Clear Sky Science · en
WTAP stabilized by USP7 contributes to enzalutamide resistance in prostate cancer via mediating AKT m6A-modification
Why some prostate cancers outsmart a key drug
Enzalutamide is a widely used pill that helps control advanced prostate cancer by shutting down the male hormone signals that feed tumor cells. Many patients initially respond well, but over time the cancer often learns to grow again despite the drug. This study uncovers a molecular "trick" that tumor cells use to escape enzalutamide, and points to new ways doctors might block this escape route.
A growth helper hiding in plain sight
The researchers focused on a protein called WTAP, already known to be involved in regulating how genetic messages are processed inside cells. By examining tissue samples from 60 men with prostate cancer, and comparing them with nearby noncancerous tissue, they found that WTAP levels were consistently higher in tumors. When they artificially increased WTAP in prostate cancer cells grown in the lab, the cells formed more colonies and, when implanted into mice, produced faster-growing tumors. Conversely, dialing down WTAP slowed tumor cell growth. These results show that WTAP acts as a growth helper for prostate cancer.
How hormone signals boost this growth helper
Prostate cancer is strongly driven by the androgen receptor, a protein that senses male hormones and switches many genes on or off. The team discovered that WTAP itself is one of the genes controlled by this receptor. When they activated the androgen receptor with a hormone-like compound, WTAP levels rose; when they blocked or reduced the receptor, WTAP fell. Detailed DNA-binding and reporter experiments showed that the receptor can latch directly onto the WTAP gene’s control region and turn it up. In other words, standard hormone signals encourage tumor growth in part by raising WTAP.
When blocking hormones is no longer enough
Enzalutamide is designed to silence the androgen receptor, and early in treatment it indeed lowers WTAP levels. But the story changes in drug‑resistant cells. In prostate cancer cells that had been exposed to enzalutamide for months and become resistant, WTAP levels rebounded and were again high, even though hormone signaling was still blocked. The researchers traced this rebound to another protein, USP7, which acts like a molecular bodyguard. Normally, WTAP can be tagged for destruction and broken down; in resistant cells, USP7 binds WTAP and removes these tags, preventing its disposal and allowing WTAP to accumulate. Inhibiting USP7 reduced WTAP levels and increased the chemical marks that signal WTAP for breakdown.
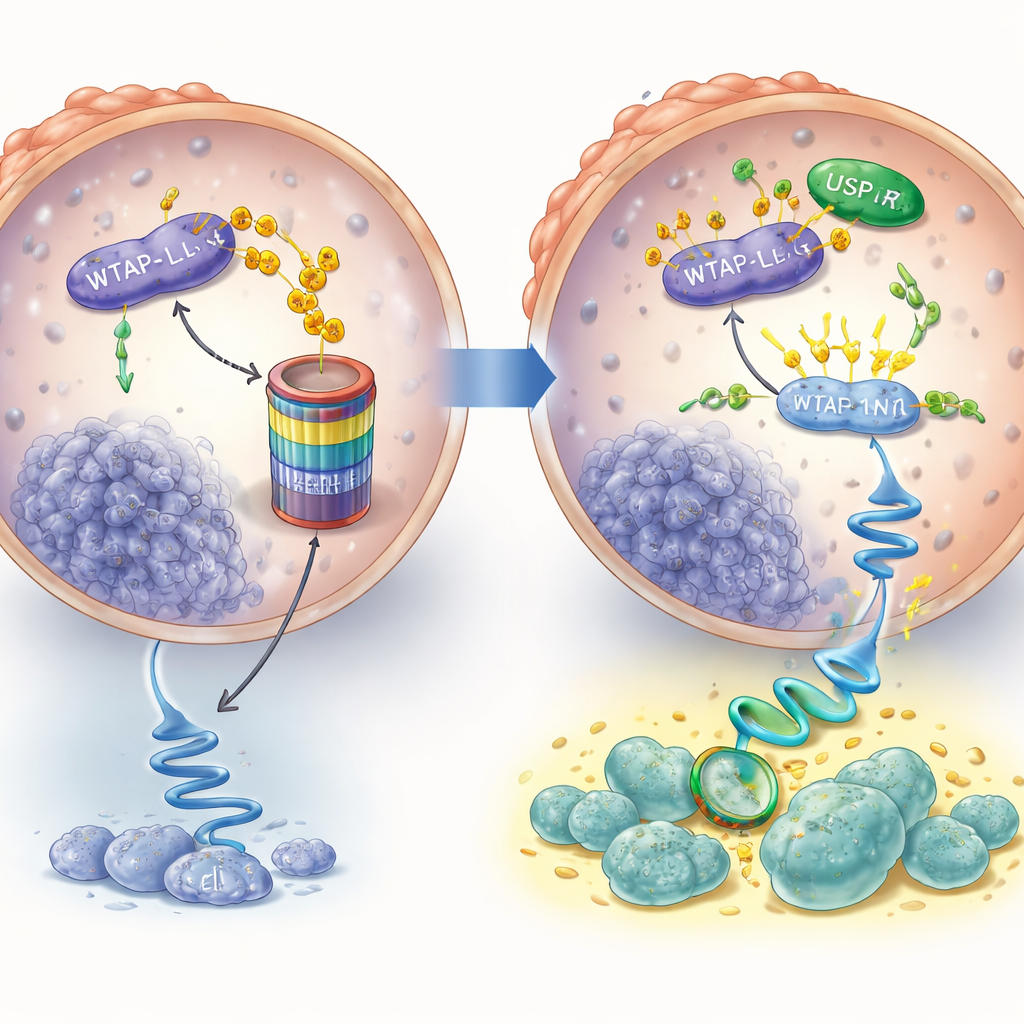
Rewiring messages to switch on a survival pathway
High WTAP levels would matter little unless they changed how cancer cells behave. The team used large‑scale RNA analysis and found that a major growth and survival pathway controlled by a protein called AKT was especially active in resistant cells. They showed that WTAP helps modify the genetic message for AKT in a way that makes this message more stable, so cells produce more AKT protein. When WTAP or its partner enzymes were blocked, AKT levels dropped and the message decayed faster. Functionally, extra WTAP allowed cancer cells to keep proliferating even in the presence of enzalutamide, but adding an AKT‑blocking drug erased this advantage. Similarly, reducing WTAP or blocking AKT made resistant cells more sensitive to enzalutamide.
What this means for patients
Taken together, the work outlines a chain of events: prostate cancer cells raise WTAP to fuel growth; long‑term enzalutamide treatment removes the initial hormone control over WTAP, but resistant cells compensate by using USP7 to protect WTAP from destruction; in turn, WTAP helps stabilize messages that boost AKT, a key survival switch, allowing tumors to ignore the drug. For patients, this suggests that measuring WTAP or USP7 might help identify cancers likely to resist enzalutamide, and that drugs targeting USP7 or AKT, combined with hormone‑blocking therapy, could offer a more durable way to keep advanced prostate cancer in check.
Citation: Shi, R., Gu, K., Li, H. et al. WTAP stabilized by USP7 contributes to enzalutamide resistance in prostate cancer via mediating AKT m6A-modification. Cancer Gene Ther 33, 277–288 (2026). https://doi.org/10.1038/s41417-026-01013-y
Keywords: prostate cancer, drug resistance, enzalutamide, AKT signaling, RNA modification