Clear Sky Science · en
From gut to brain: effects of fecal microbiota transplants from humans to rats on hippocampal gene regulation - a study on anorexia nervosa
Why Your Gut Might Matter to Your Mind
Anorexia nervosa is often seen as a disorder of thoughts and feelings about food and body image, but mounting evidence suggests that tiny organisms in our intestines may also play a role. This study explores whether gut bacteria from people with anorexia can influence brain biology when transferred into rats. By tracking changes in the animals’ gut communities and a memory‑critical brain region called the hippocampus, the researchers ask a simple but far‑reaching question: can remixing the microbiome help—or harm—the brain in eating disorders?
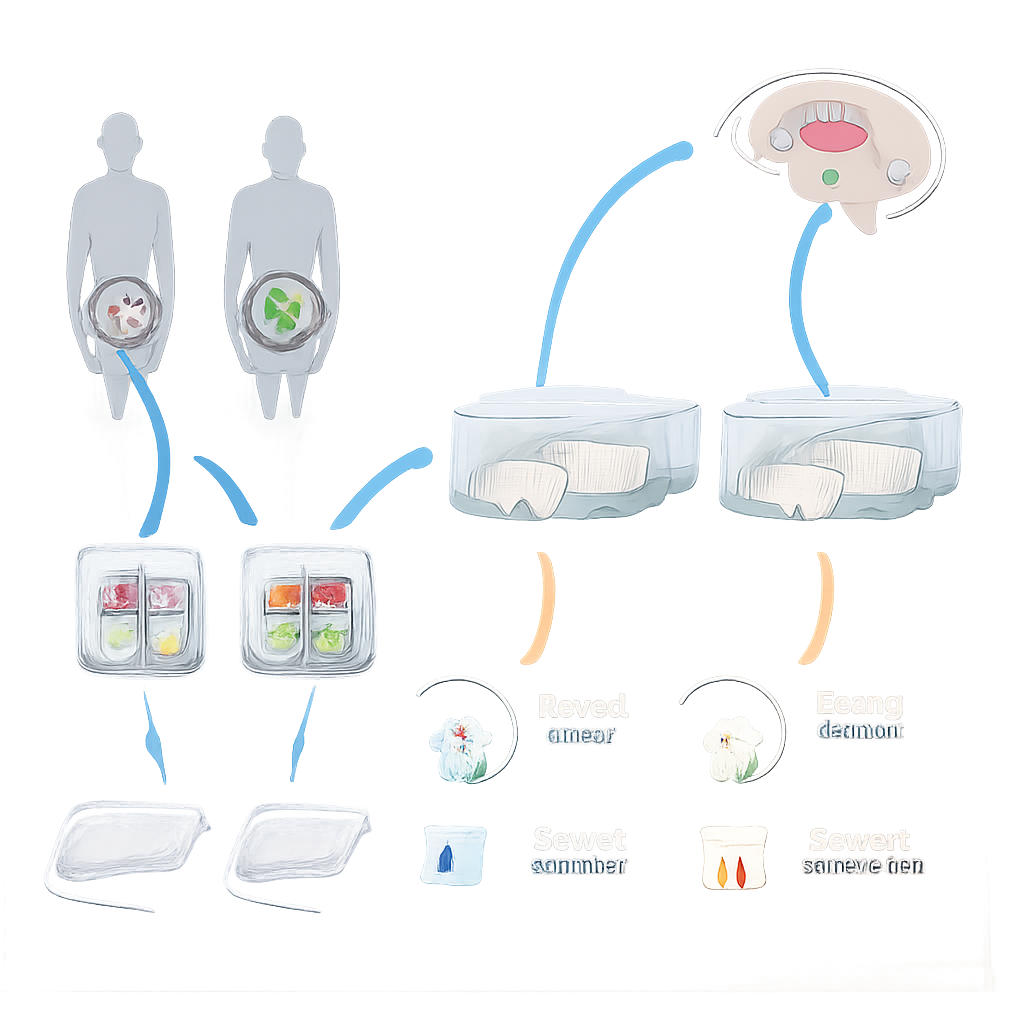
Moving Microbes from People to Rats
The team worked with stool samples from adolescent girls with anorexia and from healthy peers of similar age and sex. These samples were processed to preserve living bacteria and then frozen. Young female rats first received a cocktail of antibiotics in their drinking water for a week to dramatically reduce their own gut bacteria. After this “clear‑out” phase, some rats were given repeated fecal microbiota transplants (FMT) from healthy donors, some from donors with anorexia, and others only water. All animals had unrestricted access to food, water, and running wheels, and the scientists monitored their weight, food intake, and activity for several weeks.
What Changed in the Gut
Antibiotic treatment strongly disturbed the rats’ gut ecosystems, lowering overall microbial biomass and shifting which bacterial groups were present. Over time, FMT partially rebuilt these communities: human‑derived bacteria successfully took hold in the rats, especially in those receiving healthy‑donor stool. Still, the transfer was far from complete—only a modest fraction of human bacterial types established themselves. Importantly, despite these microbiome shifts, rats receiving stool from patients with anorexia did not develop classic disease‑like features: their body weight, food intake, and running activity remained similar to all other groups.
What Changed in the Brain
To see how gut changes might affect the brain, the researchers examined the hippocampus, a region essential for learning, memory, and flexible thinking—abilities often impaired in anorexia. They measured both the presence of key brain cell types and the activity of genes tied to cell growth, support cells, and inflammation. Antibiotics alone lowered activity of genes linked to oligodendrocytes, cells that help insulate nerve fibers. They also reduced expression of Bdnf, a growth factor crucial for maintaining healthy brain circuits, and dampened a marker of new cell production (Mki67). When rats received FMT from healthy donors, these negative effects were largely reversed: Bdnf and Mki67 expression rebounded, and inflammatory signaling molecules in the hippocampus increased in a pattern consistent with active repair and remodeling. In contrast, FMT from donors with anorexia failed to fully restore these measures, leaving growth‑related and inflammatory signals comparatively blunted.
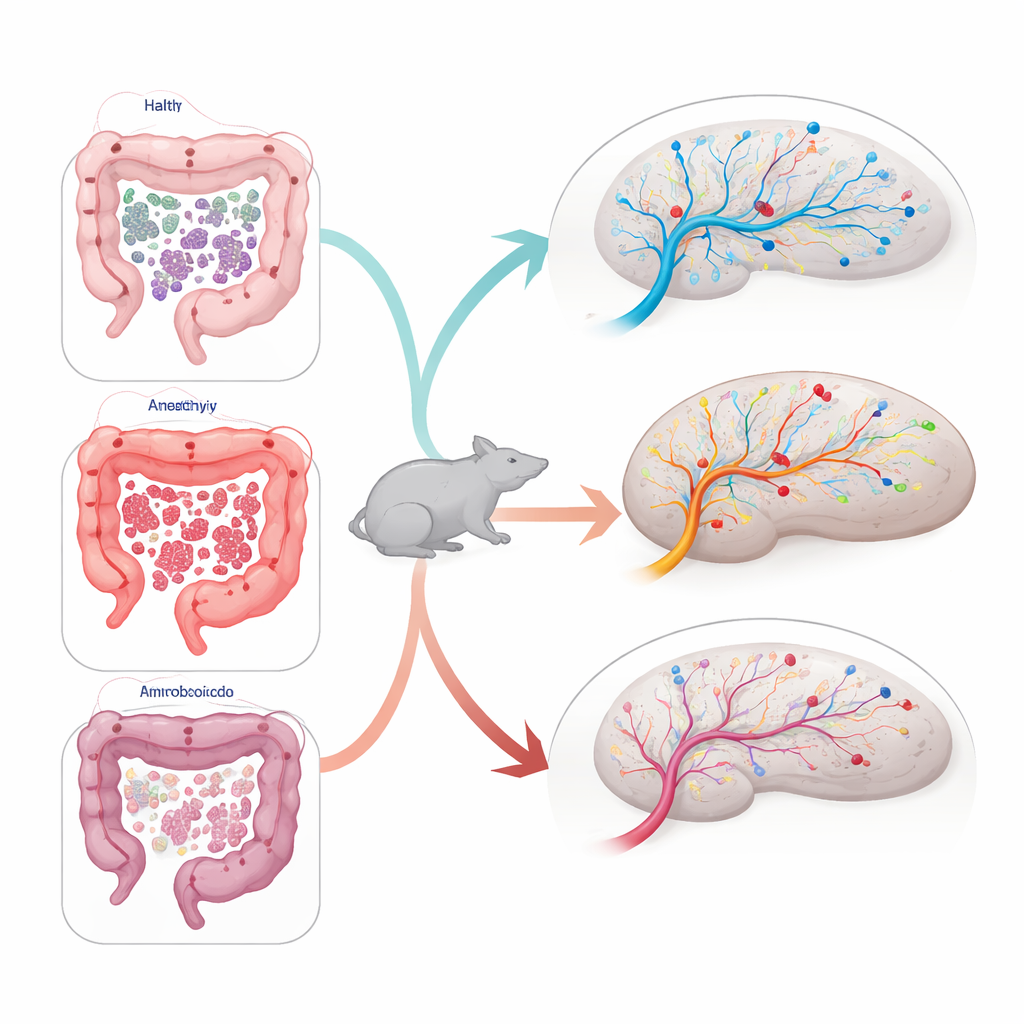
Links Between Bacteria and Brain Signals
Digging deeper, the scientists explored how specific bacterial groups related to brain gene activity. Certain genera that have been implicated in mood, metabolism, or inflammation—such as Akkermansia, Blautia, Prevotella, and Butyricicoccus—showed notable associations with markers of neuroplasticity and immune signaling in the hippocampus. For example, some bacteria were tied to higher expression of genes involved in inflammation and cell proliferation, while others showed the opposite pattern. These patterns differed between rats that received stool from healthy donors and those that received stool from patients with anorexia, hinting that distinct microbial “signatures” may nudge brain repair processes in different directions.
What This Means for Anorexia and Future Treatments
The study shows that while fecal transplants from people with anorexia do not, by themselves, recreate the disorder in rats, they do shape how the hippocampus responds to a major disruption of the microbiome. Antibiotics appeared to dampen brain plasticity and supportive cell function, and only healthy‑donor microbiota reliably restored growth‑ and inflammation‑related signals. In plain terms, the gut community seems able to push the brain toward or away from recovery after a hit. These findings support the idea that anorexia is not purely a psychological illness but also involves a gut–brain loop. They point toward a future in which tailored microbial mixes—or the beneficial molecules they produce—could complement existing therapies, especially when combined with careful nutritional support and strategies that directly address starvation.
Citation: Korten, N.M., Blischke, L., Thelen, A.C. et al. From gut to brain: effects of fecal microbiota transplants from humans to rats on hippocampal gene regulation - a study on anorexia nervosa. Transl Psychiatry 16, 238 (2026). https://doi.org/10.1038/s41398-026-04056-9
Keywords: anorexia nervosa, gut microbiome, fecal microbiota transplant, hippocampus, neuroinflammation