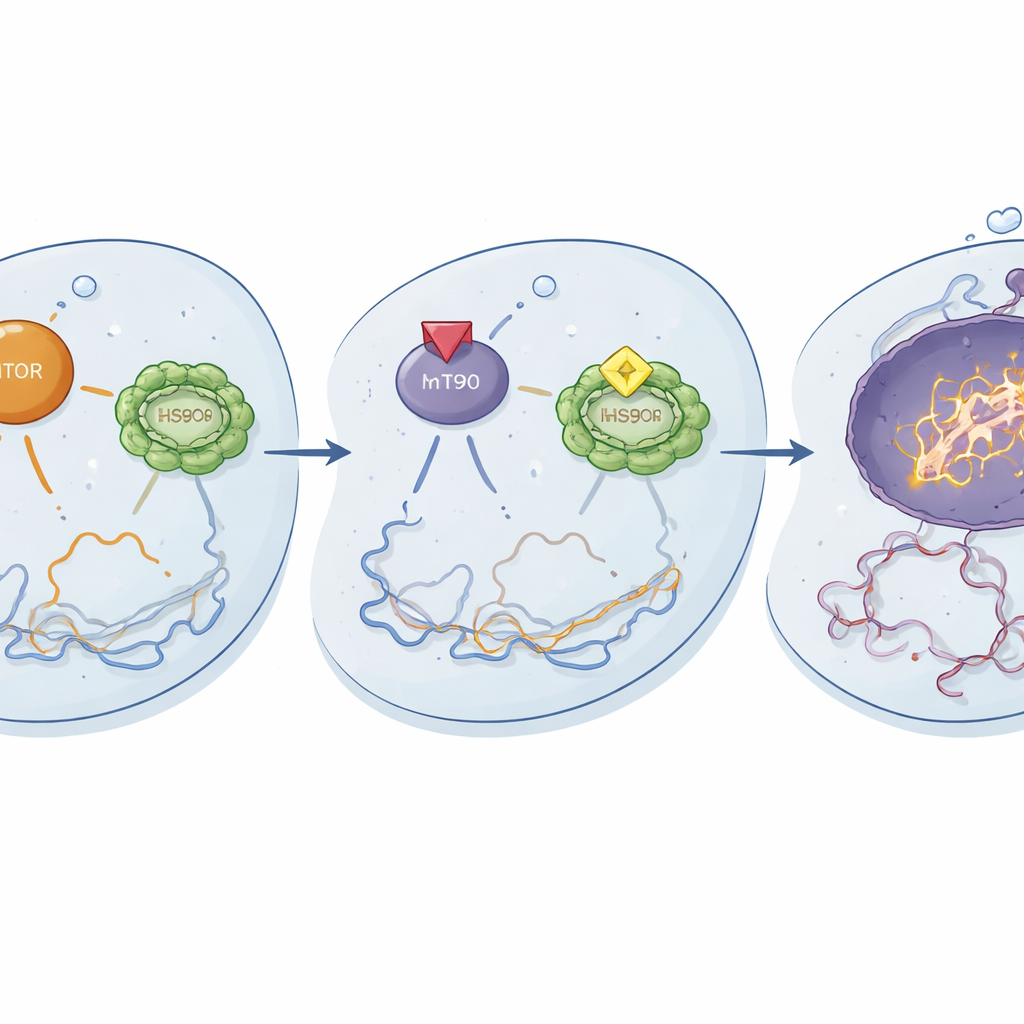
How the new combination works inside the cell
Mechanistic studies revealed what was happening at the molecular level. When used together, ganetespib and temsirolimus dampened the activation of key components of the mTOR pathway and reduced the activity of HSF1 and its chaperone partners, including HSP90. This dual shutdown seemed to dismantle important survival and stress-handling circuits in resistant cells. As a result, cisplatin caused more intense DNA damage, as shown by increased DNA break markers, and triggered stronger apoptosis, the controlled self-destruct program. Early phosphoprotein changes appeared within 24 hours and became more pronounced by 48 hours, suggesting that the triple therapy quickly rewires signaling networks away from survival and toward cell death.
What this could mean for patients
In practical terms, this work proposes a new way to tackle cancers that no longer respond to platinum chemotherapy by exploiting their reliance on mTOR signaling and HSP90-driven stress protection. While earlier trials of HSP90 or mTOR inhibitors alone have shown limited benefit, the evidence here suggests that combining both with cisplatin may convert a previously resistant tumor back into one that is vulnerable to treatment. The authors argue that future clinical studies should test this triple approach in patients with platinum-resistant ovarian and other cancers, ideally selecting those whose tumors show high activity in these pathways. If successful, such a strategy could open a new route to extend the lives of patients for whom current options are few.
Citation: Lombardi, R., Addi, L., Pucci, B. et al. Dual inhibition of mTOR and HSP90 enhances cisplatin efficacy and overcomes resistance in ovarian cancer. Cell Death Dis 17, 417 (2026). https://doi.org/10.1038/s41419-026-08533-3
Keywords: ovarian cancer, cisplatin resistance, mTOR inhibition, HSP90 inhibitors, combination therapy